| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Original Research
Volume 33, March 2026, pages 73-84
Preparation and characterization of phosphopeptides derived from egg yolk granules
Jiandong Ren†, Jianping Wu*
Department of Agricultural, Food and Nutritional Science, 4-10 Ag/For Building, University of Alberta, Edmonton, AB T6G 2P5, Canada
*Corresponding author: Jianping Wu, Department of Agricultural, Food and Nutritional Science, 4-10 Ag/For Building, University of Alberta, Edmonton, AB T6G 2P5, Canada. E-mail: jwu3@ualberta.ca
DOI: 10.26599/JFB.2026.95033441
Received: December 8, 2025
Revised received & accepted: February 1, 2026
| Abstract | ▴Top |
Phosphorylated peptides derived from egg yolk phosvitin (PPP) have been reported with various physiological functions such as mineral absorption promoting, antioxidant, antimicrobial and anti-inflammatory activities. However, the animal/clinical study and commercial application of PPP was hampered by the difficulties in phosvitin purification and peptide fragmentation. This study introduced a new approach to prepare phosphopeptides from egg yolk granules to avoid difficulties obtaining purified phosvitin. The egg yolk granules were first dephosphorylated by 0.2 N NaOH for 0.5 h and then hydrolyzed by eight enzymes, respectively. Pancreatin produced the highest degree of hydrolysis of 24.2% with a protein recovery of 41.8% and nitrogen to phosphorus atomic ratio of 19.1, which is comparable to commercial casein phosphopeptides. In the pancreatin hydrolysate 15 peptide sequences were characterized from three phosvitin domains: AKTSSSSSSASSTATSSSSSSASSPN (PV 9-34), DEEENDQV (PV 40-47) and SGHLEDDSSSSSSSSVLSKIWG (PV 190-211). In addition, 54 peptides were characterized from lipovitellins, and 26 of them were phosphorylated. This experiment suggests that it is feasible to produce phosphopeptides from granules instead of purified phosvitin.
Keywords: Granules; Phosvitin; Phosphopeptides; Hydrolysis; Peptide sequence
| 1. Introduction | ▴Top |
Bioactive peptides have been discovered from various food proteins with positive effects on the functions of cardiovascular, digestive, immune and nervous systems due to their bioactivities including antioxidant, antimicrobial, antihypertensive, and immunomodulating activities (Daliri et al., 2017). Phosphopeptides are specific peptides cleaved from proteins containing phosphorylated serine or threonine residues. The presence of the cluster of phosphorylated residues plays a key role in these activities by solubilizing minerals or interacting with cell receptors (Lee et al., 2022; Liu et al., 2024). Phosvitin, the major phosphoprotein from egg yolk, has a unique structure whereby nearly all the serine residues, accounting for half of the amino acids in phosvitin, are phosphorylated (Marcet et al., 2022). Previously, phosvitin derived phosphopeptides were studied for their antioxidant, anti-inflammation, and antimicrobial activities (Wang et al., 2011; Xu et al., 2012; Shipman et al., 2020). Later phosvitin was reported to promote bone formation in tissue culture and cell culture experiments. It was proposed that phosvitin provided antioxidant capacity to mirror the physiological role of ascorbic acid, which cannot be obtained during the embryo development to support skeleton formation (Liu et al., 2013; Chakrabarti et al., 2020). More recent studies have reported the positive impact of phosvitin phosphopeptides on a wide range of health conditions such as immune disorder, ageing, and cancer (Yilmaz and Ağagündüz, 2020).
Commercial production of PPP is unavailable probably due to the lack of a cost-effective method of purifying phosvitin from egg yolk. Most of the phosvitin extraction protocols are of low recovery and involve organic solvents or non-food grade chemicals. One alternative approach is to prepare phosphopeptides directly from egg yolk proteins without purifying phosvitin. Delipidated egg yolk protein hydrolysate prepared and fractionated by ultra-filtration membranes was reported with antioxidant activities (Young et al., 2011; Chay Pak Ting et al., 2011). However, these fractions represented a nitrogen to phosphorus (N/P) atomic ratio of >60 which were not comparable with the commercially available casein derived phosphopeptides (CPP) with N/P ratios ranging from 6 to 18 (Zhao et al., 2007). For phosphopeptides, a lower N/P ratio is preferable since a lower value corresponds to a higher density of phosphate groups.
Egg yolk can be easily separated into two fractions after water dilution and centrifugation. The light phase contains most of the yolk lipids in form of low-density lipoprotein is called plasma fraction, while the protein-rich pellets are called granules mainly composed of high-density lipoprotein and phosvitin (Anton et al., 2007). Granules are high density, insoluble particles in egg yolk. It was reported that nearly all the phosvitin exists in egg yolk granules (Ren and Wu, 2014). It would be more efficient to prepare phosvitin phosphopeptides directly from granules instead of purified phosvitin or defatted egg yolk protein. However, phosvitin constitutes only 16% of granules and exists in compact complexes due to electrostatic charge, and therefore it is difficult to be degraded by enzymatic hydrolysis (Marcet et al., 2022). It is reported that purified phosvitin can be hydrolyzed into phosphopeptides after partial dephosphorylation by NaOH treatment (Jie et al., 2018). However, it remains unknown whether phosvitin within granules could be dephosphorylated by NaOH and susceptible to enzymatic hydrolysis or not. It is also of interest to evaluate the effects of different enzymes on the digestibility of dephosphorylated granules and peptides yield/recovery. Therefore, this study aimed to develop a protocol to prepare and characterize phosphopeptides from granules by alkaline dephosphorylation and enzymatic hydrolysis.
| 2. Materials and methods | ▴Top |
2.1. Reagents
Glycine, Precision Plus Protein Dual Xtra Standards, Laemmli sample buffer, sodium dodecyl sulfate (SDS), and precast gels (10–20% Tris-HCl) were purchased from Bio-Rad Laboratories, Inc. (Hercules, CA, USA). Phosvitin standard (P1253, from chicken egg yolk) and leucine (L8000) were purchased from Sigma-Aldrich, Ltd. (Oakville, ON, Canada). The deionized distilled water (DD water) was produced by a Barnstead water purification system (Thermo Scientific, Asheville, NC, USA). Trypsin (T4799-10G, from porcine pancreas), pancreatin (p7545-25G, from porcine pancreas), pepsin (p7000-25G, from porcine gastric mucosa), thermolysin (p1512-25MG, from Bacillus thermoproteolyticus rokko) and α–chymotrypsin (c4129-1G, from bovine pancreas) were purchased from Sigma-Aldrich, Ltd. (Oakville, ON, Canada). Protex 6L (4880883444, from Bacillus licheniformis), protex 30L (4880873541, derived from Bacillus subtilis) and protex 7L (4860881508, derived from Bacillus amyloliquefaciens) were purchased from Danisco US Inc. (Rochester, NY, USA). All the other chemicals were purchased from Sigma-Aldrich, Ltd. (Oakville, ON, Canada) or Fisher Scientific (Nepean, ON, Canada) otherwise specified.
2.2. Granules preparation
Eggs were purchased from a local supermarket. Granules were prepared according to the protocol of McBee and Cotterill (1979) with slight modifications. Briefly granules were obtained after yolk was diluted two times with deionized water and centrifuged at 10,000 g for 45 min at 4 °C.
2.3. Alkaline dephosphorylation of granules
Granules were first homogenized by an IKA T25 homogenizer (Labsavers LLC, Astoria, NY, USA) at 15 000 rpm for 5 min, and then suspended in 0.2 M NaOH (1/20, w/w) at room temperature (25 °C) for 0.5, 1, 2, 3, 6, and 24 h with magnetic stirring, respectively. Reaction was stopped by adjusting pH to 8.0 with 3 M HCl. Trypsin was added in the ratio of 1/100 (enzyme/granules, w/w). Incubation was carried out in a 100 mL jacket beaker at 37 °C for 3 h. The temperature was maintained by using a heating circulator LAUDA A103 (LAUDA-Brinkmann, LP., Lauda-Koenigshofen, Baden-Württemberg, Germany) and maintained at pH 8.0 by addition of 0.2 M NaOH. The enzymatic reactions were terminated by raising the temperature to 95 °C for 15 min. In another experiment, granules were dephosphorylated for 0.5 h by 0.05, 0.1, 0.2 and 0.4 M NaOH, respectively, and then hydrolyzed by trypsin as described above. Hydrolysates were centrifuged at 10 000 g for 30 min at 4 °C and then lyophilized for the following assay.
2.4. Hydrolysis of dephosphorylated granules and determination of degree of hydrolysis (DH)
Granules were first dephosphorylated by 0.2 M NaOH for 0.5 h (the dephosphorylation conditions was obtained from section 2.3), and then was incubated with one of the following enzymes: pepsin (37 °C, pH 2.0), pancreatin (40 °C, pH 7.5), thermolysin (37 °C, pH 7.5), α-chymotrypsin (25 °C, pH 7.8), protex 6L (60 °C, pH 9.5), protex 30L (65 °C, pH 8.0), protex 7L (60 °C, pH 6.5) or trypsin (37 °C, pH 8.0), at a ratio of 1/100 (enzyme/granules, w/w) for 3 h as described above. Hydrolysates were centrifuged and lyophilized for the following assay.
The degree of hydrolysis was determined according to Adler-Nissen (1979). Briefly, the hydrolysate was dispersed in 1% sodium dodecyl sulphate and incubated with 0.1% trinitrobenzenesulfonic acid to determine the free amino groups released during hydrolysis by measuring absorbance at 340 nm. Leucine prepared at different concentrations was used as a standard. The DH was calculated by using the formula: DH(%) = (H/Htot)*100. H is the free amino groups released by hydrolysis and Htot is the total amino groups in the original protein, derived from its amino acid composition.
2.5. Nitrogen and protein determination
Nitrogen content was determined by Leco-N nitrogen determinator (Model FP-428, Leco Corporations, St. Joseph, MI, USA) and crude protein was calculated as N x 6.25.
2.6. Phosphorus determination
Malachite green phosphate assay kit (Bioassay Systems, Hayward, CA, USA) was used to determine the phosphorus content following the manufacturer’s instructions.
2.7. Calculation of recovery and yield
The peptide/protein yield was calculated as the amount of protein in the hydrolysate from 100 g yolk solids. The peptide/protein recovery was calculated as a percentage based on the weight ratio of amount of peptide/protein in the hydrolysate to the total protein in the egg yolk.
2.8. Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE)
Samples were analyzed by using 10–20% linear gradient gels in a Mini-Protean Tetra Cell (Bio-Rad, Hercules, CA, USA) as described by Ren and Wu (2014). Briefly samples were prepared at 1 mg/mL in Laemmli buffer (Bio-Rad, Hercules, CA, USA) and heated to 95 °C for 5 min before loading to the gel. For each well, 20 µL sample was loaded. Gel was running in a tris-glycine running buffer (14.4 g/L tris, 3g/L glycine, and 1g/L SDS) at a constant voltage of 150 V for approximately 50 min until the blue dye moved out of the gel. Staining and destaining were performed according to Hegenauer et al. (1977). Staining buffer was prepared as 0.05% Coomassie brilliant blue R-250 in a solution of 0.1 M aluminum nitrate/25% isopropanol/10% acetic acid/1.0% Triton X-100, and destaining buffer was 7% acetic acid solution. Images of gels were captured by an Alphachem SP machine. Molecular weight was analyzed by AlphaEase FC software (version 6.0.0, Alpha Innotech Corporation, Santa Clara, CA, USA).
2.9. Molecular weight (MW) distribution of hydrolysates by chromatography
A Superdex peptide 10/300 GL column (GE Healthcare, Piscataway, NJ, USA) was used to determine the molecular weight distribution of hydrolysates on an AKTA explorer 10S system (GE Healthcare, Piscataway, NJ, USA). Samples were prepared at 0.5 mg/mL in 30% aqueous acetonitrile containing 0.1% trifluoracetic acid (TFA) was injected into the column at the volume of 100 µL and eluted with the same buffer. Elution was monitored at 215 nm. Molecular weights were calculated according to a calibration curve created by running molecular weight markers under identical conditions. Molecular weight markers used in this experiment were all purchased from Sigma-Aldrich, Ltd. (Oakville, ON, Canada): Cytochrome c from equine heart (C7752, MW of 12,384 Da), Aprotinin (C6279, MW of 6,512 Da), Vitamin B12 (V2876, MW of 1,355 Da), Gly-Gly-Gly (G1377, MW of 189 Da) and Gly (G7126, MW of 75 Da).
2.10. nion exchange chromatography of pancreatic hydrolysates
Pancreatic hydrolysate from granules was prepared at 15 mg/mL with buffer A (0.05M tris-HCl at pH 8.0) and filtered through 0.45 µm membrane (Millipore, Billerica, MA, USA) before loading to a HiPrep 16/10 Q FF anion exchange column (GE Healthcare, Piscataway, NJ, USA). The elution was monitored at 215 nm and operated by an AKTA explorer 10S system (GE Healthcare, Piscataway, NJ, USA) from 100% buffer A to 100% buffer B (1.0M NaCl in buffer A). Fractions were collected and lyophilized for analysis.
2.11. Dephosphorylation of fraction G-4
Fraction G-4 from anion exchange chromatography was treated with phosphatase (E/S=1/50, w/w) in a buffer containing 10 mM tris-HCl, 5 mM MgSO4, and 1 mM ZnCl2 at pH 9.0 according to the manufacturer’s specification. The incubation was carried out at 37 °C for 3 h and terminated by increasing temperature to 80 °C for 10 min. Free phosphates and salts were removed by ZipTip pipette tip (reverse phase, 10 µL, Millipore, Billerica, MA, USA) and peptide samples were concentrated by Savant SpeedVac Concentrators (Fisher Scientific ltd., Nepean, On, Canada) for mass spectrometry analysis.
2.12. Liquid Chromatography-Mass Spectrometry-Mass Spectrometry (LC-MS/MS)
Identification of peptides in both native and dephosphorylated G-4 fraction was carried out by a Waters ACQUITY UPLC system connected to a Waters Micro mass Q-TOF Premier Instrument (Milford, MA, USA). Samples were first separated by a Waters Atlantis dC18 UPLC column (150 mm×75 μm, 3 μm; Milford, MA, USA) before MS/MS analysis. Solvent A was 0.1% formic acid in water, and solvent B was 0.1% formic acid in acetonitrile. Sample (5–10 µL) was injected to a 5 μm trapping column and trapped for 2 min at a flow rate of 10 μL/min by running 99% solvent A. Then the flow rate decreased to 0.35 μL/min when gradient decreased from 99% A to 90% A in 5 min, to 70% A in 30 min, to 60% in 3 min, and to 5% A in 1 min. The flow rate was increased to 0.50 μL/min and held for another 2 min, and then the gradient was increased from 5% A to 98% A in 1 min and held for another 27 min. At last, the flow rate decreased to 0.35 μL/min in 1 min. The elute was ionized through a nanoLockspray ionization source in a positive ion mode (capillary voltage of 3.80 kV and source temperature of 100 °C) before loaded into the mass spectrometer. A Q-TOF analyzer was used to determine the mass of peptides and the mass/charge (m/z) range was set as 400–1,600 in MS mode and 50–1,990 in MS/MS mode. Instrument was controlled and data was collected by using MassLynx software (Micromass U.K. Ltd., Wythenshawe, Manchester, U.K.). Peaks Viewer 5.2 (Bioinformatics Solutions Inc., Waterloo, ON, Canada) was used for peptide sequencing.
2.13. Statistical analysis
Experiments were repeated three times and values are presented as means ± SD. Statistical analyses were carried out using Statistical Analysis System Software, SAS version 9.0 (SAS Institute, Cary, NC). One way Analysis of variance (ANOVA) was used, and significant difference was determined by the Tukey range post-hoc comparisons and accepted at p < 0.05.
| 3. Results and discussion | ▴Top |
3.1. Effects of dephosphorylation time on hydrolysis
Both native and dephosphorylated granules were hydrolyzed by trypsin to study the effects of dephosphorylation on protein fragmentation. Granules were first dephosphorylated by 0.2 M NaOH for 0.5, 1.0, 2.0, 3.0, 6.0 and 24 h, respectively, and then hydrolyzed by trypsin. Native granules without dephosphorylation were also treated with trypsin as comparison. As shown in Figure 1, the native granules (Lane 3) were degraded by trypsin into at least 17 bands with molecular weights ranging from 3.8 to 122.0 KDa without dephosphorylation (lane 4). After dephosphorylation, a significant improvement in hydrolysis was observed as both the quantity and the intensity of the bands were decreased. There were a few faint bands after 0.5 h of dephosphorylation, and the intensity of these bands further decreased at prolonged dephosphorylation, leaving four major bands from approximately 5 to 30 kDa, with 5 kDa band was dominant. Phosvitin band was not detected in the hydrolysates of dephosphorylated granules (lane 5–10), which indicated a complete digestion of phosvitin.
 Click for large image | Figure 1. SDS-PAGE profiles of tryptic hydrolysates of granules dephosphorylated by 0.2 M NaOH for 0–24 h. 1, molecular weight markers; 2, phosvitin standard from Sigma; 3, egg yolk granules; 4, tryptic hydrolysates of granules without dephosphorylation; 5, tryptic hydrolysates of granules dephosphorylated for 0.5 h; 6, tryptic hydrolysates of granules with 1 h dephosphorylation; 7, tryptic hydrolysates of granules with 2 h dephosphorylation; 8, tryptic hydrolysates of granules with 3 h dephosphorylation; 9, tryptic hydrolysates of granules with 6 h dephosphorylation; 10, tryptic hydrolysates of granules with 24 h dephosphorylation; Granules hydrolysates were prepared as described in Materials and Methods. Phosvitin standard was purchased from Sigma. |
The DH, recovery and yield were summarized in Table 1. The DH of native granules without dephosphorylation was 5.0%, whereas DH of dephosphorylated granules showed higher values ranging from 6.7% to 7.8%. These results suggested that dephosphorylation increased DH, except for 24 h, compared to control. Similar trend was observed in the yield and recovery as well. The highest recovery of 39.8% was obtained at 0.5 h of dephosphorylation, which was significantly higher than 18.5% of the control group. The above results suggested that controlled dephosphorylation could improve efficiency of phosvitin hydrolysis, which was in good agreement with previous report (Liu et al., 2022). The low DH and recovery in control group may be due to the compact structure of native granules which might interfere with the physical access of enzymes to the cleavage site of the peptide bond. Granules are particles composed of high-density lipoprotein (HDL, or lipovitellin), low-density lipoprotein (LDL) and phosvitin. In granules, phosvitin exists in the form of lipovitellin-phosvitin complex through phosphocalcic bridge (Marcet et al., 2022). This complex is relatively stable unless it is subjected to high ionic strength (above 0.58 M NaCl) or high pH (above pH 9.0) (Lei and Wu, 2012). Even if this complex is disrupted, phosvitin is still resistant to enzymatic hydrolysis because of the high density of negatively charged phosphorylated serine (Khan et al., 1998). However, alkaline dephosphorylation could disrupt the ion bridge between phosphoserine residues and calcium, enhance electrostatic repulsions by increasing negatively charged COO- groups in proteins, and eventually destruct the structure of granules and solubilize all the components (Causeret et al., 1991). At the same time, the dephosphorylation made phosvitin molecules more vulnerable for enzymatic hydrolysis by partially removing the negatively charged phosphate groups (Jiang and Mine, 2000). Therefore, the dephosphorylated granules produced higher DH and recovery than native granules without dephosphorylation.
 Click to view | Table 1. Effects of time of dephosphorylation on DH, yield and recovery of granules hydrolysate |
3.2. Effects of NaOH concentrations on hydrolysis
Granules were treated by 0.05, 0.1, 0.2 and 0.4 M NaOH for 0.5 h prior to hydrolysis, and the hydrolysates were analyzed by SDS-PAGE. As shown in Figure 2, hydrolysates of granules dephosphorylated by 0.05 M and 0.1 M NaOH contained mainly 12 bands with molecular weights ranging from 3.7 to 84.6 kDa. The most significant band at 31.3 kDa could be considered as phosvitin fragments according to Goulas et al. (1996) and Khan et al. (1998), who obtained a phosvitin core with 28–29 kDa without dephosphorylation prior to hydrolysis. Our results suggested that 0.05 and 0.1 M NaOH may not be sufficient to dephosphorylate phosvitin molecules within granules and make them vulnerable for enzymatic hydrolysis; instead, trypsin may only cleave from C- and N-terminus of phosvitin, leaving a large intact core of protein as shown in SDS-PAGE (Lane 3 and 4 cycled area, Figure 2) (Goulas et al., 1996; Khan et al., 1998). However, no significant large protein was detected in granule hydrolysates treated with different concentrations of NaOH, which suggested that other proteins in granules should be hydrolyzed into small fragments after dephosphorylation. With treatments of 0.2 M and 0.4 M NaOH, the possible phosvitin core at around 30 kDa (Lanes 3, 4, 5 and 6 rectangle area, Figure 2) was not detected, indicating that phosvitin was eventually hydrolyzed into small peptides.
 Click for large image | Figure 2. SDS-PAGE profiles of tryptic hydrolysates of granules dephosphorylated by various NaOH concentrations. 1, molecular weight markers; 2, phosvitin standard from Sigma; 3, tryptic hydrolysates of granules dephosphorylated by 0.05 M NaOH; 4, tryptic hydrolysates of granules dephosphorylated by 0.1 M NaOH; 5, tryptic hydrolysates of granules dephosphorylated by 0.2 M NaOH; 6, tryptic hydrolysates of granules dephosphorylated by 0.4 M NaOH; 7, egg yolk granules. Granules hydrolysates were prepared as described in Materials and Methods. Phosvitin standard was purchased from Sigma. |
Table 2 summarized the DH, yield and recovery of tryptic hydrolysates of granules dephosphorylated by 0.05, 0.1, 0.2 and 0.4 M NaOH. The DH ranged from 7.6% to 11.0%. Yield and recovery were not significantly different among all the treatments. However, the electrophoretic patterns indicated the hydrolysates with 0.05 M and 0.1 M NaOH treatments contain partially digested phosvitin and other large fragments, whereas dephosphorylation by 0.2 and 0.4 M NaOH led to more extensive hydrolysis of granule. Therefore, the dephosphorylated granules produced higher DH and recovery than native granules without dephosphorylation.
 Click to view | Table 2. Effects of NaOH concentrations on DH, yield and recovery of trypsin-digested granules |
3.3. Hydrolysis of granules by different enzymes
Effects of different enzymes on phosvitin hydrolysis were examined. Granules were dephosphorylated by 0.2 M NaOH for 0.5 h prior to hydrolysis. The electrophoretic patterns in Figure 3 showed that all the enzymes could degrade dephosphorylated granules into smaller fragments, though some of them produced fragments as large as 10–15 kDa (lane 3, pepsin).
 Click for large image | Figure 3. SDS-PAGE profiles of granules hydrolysates prepared by different enzymes (granules were dephosphorylated by 0.2 M NaOH for 0.5 h before hydrolysis). 1, molecular weight markers; 2, phosvitin standard from Sigma; 3, pepsin hydrolysates of granules; 4, pancreatin hydrolysates of granules; 5, thermolysin hydrolysates of granules; 6, chymotrypsin hydrolysates of granules; 7, protex 6L hydrolysates; 8, protex 30L Granules hydrolysates; 9, protex 7L hdyrolysates; 10, trypsin hydrolysates; 11, granules. |
To further determine the molecular weight distribution of the hydrolysates, a Superdex peptide 10/300 GL column was used for gel filtration chromatography. The whole hydrolysate without fractionation was a mixture of various protein fragments covering a wide range of molecular weights as shown in Figure 4. In all the enzymatic hydrolysates except for pancreatin hydrolysate, the fractions were eluted from 9 mL to 21 mL, corresponding to molecular weights ranging from 129 to 16 913 Da (calculated from a predetermined standard curve). Large proteins such as lipovitellin (200 kDa) and phosvitin (37–45 kDa) were not detected. The pancreatic hydrolysate, with the highest DH, showed better separation than other hydrolysates. It was resolved into five major fractions with peak molecular weights of 1 253 Da, 718 Da, 250 Da, 176 Da and 56 Da. It is noteworthy that the small fractions (250 Da, 176 Da and 56 Da) take larger proportion in pancreatic hydrolysate than in other hydrolysates. Besides, small fractions such as 176 Da and 56 Da were only found in pancreatic hydrolysate.
 Click for large image | Figure 4. Gel filtration chromatogram of granules hydrolysates by different enzymes. |
The atomic ratio of nitrogen to phosphorus (N/P) is frequently used to describe phosphopeptides such as the commercially available CPP. Usually a lower N/P ratio is preferable since a lower value corresponds to a higher density of phosphate groups (Zhao et al., 2007). As shown in Table 3, Protex 7L hydrolysate represented the lowest N/P value of 15.2, and the lowest recovery of 19.4%. Pepsin hydrolysate represented the largest N/P ratio of 45. It was reported the N/P of delipidated egg yolk protein hydrolysates was 69.1 (Chay Pak Ting et al., 2011), which is much higher than the values in this study. The purified phosvitin usually has N/P values of ∼3.0 depending on extraction methods, and we previously characterized phosphopeptides from highly purified phosvitin with a N/P value of 3.9 and a DH of 12.9% (Ren et al., 2015). Considering phosvitin accounts for 16% of granules, the N/P values obtained in this study were at a low level, which suggested relatively high density of phosphate groups in these peptides. It is generally believed that small peptides may survive digestion and thus exert their physiological functions; therefore, a higher DH is desirable to prepare bioactive peptides. Chay Pak Ting et al. (2011) reported a DH of 25.3% by using a combination of alcalase and protease N to hydrolyze egg yolk protein. In this study, pancreatic hydrolysate showed the highest DH of 24.2% which is comparable to that of egg yolk protein. Pancreatin is a mixture of several enzymes, including amylase, trypsin, lipase, ribonuclease and protease. In this study pancreatin appeared to be a good choice for granules hydrolysis because granules consist of both lipids and proteins, and these components could be hydrolyzed by lipase and trypsin (protease), respectively. Pancreatic hydrolysate ranked the second highest recovery of 41.8% with corresponding yield of 138.4 mg per gram yolk solids, after the Protex 6L hydrolysate with the highest recovery of 43.4% with a yield of 144.0 mg per gram yolk solids. For CPP, which were usually prepared by trypsin or pancreatin hydrolysis, the DH varied from 10% to 25%, and the yield ranged from 100–200 mg per gram caseinate (Adamson and Reynolds, 1995; Ellegard et al., 1999). Both the DH and yield in this study were comparable to those of CPP.
 Click to view | Table 3. DH, yield, recovery and N/P value of granules hydrolysates |
3.4. Fractionation of pancreatic hydrolysate by Anion exchange chromatography
The pancreatic hydrolysate derived from dephosphorylated granules (0.2 M NaOH for 0.5 h) was eluted on a HiPrep 16/10 Q FF anion exchange column (GE Healthcare Bioscience, USA) as shown in Figure 5. HiPrep 16/10 Q FF is a strong anion exchange column, which will interact with the negatively charged phosphoserine residues from phosphopeptides. With the increase of NaCl gradient, the eluted fractions are supposed to take more negative charge. In Figure 5, the pancreatic hydrolysate could be roughly resolved into four fractions representing 1.5%, 8.1%, 5.4% and 85.1% of the total phosphorus content in all fractions, which was in good agreement with the results of Katayama et al. who reported similar chromatography patterns with purified phosvitin hydrolysate (Katayama et al., 2006).
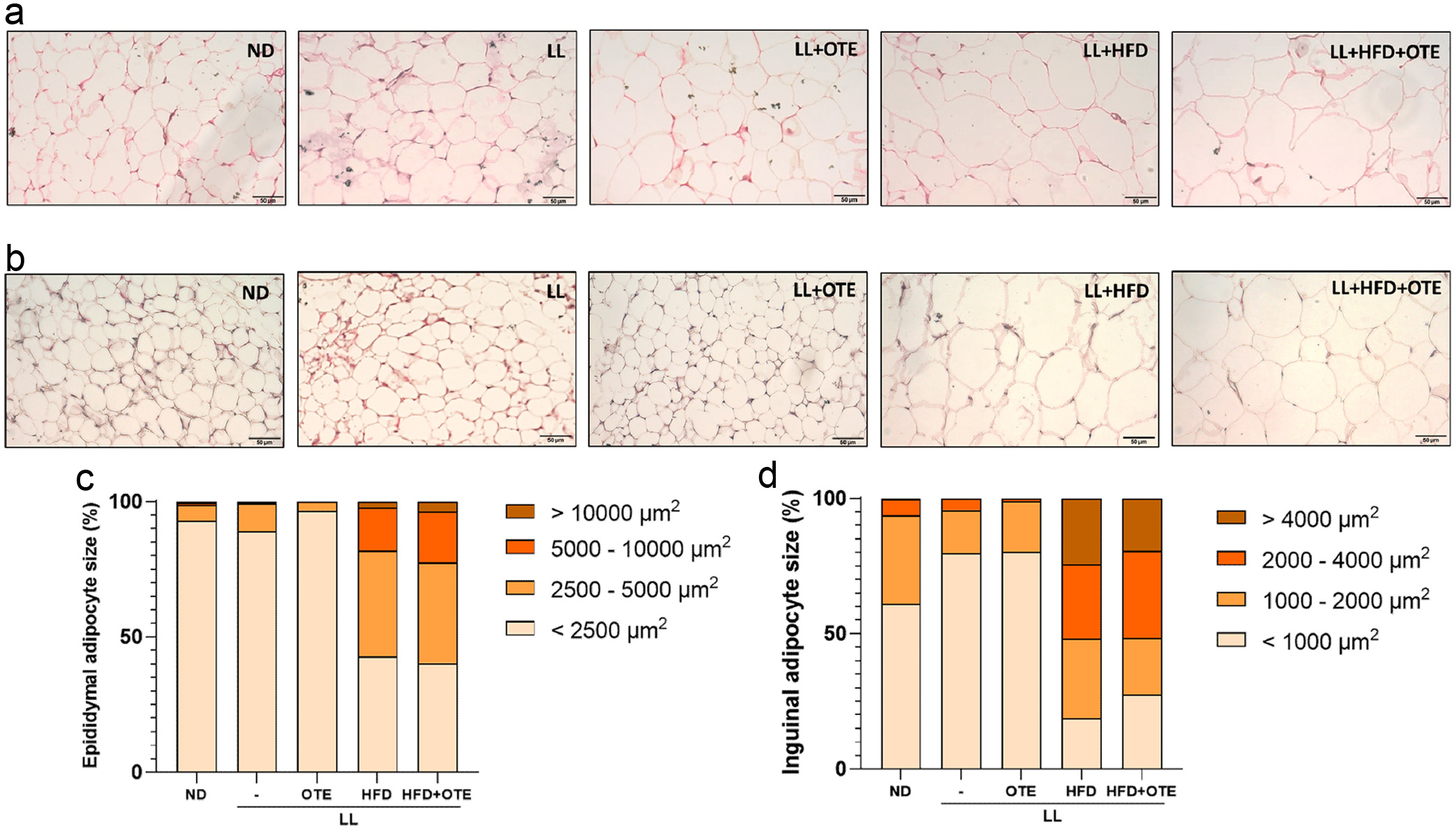 Click for large image | Figure 5. Anion exchange chromatography of pancreatic hydrolysates of granules. |
3.5. Identification of peptides in fraction G-4 by LC-MS/MS
The G-4 fraction containing the most phosphorus was collected for sequence identification by LC-MS/MS. Sequence identification of multiple phosphorylated peptides is always problematic due to the strong negative charges from the phosphoserine residues (Potel et al., 2018). These peptides are too hydrophilic to bind the dC-18 column and are also difficult to ionize to generate detectable signals for MS/MS analysis. In this study, an aliquot of G-4 fraction was dephosphorylated with phosphatase prior to LC-MS/MS to detect peptide backbone sequences. At the same time, native G-4 fraction without dephosphorylation was also subjected to LC-MS/MS to identify the phosphorylation sites of the peptides. An example of de novo sequencing was provided in Figure 6.
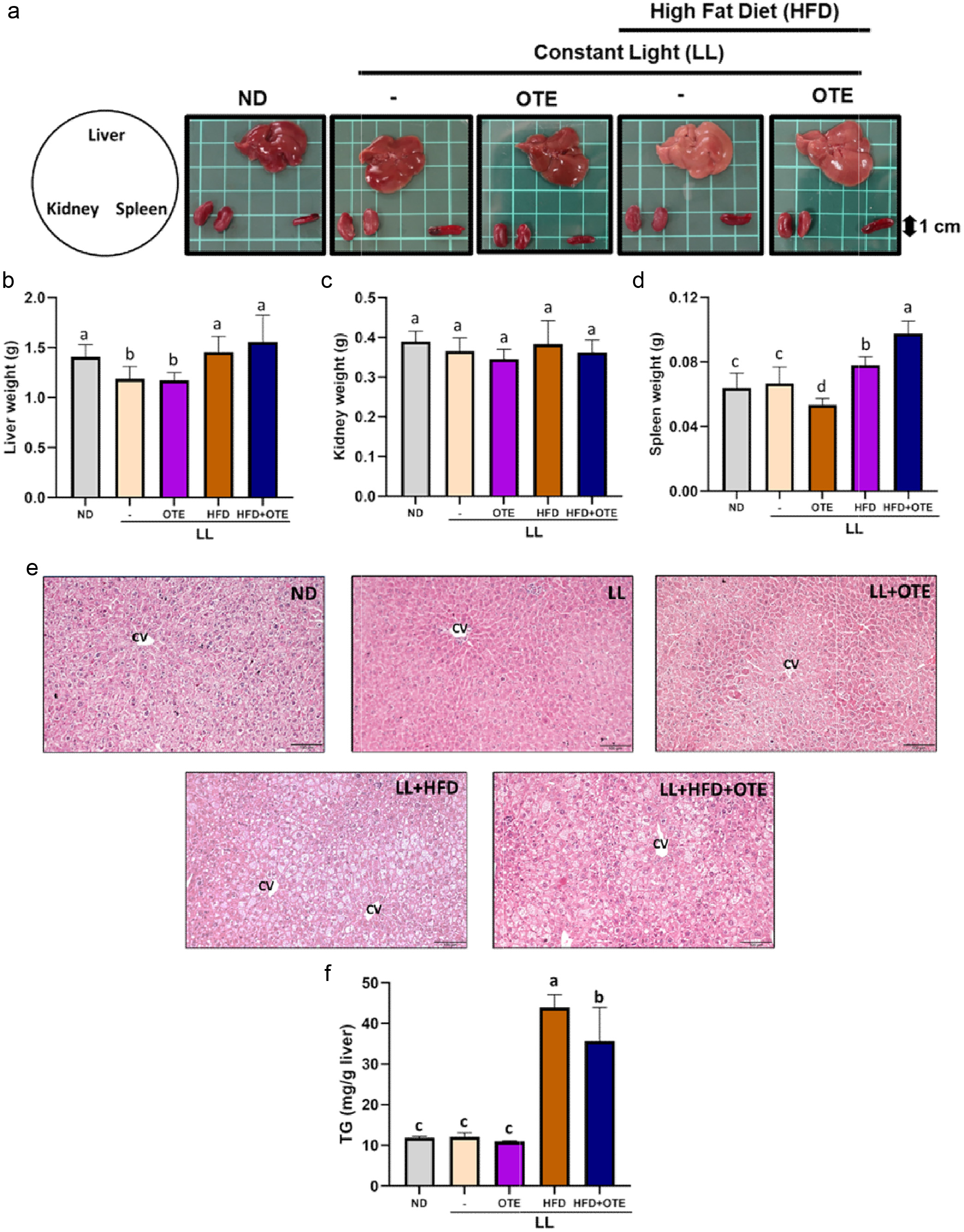 Click for large image | Figure 6. One representative peptide LEDDSSSSSSSSVLSK from fraction G-4 was de novo sequenced by using its MS/MS spectra (a) Total ion chromatogram of G-4 fraction. (b) Mass spectrum of a selected ion with m/z of 807.9. (c) MS/MS spectrum of the ion m/z 807.9. |
In G-4 fraction, 15 peptides were identified from 3 domains in phosvitin sequence: AKTSSSSSSASSTATSSSSSSASSPN (PV 9-34), DEEENDQV (PV 40-47) and SGHLEDDSSSSSSSSVLSKIWG (PV 190-211); 54 peptides were identified from 5 domains in lipovitellin sequence: IGNIYAPE (VTG 116-123), TVDLNNCQEK (VTG 182-191), MTPPLTGDF (VTG 821-829), TSILPEDAPLDVTEEPF (VTG 929-945) and IITEVNPESEEEDESSPYEDIQA (VTG 1056-1078) (Table 4). It is not surprising to characterize more peptides from other egg yolk proteins than from phosvitin, since granules are a mixture of 60% lipovitellin, 16% phosvitin and 12% low-density lipoproteins (Marcet et al., 2022). This study successfully identified phosvitin peptide sequences from dephosphorylated G4 fraction with MW ranging from 793.3 (EFGTEPD) to 2,137.9 (TSSSSSSASSTATSSSSSSASSPN) but was unable to determine the phosphorylation sites in these peptides in the native G4 fraction, probably due to the difficulty in ionizing peptides with multiple phosphorylation. Although we observed fragments in LC-MS mode which might represent phosvitin phosphopeptides, their signals were too weak to be chosen for the MS/MS analysis. Considering the low N/P value of these peptides, it is reasonable to assume some of the non-phosphorylated phosvitin peptides might also be phosphorylated. Peptides derived from the core of phosvitin (PV 48-189) were not detected by LC-MS/MS, probably due to the high amount of phosphorus as well. Interestingly, we found many peptides from lipovitellin containing 1–2 phosphoserine or phosphothreonine residues. This result could be explained by the fact that lipovitellin, the major protein in egg yolk granules, is also a phosphoprotein containing 0.2–0.4% protein-bound phosphorus (Kurisaki et al., 1981). There was few information about phosphopeptides derived from lipovitellin so far. It will be interesting to study these phosphopeptides and compare their physiological activities with phosvitin derived phosphopeptides.
 Click to view | Table 4. Peptides identified in fraction G-4 |
| 4. Conclusions | ▴Top |
Casein phosphopeptides have been reported to have calcium-absorption promotion, antioxidant and anti-inflammatory activities. However, the commercial application of phosvitin phosphopeptides has been hindered by the lack of a scalable protocol to prepare phosvitin phosphopeptides. In this study we prepared phosphopeptides directly from egg yolk granules, with comparable N/P ratio to purified CPP. NaOH dephosphorylation of granules prior to enzymatic hydrolysis was proved to be critical to hydrolyze phosvitin within granules. Pancreatin hydrolysis of dephosphorylated granules (0.2 M NaOH for 0.5 h) showed the highest DH of 24.2% with recovery of 41.8%. In this pancreatic hydrolysate, 15 peptides were identified from 3 domains in phosvitin, and 54 peptides were identified from 5 domains in lipovitellin, 26 of which were confirmed to be phosphorylated. This study suggested it is feasible to prepare phosphopeptides directly from egg yolk granules.
Acknowledgments
This research was supported by grants from Alberta Agriculture and Forestry, Egg Farmers of Canada, Michael Foods Ltd., and Natural Sciences and Engineering Research Council (NSERC) of Canada to J. Wu. The funders had no role in the study design, data collection and analysis, decision to publish or preparation of the manuscript. We thank Jing Zheng (Department of Chemistry, University of Alberta, Edmonton, Canada) for her excellent technical support in LC–MS/MS analysis.
Data availability statement
The data that support the findings of this study are available upon request from the corresponding author.
Current Address: School of Biological and Food Engineering, Guangdong University of Education, 510310, Guangzhou, China.
Conflict of interest
The authors declare no conflicts of interest.
Author contributions
Conceptualization, Jianping Wu; methodology, Jiandong Ren; formal analysis, Jiandong Ren; investigation, Jianping Wu and Jiandong Ren; writing—original draft preparation, Jiandong Ren; writing—review and editing, Jianping Wu; supervision, Jianping Wu; funding acquisition, Jianping Wu. All authors have read and agreed to the published version of the manuscript.
| References | ▴Top |