| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Review
Volume 14, June 2021, pages 53-59
Recent study on the anticancer activity of nobiletin and its metabolites
Ke Lva, b, Lingling Zhanga, b, *, Hui Zhaoa, *, Chi-Tang Hoc, Shiming Lib
aTianjin Key Laboratory of Food and Biotechnology, School of Biotechnology and Food Science, Tianjin University of Commerce, Tianjin 300134, China
bHubei Key Laboratory of EFGIR, Huanggang Normal University, Huanggang, Hubei, 438000, China
cDepartment of Food Science, Rutgers University, New Brunswick, New Jersey 08901, United States
*Corresponding author: Lingling Zhang and Hui Zhao, Tianjin Key Laboratory of Food and Biotechnology, School of Biotechnology and Food Science, Tianjin University of Commerce, Tianjin 300134, China; Hubei Key Laboratory of EFGIR, Huanggang Normal University, Huanggang, Hubei, 438000, China. E-mail: 1243536645@qq.com (LZ), zhaohui@tjcu.edu.cn (HZ)
DOI: 10.31665/JFB.2021.14267
Received: June 25, 2021
Revised received & accepted: June 30, 2021
| Abstract | ▴Top |
The exploration of naturally occurring phytochemicals with antitumor potential has been the focus of many studies. Nobiletin, a polymethoxyflavone (PMF) exclusively derived from citrus peels, has been reported as a promising candidate for the prevention and/or treatment of cancers. Additionally, multiple demethylated derivatives of nobiletin from in vivo biotransformation, including 3′-demethylnobiletin (3′-DMN), 4′-demethylnobiletin (4′-DMN), 3′,4′-didemethylnobiletin (3′,4′-DDMN) and 5-demethylnobiletin (5-DMN), among others, have been found to show anti-cancer activity. In this review, the anticancer activity of nobiletin and its derivatives in cancer prevention are comprehensively described.
Keywords: Nobiletin; Cancer; Anti-Inflammation; Apoptosis; Autophagy
| 1. Introduction | ▴Top |
Cancer is becoming a major public concern because it is a leading cause of death worldwide. According to a recent statistic from the American Cancer Society, 1,898,160 new cancer cases and 608,570 cancer deaths are projected to occur in the United States in 2021 (Siegel et al., 2021). As a result of improvements in early detection and treatment, the cancer death rate dropped by 31% from its peak in 1991 until 2018 (Siegel et al., 2021). In recent years, attention has been focused on finding effective and low toxic anticancer agents as the alternative and complementary medicine for cancer treatment and prevention. Notably, accumulating data suggest that compounds from natural plants have been used to target cancer for decades because they usually have fewer side effects than non-natural anticancer drugs.
Nobiletin (5,6,7,8,3′,4′-hexamethoxyflavone, Figure 1) is a natural flavonoid extracted exclusively from citrus peel (Wang et al., 2018) and exhibits a good safety profile and a broad spectrum of pharmacological activities, including anti-inflammatory (Ozkan et al., 2020; Hagenlocher et al., 2019; Yang et al., 2019), antitumor (Ashrafizadeh et al., 2020) and neuroprotective activities (Amarsanaa et al., 2021). Not only does nobiletin itself exhibit anti-cancer properties, but also its in vivo metabolites, including 3′-demethylnobiletin (3′-DMN), 4′-demethylnobiletin (4′-DMN), 3′,4′-didemethylnobiletin (3′,4′-DDMN) and 5-demethylnobiletin (5-DMN) also have anti-cancer activity (Figure 1).
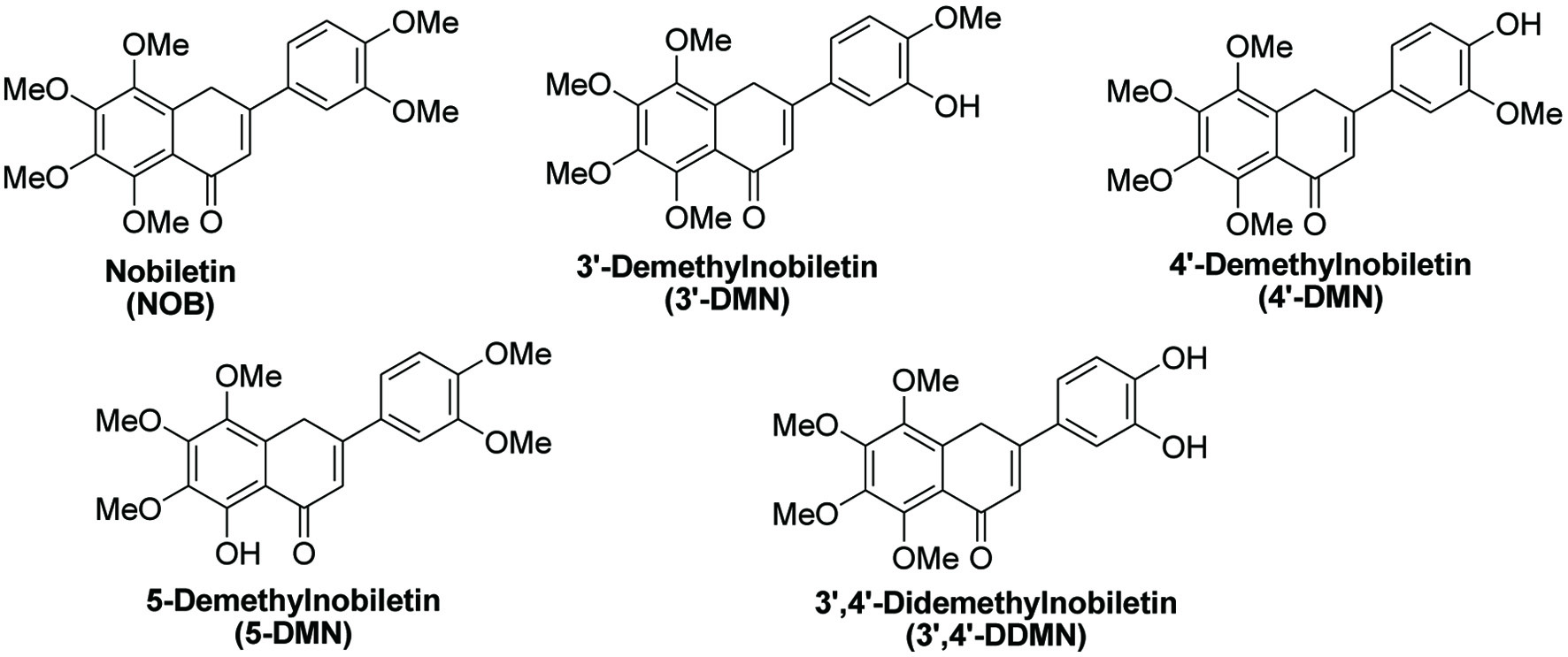 Click for large image | Figure 1. Structures of nobiletin and its in vivo metabolites. |
In this review, we summarize the anticancer activity of nobiletin and its derivatives in cancer therapy based on the newly published articles.
1.1. Chemical aspects of nobiletin and its derivatives
Recently, nutritionists have shown interest in natural compounds extracted from plants and fruits in the prevention and treatment of various illnesses including cancer. Nobiletin can be extracted exclusively from citrus fruits, particularly in the peels of sweet oranges (Citrus sinensis) (Li, Yu and Ho, 2006) and mandarin oranges (Citrus reticulate) (Nogata et al., 2006) through different types of chemical extraction processes. There is concurrently ongoing research looking into the effect of nobiletin on different types of cancers, such as leukemia (Yen et al., 2020), colon cancer (Goh et al., 2019), breast cancer (Surichan et al., 2018), ovarian cancer (Zhang et al., 2020), thyroid cancer (Sousa et al., 2020), lung cancer (Da et al., 2016), and other tumor types. The metabolism of nobiletin comprises two phases, including phase I and phase II metabolism. Three common metabolites of nobiletin include 3′-DMN, 4′-DMN, and 3′,4′-DDMN (Ashrafizadeh et al., 2020). Under long-term storage, nobiletin can be degraded to 5-DMN through the process of microbial autohydrolysis (Wang et al., 2018). Figure 1 illustrates the chemical structures of nobiletin and its derivatives.
| 2. The anticancer activity of nobiletin and its derivatives | ▴Top |
Dietary factors play key roles in the development and prevention of various diseases, including cancer. Nobiletin as a prospective anticancer agent for chemoprevention, is known to inhibit different pathways leading to cancer via a number of different mechanisms, which includes inhibiting cell cycle progression, limiting inflammation, inducing apoptosis, preventing angiogenesis and reducing tumor formation. Figure 2 schematizes the main anticarcinogenic pathways of nobiletin, its autohydrolysis product, 5-DMN and its three common metabolites, 3′-DMN, 4′-DMN, and 3′,4′-DDMN, in chemoprevention of different cancers in detail and also are summarized in Table 1.
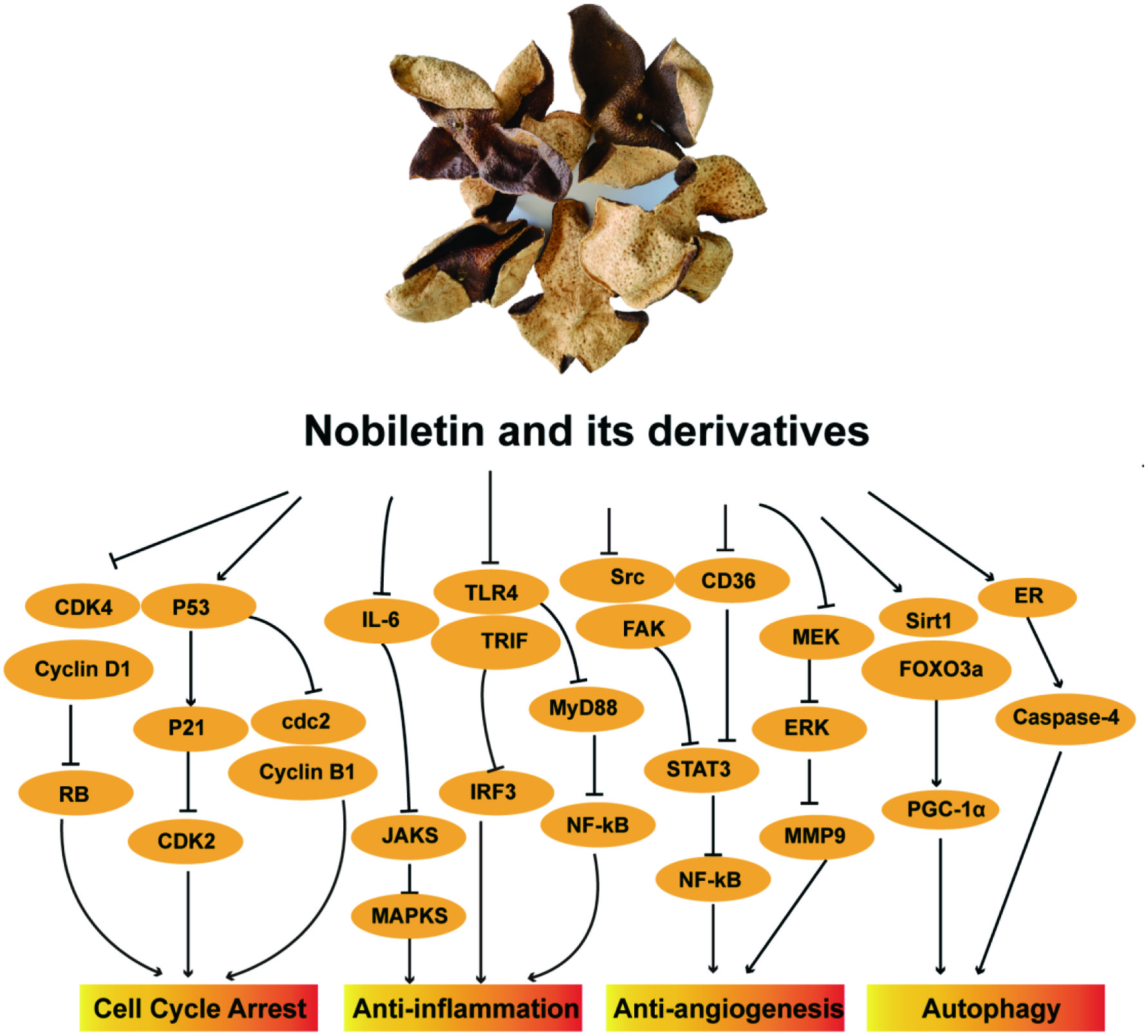 Click for large image | Figure 2. Schematic diagram showing the mechanisms underlying the anti-cancer effects of nobiletin and its derivatives through cell cycle arrest, anti-inflammation, anti-angiogenesis and induction of autophagy. |
 Click to view | Table 1. The anticancer activity of nobiletin and its derivatives |
2.1. Cell cycle arrest
The cell cycle is a complex sequence of events through which a cell duplicates its contents then divides to two genetically identical daughter cells. Mutations and deficiencies in regulation throughout the cell cycle can lead to serious diseases such as cancer (Wenzel and Singh, 2018). Therefore, one way to suppress cancer is to block its cell cycle progression. Studies in recent years elucidated that nobiletin inhibited cancer behavior through cell cycle arrest. For example, Jiang et al. (2020) demonstrated that nobiletin inhibits the growth and metastasis of human pancreatic cancer cells via induction of G0/G1 cell cycle arrest and showed an IC50 of 6.12 µM. In glioma cell, the flow cytometry test showed that 50 and 100 μM nobiletin induced dose-dependently increase in the G0/G1 subpopulation by approximately 8.7 and 17.2%, respectively, compared with the solvent control (0.5% DMSO), indicating that nobiletin can arrest the cell cycle in the G0/G1 phase (Lien et al., 2016). Combining different chemopreventive agents is a promising strategy to reduce cancer incidence and mortality due to potential synergistic interactions between these agents. Co-treatment with 4′-DMN and atorvastatin (a lipoprotein-lowering drug) strongly inhibited the growth of human colon cancer cells by inducing G0/G1 cell cycle arrest and apoptosis (Wu et al., 2018). In HCT116 human colon cancer cells, the combination of curcumin and 3′,4′-DDMN, a primary metabolite of nobiletin, induced significant G2/M cell-cycle arrest and extensive apoptosis, which greatly exceeded the effects of individual treatments with curcumin or 3′,4′-DDMN (DiMarco-Crook et al., 2020). In vitro tests using lung cancer cells revealed 5-DMN, 4′-DMN, and 3′,4′-DDMN inhibited cell proliferation by inducing G2/M cell cycle phase arrest (Chen et al., 2015, Song et al., 2016).
2.2. Anti-Inflammation
Inflammation is the body’s response to tissue damage, caused by physical injury, ischemic injury, infection, exposure to toxins, or other types of trauma. Inflammation is often associated with the development and progression of cancer. The triggers of chronic inflammation that increase cancer risk or progression include infections, autoimmune diseases, and inflammatory conditions of uncertain origin (Singh et al., 2019). In addition, cancer cells as well as surrounding stromal and inflammatory cells engage in well-orchestrated reciprocal interactions to form an inflammatory tumor microenvironment (Greten and Grivennikov, 2019).
Increasing evidence showed that inflammatory cytokines and chemokines, which can be produced by the tumor cells and/or tumor-associated leukocytes and platelets, may contribute directly to malignant progression. Therefore, one way to suppress cancer is to inhibit the secretion of pro-inflammatory cytokines. Ozkan et al. (2020) reported that the anti-inflammatory effect of nobiletin on TLR4/TRIF/IRF3 and TLR9/IRF7 signaling pathways in PCa cells for the first time. Treatment with nobiletin resulted in a noteworthy reduction of cell growth and the release of IFN-α and IFN-β. Besides nobiletin, multiple studies have shown that its metabolites, especially, 4′-DMN, and 3′,4′-DDMN also exhibit significant anti-inflammation effect towards nitric oxide production, iNOS and cyclooxygenase (COX) expressions in cancer (Wu et al., 2017). By administering nobiletin and its metabolites at a concentration equivalent in the LPS-induced RAW 246.7 macrophages decreased iNOS and COX-2 protein expression levels and induced heme oxygenase-1 (HO-1) protein expression (Wu et al., 2017). Additionally, the anti-inflammatory activity of 3′,4′-DDMN can be demonstrated by its effect on iNOS, COX-2, VEGF, matrix metalloproteinase-9 (MMP-9) and ODC expression in TPA-stimulated mouse skin (Lai et al., 2008) . iNOS and COX-2 are frequently regulated by activating the NF-кB signaling pathway (Chun et al., 2004). 3′,4′-DDMN treatment caused a significant reduction of TPA-induced nuclear translocation and phosphorylation of p65/RelA, the functionally active subunit of NF-кB protein (Lai et al., 2008).
2.3. Anti-angiogenesis
Angiogenesis is a biological process in which novel blood vessels are formed, providing tissues with oxygen and nutrients. Folkman (1971) first proposed that angiogenesis was related to tumor growth, forming the hypothesis of targeting angiogenesis to treat cancer. Since then, many efforts have been made to explore the possibility of curing cancer by targeting angiogenesis.
Steroid receptor coactivator (Src) and focal adhesion kinase (FAK) are intracellular tyrosine kinases that play an important role in tumor angiogenesis. Nobiletin inhibited tumor angiogenesis by regulating Src, FAK, and STAT3 signaling through PXN in ER+ breast cancer cells (Sp et al., 2017). CD36 is implicated in tumor metastasis through its roles in fatty acid metabolism. Recent research showed that nobiletin inhibited angiogenesis by suppressing CD36 expression and decreasing the expression of TSP-1 and TGF-β1 (Sp et al., 2018). In the zebrafish angiogenesis model, nobiletin showed anti-angiogenic activity by blocking new blood vessel formation (Lam et al., 2011). Leptin is thought to be a mitogenic factor that leads to the development of colon cancer. Treatment with nobiletin suppressed cell proliferation induced by leptin through inhibition of mitogen-activated protein/extracellular signal-regulated kinase (MEK) 1/2 (Miyata et al, 2008).
2.4. Autophagy
Autophagy plays a dichotomous role in cancer by suppressing benign tumor growth but promoting advanced cancer growth. In the past decade, numerous researches have established autophagy as a potential therapeutic target in cancer.
Hepatic ischemia and reperfusion injury are characterized by impaired autophagy, mitochondrial dysfunction, and subsequent damage of cellular homeostasis after hepatic surgery or transplantation. Treatment with nobiletin (5 mg/kg) ameliorated oxidative damage, inflammation, and cell death caused by hepatic ischemia and reperfusion through the activation of SIRT-1/FOXO3a-mediated autophagy and mitochondrial biogenesis (Dusabimana at al., 2019). Autophagy is a cellular defense mechanism involving the degradation and recycling of cytoplasmic constituents. Nobiletin suppressed autophagic degradation via over-expressing AKT pathway and enhances apoptosis in multidrug-resistant SKOV3/TAX ovarian cancer cells (Jiang et al., 2018). ER stress-induced apoptotic signaling has been studied to identify potential targets for therapeutic intervention in diseases associated with ER stress (Puthalakath et al., 2007). Studies have shown that nobiletin-induced apoptosis in SNU-16 cells was mediated by pathways involving intracellular ER stress-mediated protective autophagy (Moon and Cho, 2016). Notably, different metabolites of NOB work by different mechanisms against different cancer. In vivo tests using xenograft mouse model showed that 5-DMN exhibited more preferential anti-colon cancer effects than NOB via its ability to induce p53-regulated autophagy (Song et al., 2020).
2.5. Reactive oxygen species (ROS)
Extensive research indicated that reactive oxygen species (ROS), including superoxide radical, hydroxyl radical, and hydrogen peroxide, play an important role in cancer. Although low levels of ROS can be beneficial, excessive accumulation can promote cancer. For example, they can activate pro-tumorigenic signaling, enhance cell survival and proliferation, and drive DNA damage and genetic instability (Diao et al., 2016; Prasad et al., 2017; Sarsour et al., 2008). However, excess ROS can also promote anti-tumorigenic signaling, initiating oxidative stress-induced tumor cell death (Liou and Storz, 2010; Roy et al., 2015; Stanicka et al., 2015). Anti-tumorigenic signaling of ROS can be targeted as a therapy in cancer, by the increased production of ROS levels to toxic levels and exhaustion of the antioxidant system capacity causing programmed cell death. Numerous studies from preclinical to clinical models have indicated that compounds from natural plants are rich in antioxidants provided a protective effect in the development of cancer (Li et al., 2020; Wang et al., 2018; Zhang et al., 2018). Pyroptosis is a pro-inflammatory form of programmed cell death that is triggered by a series of cytosolic sensors to induce cancer cell death (Man, Karki and Kanneganti, 2017). Since nobiletin can trigger ROS-mediated pyroptosis, it is considered a promising new anti-ovarian cancer drug candidate (Zhang et al., 2020). In addition, the intracellular ROS level was dose-dependent with the increase of NOB concentration (Yang et al., 2020).
Inhibition of ROS production in tumor cells could well result in suppressed pro-tumorigenic signaling. ROS are also thought to be involved in inflammatory gene expression through the activation of NF-κB signaling pathway (Kabe et al., 2005). Studies have shown that nobiletin inhibited the DNA-binding activity of NF-κB and ROS production in RAW 264.7 cells (Choi et al., 2007). Treatment with nobiletin reduced ROS levels and apoptotic morphological changes and decreased levels of lipid peroxides and glutathione content in H. pylori-infected GES-1 cells (Ouyang, Li and Ling, 2020).
It is becoming increasingly evident that ROS play an important role in carcinogenesis. Such dual roles of ROS indicate their critical importance in cellular homeostasis. Therefore, targeting ROS could be a promising approach for cancer therapeutics.
| 3. Conclusions | ▴Top |
Overall, there is mounting interest in the therapeutic utility of nobiletin and its autohydrolysis product 5-DMN and several metabolites of nobiletin such as 3′-DMN, 4′-DMN, and 3′,4′-DDMN. However, biopharmaceutical limitations including low aqueous solubility, poor permeability across biological barriers and low bioavailability hinder further accessibility for targeting hard-to-reach cancers. Efforts in exploring and developing effective strategies to overcome the biopharmaceutical challenges associated with nobiletin seems to be a promising way to enhance its therapeutic effects.
Acknowledgments
This work was supported by the Grant from Hubei Province, China (GRANT number 2019ABA100).
| References | ▴Top |