| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Review
Volume 3, September 2018, pages 8-75
Bioactives in spices, and spice oleoresins: Phytochemicals and their beneficial effects in food preservation and health promotion
Feriedoon Shahidi*, Abul Hossain
Department of Biochemistry, Memorial University of Newfoundland, St. John’s NL, Canada A1B 3X9
*Corresponding author: Feriedoon Shahidi, Department of Biochemistry, Memorial University of Newfoundland, St. John’s NL, Canada A1B 3X9
DOI: 10.31665/JFB.2018.3149
Received: July 2, 2018; Revised received & accepted: July 22, 2018
| Abstract | ▴Top |
Spices have broadly been used as food flavoring and folk medicine since ancient times. Numerous phytochemicals have been identified in spices, namely thymol (ajowan and thyme), anethole (aniseed), piperine (black pepper), capsaicin (capsicum), cinnamaldehyde (cinnamon), eugenol (clove), linalool (coriander), sabinene (curry leaf), limonene (dill seed), estragole (fennel seed), allicin (garlic), gingerol (ginger), safranal (saffron), and curcumin (turmeric), among others. The antioxidants in spices are very effective and also render anti-mutagenic, cardioprotective, anti-inflammatory, and anti-cancer properties. Apart from their antioxidant efficacy, spices, particularly their essential oils possess strong antimicrobial activity against bacteria, fungi, yeasts, and microbial toxins synthesis. In this contribution, a summary of the most relevant and recent findings on phytochemical composition and antioxidant properties of spices has been compiled and discussed. The content of phenolic acids, flavonoids, tannins, glycosides, steroids, and terpenoids in different spices are summarized. In addition, the beneficial effects of spices in food preservation and in health promotion and disease risk reduction are briefly described.
Keywords: Spice; Phytochemicals; Antimicrobial activity; Health beneft; Application
| 1. Introduction | ▴Top |
Spices are the dried portions of certain aromatic plants which are effective as flavorants, flavor enhancers, preservatives, colorants, as well as potential therapeutic agents. Spices may be derived from various parts of the plant such as buds, bark, flowers, leaves, fruits, roots, rhizomes, seeds or the whole plant tops. Spices contain many phytochemicals and their main flavor-active components are volatile liquids as well as oleoresins (Shahidi et al. 1995). Volatile essential oils are a concentrated liquid with strong odor obtained by steam distillation and are mainly used as preservatives. In contrast, oleoresins are natural mixtures of essential oils and resin which are responsible for the total flavor profile of spices and can be extracted from plants by using organic solvents or supercritical fluid extraction (SFE) with CO2 with or without an auxiliary solvent such as ethanol. Moreover, the oleoresins from spices provide the taste, whereas the volatile oils impart the aroma. In addition, oleoresins are less volatile and contain lipophilic components, mainly resins, waxes, and fats and provide some advantages over the dry powder of spices such as uniformity of aroma and taste as well as having a minimum microbial load. The most commonly used spices in 36 countries around the world include garlic, ginger, onions, and few species of pepper (Kaefer and Milner 2008). Asia supplies a majority of spices, particularly cinnamon, nutmeg, pepper, ginger as well as clove. Moreover, Guatemala is a major supplier of cardamom, Brazil is a leading producer of pepper, Nicaragua, the USA, and El Salvador produce sesame seed, Grenada supplies ginger and nutmeg, whereas Europe, the USA and Canada grow numerous aromatic seeds (Parthasarathy et al. 2008). People of Asia particularly China, India, Pakistan, and Bangladesh use spices as traditional medicines to cure infectious diseases, where Africans use plant extracts as a natural remedy to cure many ailments (Amrita et al. 2009; Tajkarimi et al. 2010). In addition, Egyptians used medicinal plants for preserving the dead bodies (Nkambule 2008).
Spices have been used not only for the palatability of food and as a flavoring agent but also as a source of natural bioactive compounds. The main bioactive compounds associated with spices comprise a diverse array of components such as terpenes and terpenoid components, phenolic compounds and other bioactive molecules (Lampe 2003). The most common chemical compounds found in spices are alkaloids, saponins, glycosides, phenolic compounds, and organic acids, among others (Leja and Czaczyk 2016; Parthasarathy et al. 2008). Compounds such as capsaicin in red chilies, curcumin in turmeric, eugenol in clove, piperine in black pepper, linalool in coriander, cuminaldehyde in cumin, and zingerone in ginger are known to inhibit lipid peroxidation (Nagababu and Lakshmaiah 1992; Pulla Reddy and Lokesh 1992).
Spices play a vital role in enriching flavor, improving digestion, serving antioxidants as well as supporting human health (Viuda-Martos et al. 2011). Spices comprise products of secondary metabolism such as phenolics (flavonoids, phenolic acids, tannins, and quinones) (Lai and Roy 2004). Several studies have reported that spices such as nutmeg, sage, rosemary, oregano, thyme, and ginger contain a number of phenolics which act as strong antioxidants (Cuvelier et al. 1996; Srinivasan 2014; Yanishlieva et al. 2006). Phenolic compounds exhibit a wide range of biological properties such as anti-inflammatory, anticancer, antiallergenic, antiproliferative, and antmicrobial activities. Due to the presence of these compounds, they are responsible for reducing the lipid, protein and enzymatic oxidation and increasing the shelf life of foods. Besides, these compounds are responsible for preventing or retarding off-flavor and rancidity development in foods (Kähkönen et al. 1999). Due to the degradation of essential fatty acids, the quality of products may change by developing rancid flavor and changing color as well as texture. To prevent these oxidation-derived changes in food, incorporation of antioxidants is necessary and phenolic compounds from spices can provide a good source of natural antioxidants. This is because phenolic compounds act as reducing agents, singlet oxygen quenchers and hydrogen atom donors and some also have the ability to chelate the prooxidant metal ions. Therefore, understanding the chemical structures of phenolics is necessary in order to evaluate their effects and those of their active metabolites.
The demand for food packaging systems with antioxidant activity is continuously increasing due to the desire to formulate clean label preservative-free food product. Phytochemicals obtained from spices are not only used in food but also in food packaging to increase the shelf life and quality of products. In particular, packaging of fat-based food is important as lipid oxidation can occur which is responsible for food spoilage. Therefore, incorporation of antioxidants into the packaging material enhances the quality of food due to oxidation prevention. However, synthetic antioxidants (e.g. butylated hydroxylanisole (BHA), butylated hydroxyltoluene (BHT), and tertiary butylhydroquinone (THBQ)) have fallen out of favor due to their potential adverse effects on health (Shahidi and Zhong 2010). Consequently, natural antioxidants such as phenolics and polyphenolics in spices and those isolated from essential oils of spices are important and the latter is a current trend in the food packaging sector (Silva-Weiss et al. 2013). This is because spice extracts are well known to possess antimicrobial properties, especially those of cinnamon, garlic, clove, sage, pimento, oregano, thyme, and rosemary have demonstrated inhibitory activity against pathogenic organisms. Spices extracts are rich in phenolic acids as well as essential oil component, which can control foodborne pathogens (Radha Krishnan et al. 2014). Moreover, simple phenols and phenolic acids inhibit the growth of microorganisms due to the presence of hydroxyl groups in their chemical structures. Furthermore, most of the phenolic compounds are lipophilic and hence play an important role in their antimicrobial activities. This is because lipophilic components are responsible for inflicting structural and functional damage to microorganisms by disrupting the osmotic balance of the cell and membrane permeability (Prakash et al. 2015). Mau et al. (2001) and Ouattara et al. (1997) reported that spice oils have an inhibitory effect against meat spoilage bacteria such as Bacillus subtilis, Listeria monocytogenes, Escherichia coli, Salmonella Typhimurium, Vibrio parahaemolyticus, Aspergillus flavus, Penicillium italicum, and Cinnamomum zeylanicum.
Due to their medicinal properties, spices have been used for thousands of years in Ayurvedic as well as Chinese traditional medicine to cure chronic diseases. Spices have attracted much attention because of their high antioxidative, antimicrobial, anticarcinogenic, anti-inflammatory, antimutagenic, and immune modulatory properties in cells and render a broad range of positive effects against cardiovascular, stomachache, gastrointestinal, respiratory, reproductive, convulsion, metabolic, neural, rheumatism, inflammation, intestinal worms, loss of appetite, intermittent fevers, diarrhea, leprosy, hepatic urinary discharges diseases, dyspepsia, dental diseases, diabetes, cough, and constipation (Bi et al. 2017; Kannappan et al. 2011; Kochhar 2008; Lampe 2003; Ramkissoon et al. 2012; Valko et al. 2007). Moreover, flavonoids from oregano, black pepper, thyme, and marjoram may assist in preventing intestinal cancer and cardiovascular disease, whereas gingerol from ginger acts as an intestinal stimulant (Peter 2001). In addition, onion, fenugreek, and garlic assist in lowering the cholesterol level, while capsaicin from chili pepper is used in both pharmaceuticals and cosmetics industries. Furthermore, curcumin in turmeric, capsaicin in red pepper, and eugenol in clove help to control cellular oxidative stress due to their ability to control the production of reactive oxygen species (ROS) (Rubió et al. 2013; Srinivasan 2014). Therefore, there is a real need for new remedies to develop drugs and natural products with reduced side effects, and spices can offer a promising source for such drugs.
There is also much interest in identification of biologically active compounds present in spices in order to use them in food product development. Generally, spices are used as flavoring, colorant and antimicrobial agents in food, pharmaceutical, perfumery, cosmetic, insecticide, and nutraceutical industries. The application of spices in food products with a multitude of nutritional benefits is highly relevant to the food industry. Therefore, this review summarizes the bioactive compounds in spices, and spice oleoresins, and their beneficial effects in food preservation and human health promotion. An overview of the application of spices in food is also provided.
| 2. Botanical characteristics and phytochemicals of spices and spice oleoresins | ▴Top |
Spices have exceptionally rich in vitamins, micro- and macro molecules, as well as a broad range of bioactive phytochemicals such as phenolics and saponins among others, which are known to possess antioxidant activity. Phytochemicals are highly variable due to their geographical origin, cultivar, processing factor, and analytical methodology. Therefore, this review intends to summarize the major compounds in spices that have shown biological activities. The specific phytochemicals in most common spices and spice oleoresins are also detailed.
2.1. Ajowan
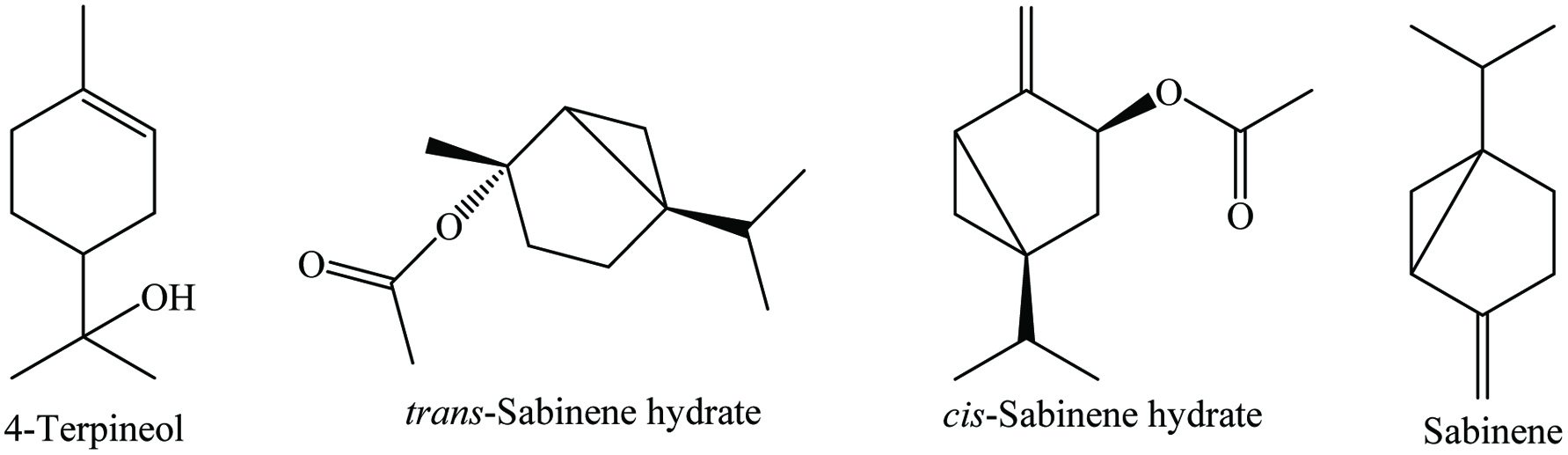
Ajowan (Trachyspermum ammi L) is an important seed spice belonging to the family Apiaceae. It is known as Sprague ex Turill, Trachyspermum copticum Linn, Ammi copticum Linn, Ptychotis coptica DC, Carum copticum Benth and Hook, Lingusticum ajowain Roxb. Ajowan is widely produced in India, Pakistan, Iran, Iraq, Saudi Arabia, Egypt, and Europe (Charles 2013). It is an annual herbaceous plant containing feathery leaves with red flowers and can grow up to 3 ft. Seeds (whole or ground), volatile oil, and oleoresins are the major products of this plant. The seeds have a strong aromatic odor with a bitter and pungent aromatic taste.
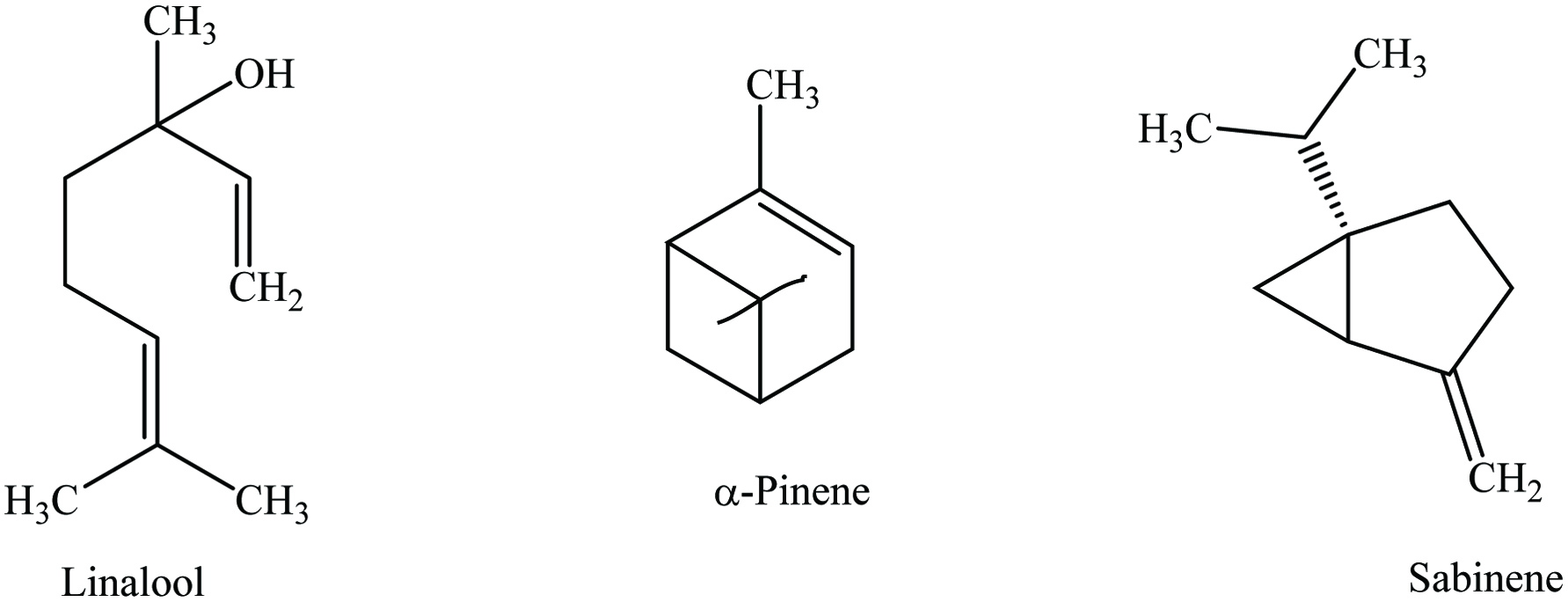
Ajowan seeds contain 9% moisture, 15.4% protein, 18.1% lipid, 38.6% carbohydrates, 11.9% crude fiber, and 7.1% ash (including 1.42% calcium and 0.3% phosphorous) with essential oil. It contains a wide range of phytochemicals (Table 1), the most abundant of which are p-cymene, γ-terpinene, and thymol (Figure 1) (Mirzahosseini et al. 2017). In addition, it contains α- and β-pinenes, α-terpinene, dipentene, carvacrol, camphene, α-3-carene, myrcene, saponin, and 6-O-β-glucopyranosyloxythymol (Bairwa 2012). Choudhury (1998) reported a yield of 1.2% for ajowan oil with its active components being carvone (46.2%), limonene (38.1%), and dillapiole (8.9%). The essential oil of ajowan contains approximately 50% of thymol that is a strong antispasmodic, germicide, and fungicide. In addition, oleoresin of ajowan is a pale green oily liquid which provides flavor and aroma.
 Click to view | Table 1. Chemical composition of ajowan |
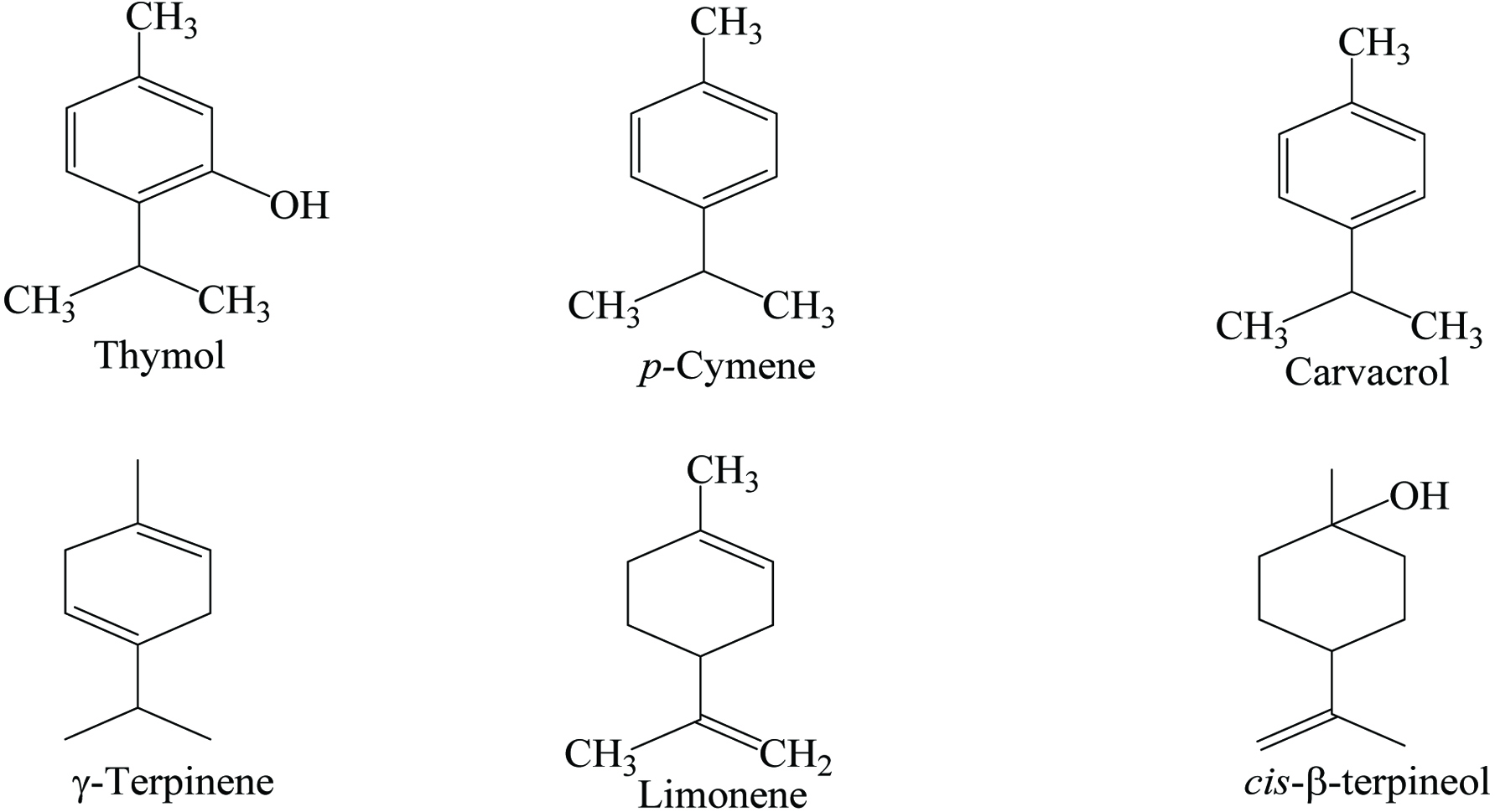 Click for large image | Figure 1. Major phytochemicals of Ajowan. |
2.2. Aniseed
Aniseed or anise (Pimpinella anisum L.) is an annual plant in the family of Apiaceae and is known as Pimpinella magna L, Anisum vulgare Gaertn, anis seed, and sweet cumin. Anise seed is widely cultivated in eastern and western Mediterranean; it is also produced in Russia, Lebanon, Japan, India, and South America. It is an herbaceous plant covered by fine hairs and can grow up to 2 ft with ternately pinnate leaves. The flowers are white whereas the fruit is pyriform or ovoid with a unique sweet smell. The main products of this plant are essential oil and oleoresin from seed, ripe dry seeds, and the fresh leaves with strong aroma (Parthasarathy et al. 2008).
Aniseed contains 9% moisture, 18% protein, 16% lipid, 35% sugars, 7% ash, 5% starch, and 12–25% crude fiber, as well as 22–28% N-free extract, and 2–7% essential oil (Pruthi 1976). Anise oleoresin is a yellowish-green to orange-brown liquid and contains approximately 15–20% volatile oil and a wide range of phytochemicals including trans-anethole, phenolics, and anisaldehyde (Table 2). The primary constituents of the aniseed oil are anethole (90%), anisketone, anisaldehyde, and methyl chavicol. Rodrigues et al. (2003) extracted essential oil (3.13–10.67%) from aniseed and its major constituents are reported as anethole (90%), p-anisaldehyde (1%), γ-himachalene (2–4%), methylchavicol (0.9–1.5%), trans-pseudoisoeugeny l-2-methylbutyrate (1.3%), and cis-pseudo l-2-methylbutyrate (3%). The main non-volatile components of aniseed are phenolic acids (caffeic acid, hydroxycinnamic acid, hydroxybenzoic acid, and p-coumaric acid) (Schultz and Herrmann 1980), flavonoids (cynaroside, luteolin, quercetin-3-glucuronide, isoorientin, rutin, apigenin-7-glucoside, and isovitexin) (El-Moghazi et al. 1979; Ozguven 2000), sterols, furanocoumarins, and glucosides (Figure 2). In addition, Kumaravel and Alagusundaram (2014) reported that the major phytochemicals of aniseed were phenolics (312 ± 4.5 mg gallic acid equivalents/100 g), phytosterols, terpenoids, alkaloids, and cardiac glycosides.
 Click to view | Table 2. Chemical composition of aniseed |
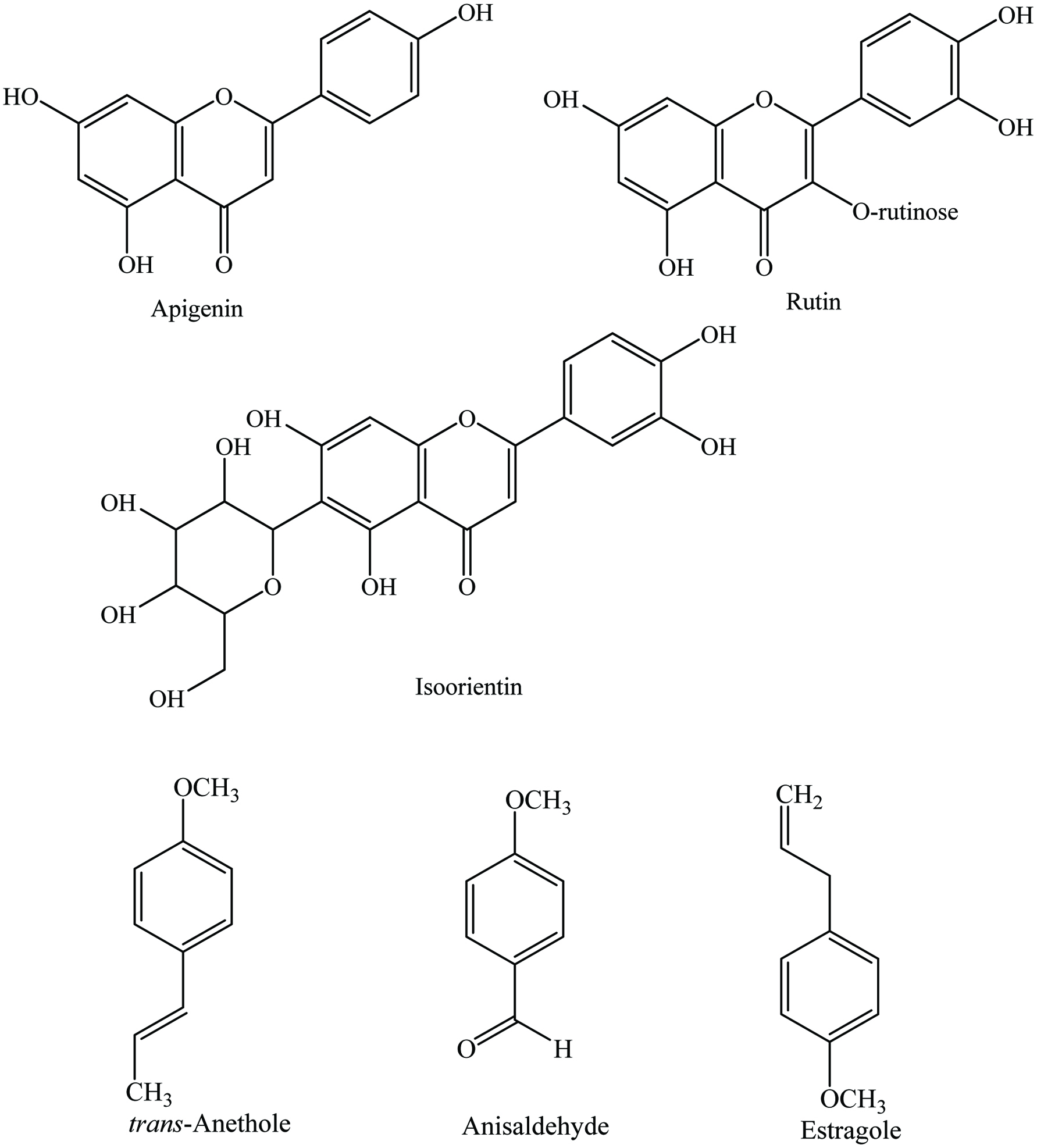 Click for large image | Figure 2. Major phytochemicals of aniseed. |
2.3. Basil
Basil (Ocimum basilicum L) is a culinary herb of the family Lamiaceae which is known as “Herbe Royale”. The synonyms of basil are Ocimum canum Sims, Ocimum americanum ssp. Americanum, comoran basil, reunion basil, and sweet basil. This annual herbaceous plant is native to Africa (mainly Egypt) and Asia (mainly Iran), and is also widely cultivated throughout Europe and the USA. It can grow up to 2 ft with grayish-green beneath, ovate leaves, and white, greenish or pinky-white flowers. Basil is used as fresh, dried, or paste and its main products are leaves and essential oil (Charles 2013).
Basil is unique in its combination and overall content of nutrients. It contains 10.30% moisture, 23% protein, 4% lipid, 37% fiber, 2.24% calcium, and vitamins. Research on basil phytochemicals started in the 1930s and currently more than 200 chemical compounds have been identified from samples produced in many parts of the world. It contains a wide range of phytochemicals including estragole, eugenol, cintronellol, apigenin, limonene, methyl cinnamate, ursolic acid, 1,8-cineole, α-terpinene, β-sitosterol, anthocyanins, p-coumaric acid, rosmarinic acid, carvacrol, farnesol, kaempherol, camphor, geraniol, menthol, quercetin, rutin, safrole, catechin, and tannin (Table 3). Chemical structures of some phenolic compounds found in basil are shown in Figure 3. The major components of Ocimum basilicum are linalool (35.1%), eugenol (20.7%) and 1,8-cineole (9.9%) (Piras et al. 2018). Juliani and Simon (2002) quantified the total phenolic content in milligram gallic acid equivalents per gram dry weight of nine basil cultivars, namely cinnamon basil (35.6), dark opal basil (126.2), holy basil (51.1), Italian large leaf basil (62.9), sweet dany lemon basil (55.8), osmin purple basil (81.7), purple ruffles basil (92.6), red rubin basil (95.1), and sweet basil (55.7). In addition, Marwat et al. (2011) reported that the chemical components of basil as monoterpene hydrocarbons, sesquiterpene hydrocarbons, oxygenated sesquiterpene, oxygenated monoterpene, flavanoids, triterpene, and aromatic compounds. Besides, rosmarinic acid was found to be the major antioxidant component in sweet basil which was confirmed by 1H NMR and characterized by mass spectrometry (Jayasinghe et al. 2003).
 Click to view | Table 3. Phenolic composition of basil |
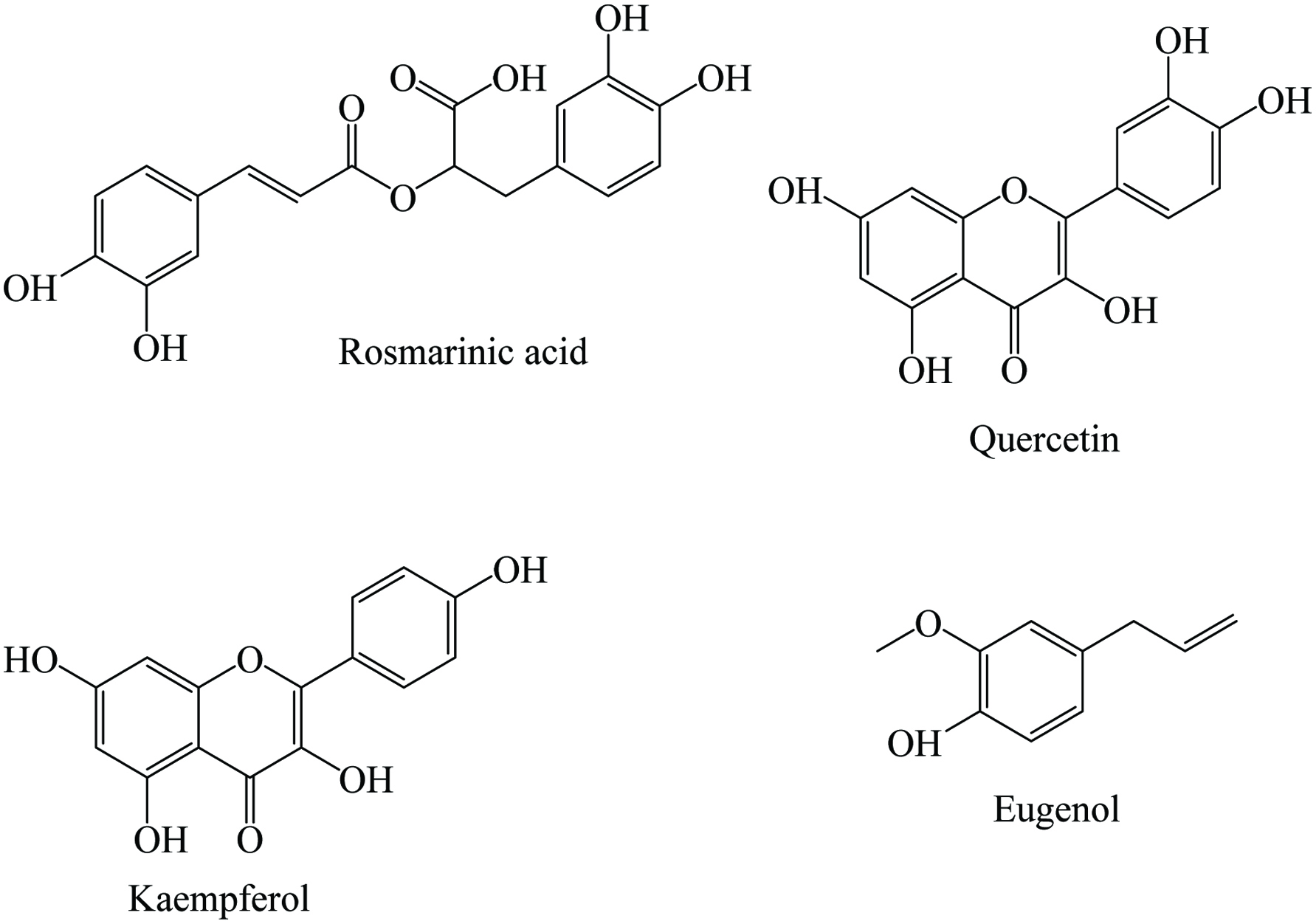 Click for large image | Figure 3. Major phenolics of basil. |
2.4. Bay leaf
Bay (Laurus nobilis L) or bay leaf belongs to the family of Lauraceae which is known as Laurus persea L, Laurus winteriana L, bay laurel, sweet bay, true bay, grecian, laurel, and true laurel. It is native to Asia and the Mediterranean region, in particular, it has been cultivated in France, Italy, Spain, Yugoslavia, Morocco, Israel, Turkey, China, and Russia. This plant can grow up to 66 ft high with dark green leaves and small yellow flowers. In addition, its fruits are succulent, cherry-like purple to black in color and have a single seed with a loose kernel. Leaves and essential oil are the main products of bay leaves used as spices (Charles 2013).
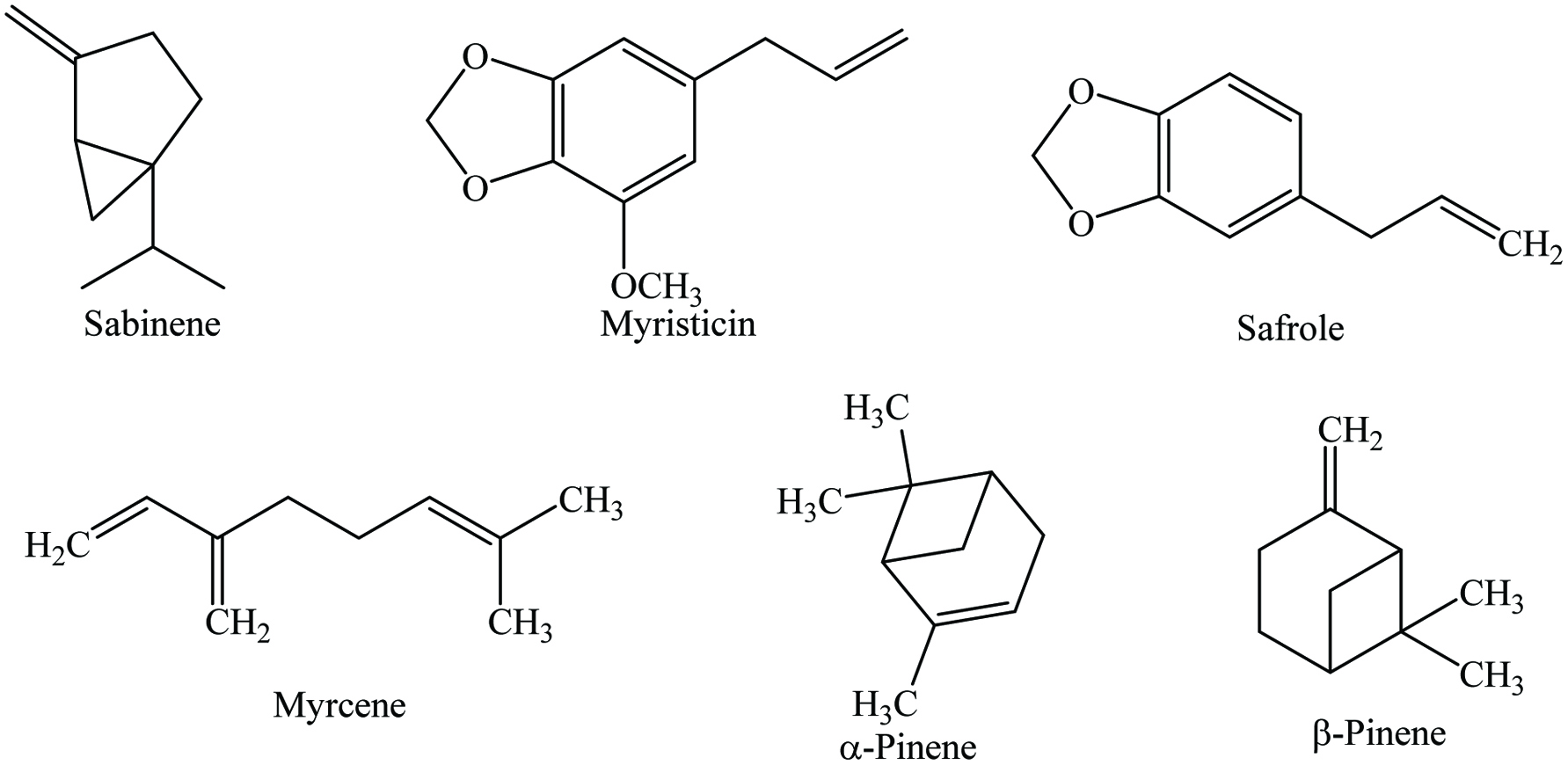
Bay leaves contain 4–10% moisture, 7–11% protein, 4–9% lipid, 65% carbohydrates, and 4% ash, as well as riboflavin, thiamine, niacin, ascorbic acid, and 0.5–3.5% essential oil. Numerous phytochemicals have been identified in bay leaves (Table 4), of which the main ones are α-terpinylacetate, cinnamtannin, 1,8-cineole, sabinene, and α-pinene (Mohammadreza 2008). Bay leaves are rich in total phenolics (1.03–289.45 mg gallic acid equivalents/g) (Boulila et al. 2015; Dall’Acqua et al. 2009; Hinneburg et al. 2006; M. Lu et al. 2011; Muñiz-Márquez et al. 2014; Siddhartha et al. 2017; Soomro 2016; Ünver et al. 2009; Vallverdú-Queralt et al. 2014; Ying et al. 2015), total flavonoids (6.34–8.72 mg quercetin equivalents/g) (Boulila et al. 2015; Kivrak et al. 2017; Siddhartha et al. 2017), and terpenoids (128 ± 13.7 mg linalool equivalents/g) (Siddhartha et al. 2017). In addition, p-coumaric acid, ferulic acid, protocatechuic acid, and catechin are the most abundant polyphenols in bay leaves. Moreover, Leja and Czaczyk (2016) reported that bay leaves contain a wide range of antioxidant compounds such as beta-carotene, ascorbic acid, tocopherol, methyl eugenol, eugenol, eudesmol, kaempferol-3-rhamnopyranoside, kaempferol, kaempferol-3,7-dirhamnopyranoside, α-terpinyl acetate, 1,8-cineole, terpinen-4-ol, and catechin. Recently, Bendjersi et al. (2016) identified the phytochemical composition of bay leaves essential oils by hydrodistillation and solvent-free microwave extraction, including 1,8-cineole (26.4–30.9%), α-terpinyl acetate (9.5–7.8%), sabinene (9–9.6%), linalool (4.9–9.5%), α-pinene (9.2–4.6%), α-terpineol (3.3–7.6%), methyl eugenol (5–6.2%), and eugenol (1.2–3.6%) (Figure 4).
 Click to view | Table 4. Phenolic composition of bay leaves and fruit |
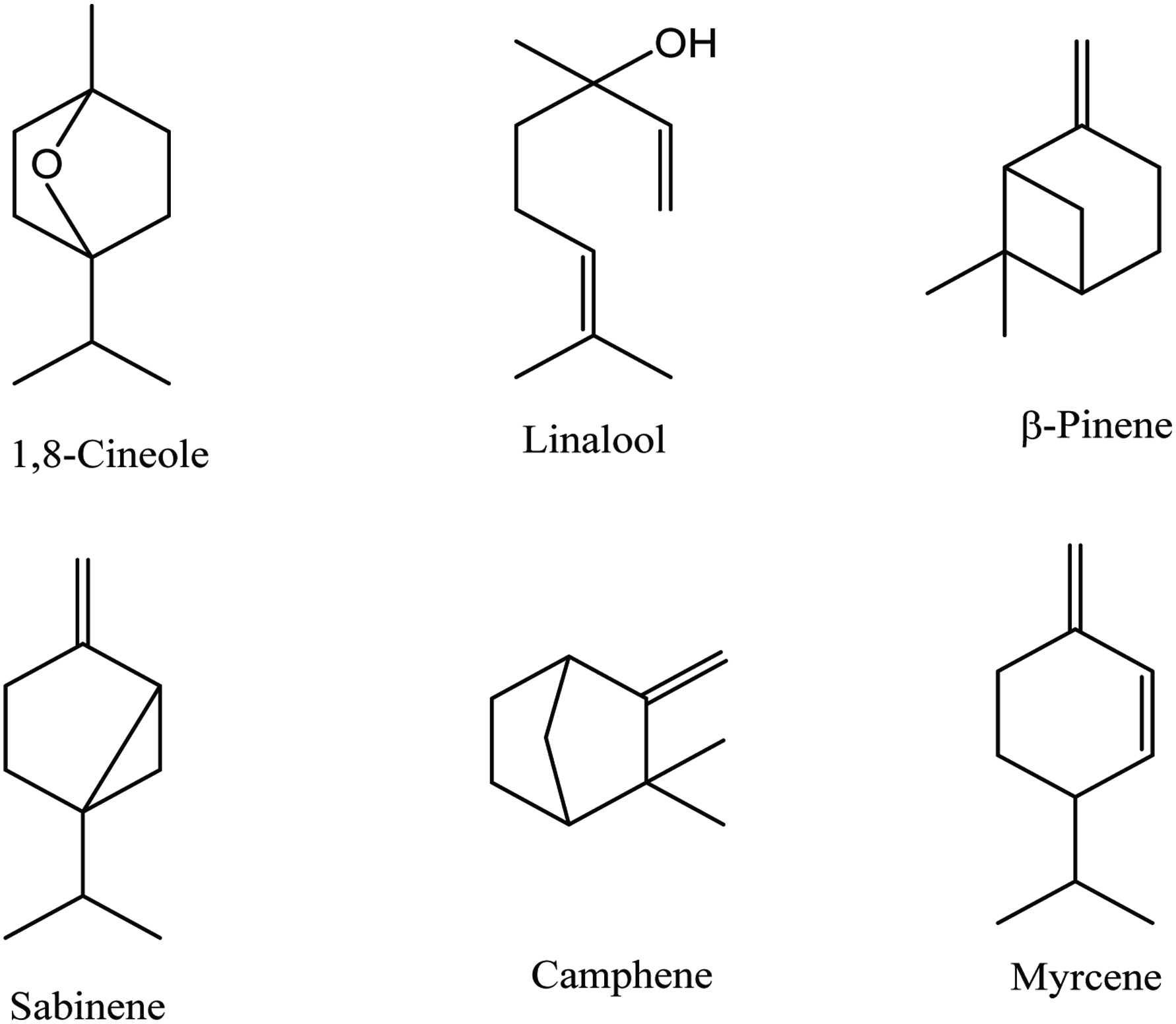 Click for large image | Figure 4. Major phytochemicals of bay leaf and fruit. |
2.5. Black pepper
Black pepper (Piper nigrum L) is the most widely used spice in the world and is known as “King of Spices”. It belongs to the family Piperaceae and is also known as piper, pepper, peper, kalimirch, pimento, and maricha. It is a perennial plant and native to South India, and some tropical regions around the world. It can grow up to 30 ft and the main products of this spice are peppercorns, essential oil and oleoresin. Black and white pepper are the fruit of the same pepper plant, but black pepper is harvested from unripe berries and dried until the outer layer becomes black, whereas white pepper obtained from ripe seeds and removal of the skin. Black pepper tastes hotter and lasts longer than the white pepper. The quality parameters of it depend on piperine that provides the pungency and the volatile oil that contributes to its flavor and aroma (Nagavekar and Singhal 2017; Peter 2001).
The volatile oil and pungent compounds are the major components of black pepper. Black pepper contains about 2.0–33.5% piperine, around 6–13% oleoresin, and about 2–3% volatile oil (Butt et al. 2013). Oleoresin of black pepper is a green, thick, and viscous liquid which is responsible for pungency and flavor of black pepper due to the presence of piperanine constituents. The oleoresins obtained from Piper nigrum showed anti-inflammatory, antioxidant, and antimicrobial activities (Nagavekar and Singhal 2017). Black pepper contains 8% moisture, 10% protein, 10.2% lipid, 66.5% carbohydrate, 4.6% ash, and vitamins (Tainter and Grenis 1993). It also contains a wide range of phytochemicals including piperine, camphene, pinene, limonene, terpenes, isoquercetin, piperidine, sarmentine, alkaloids, and flavonoids (Table 5) (Shahidi and Ambigaipalan 2015). Chemical structures of some bioactive compounds found in black pepper are shown in Figure 5. The most abundant compounds in black paper essential oil obtained by hydro-distillation are β-caryophyllene (18.64 ± 0.84%), sabinene (13.19 ± 0.17%), limonene (14.95 ± 0.13%), β-pinene (9.71 ± 0.12%), 3-carene (8.56 ± 0.11%), and α-pinene (7.96 ± 0.14%) (Bagheri et al. 2014). Numerous bioactive compounds have been identified in black pepper fruit, of which they are phenolics (5.86 ± 0.03 mg gallic acid equivalents/g), flavonoids (15 ± 0.69 mg rutin equivalents/g), terpenodis (65 ± 6.5 mg linalool equivalents/g), and alkaloids (0.9 ± 0.06 mg atropine equivalents/g) (Siddhartha et al. 2017).
 Click to view | Table 5. Phenolic composition of black pepper |
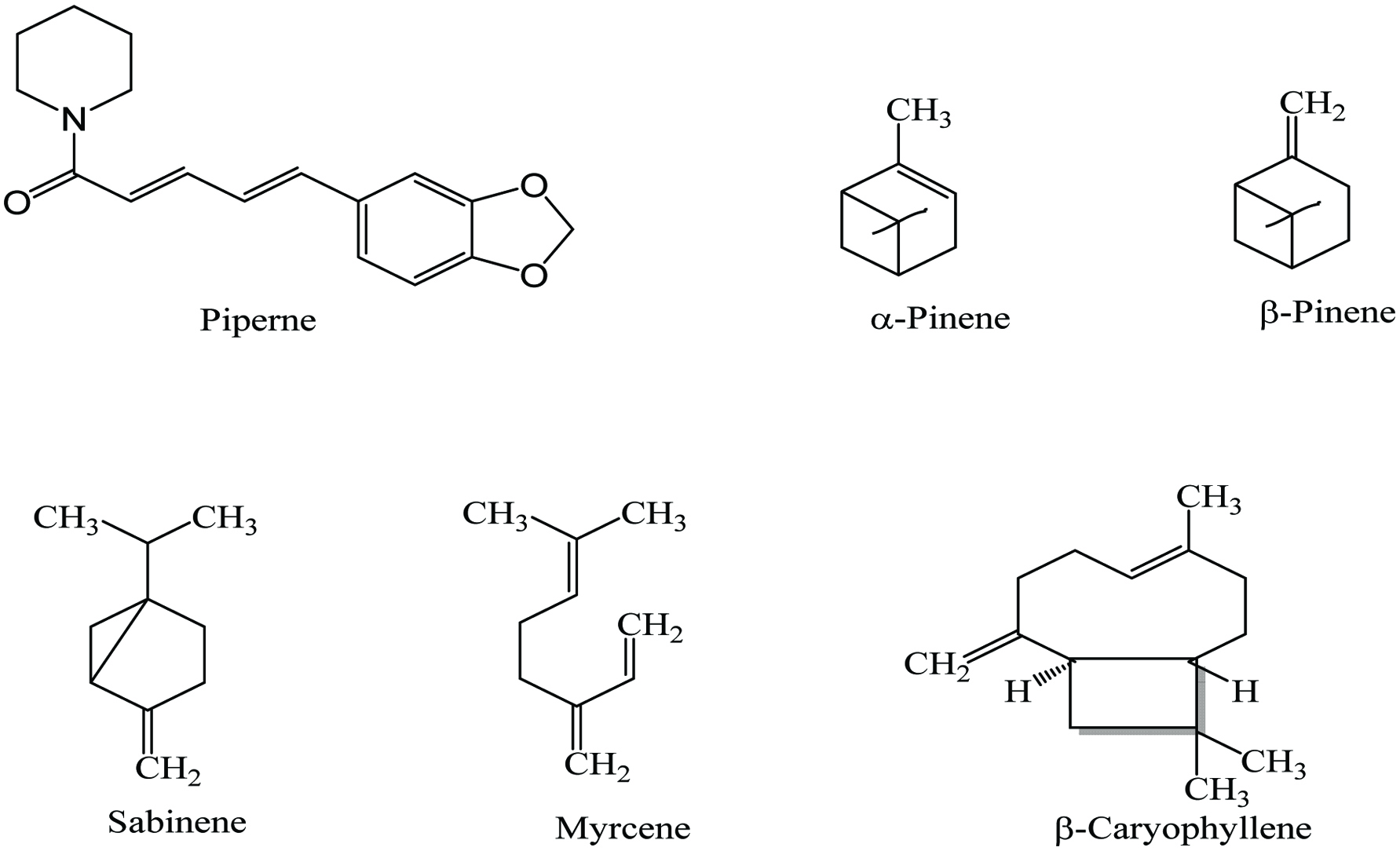 Click for large image | Figure 5. Major phytochemicals of black pepper. |
2.6. Capsicum
Capsicum (Capsicum annuum L) belongs to the family of Solanaceae and the common species are C. baccatum, C. frutescens, C. chinense, and C. pubescens. It is also known as a cayenne pepper, red pepper, tabasco pepper, hot pepper, paprika, chili pepper, cayenne, pimento pepper, and Hungarian pepper. Most of these species are perennial woody shrubs, whereas Capsicum annumis is an annual herb. It is native to tropical America and West Indies but it is cultivated all over the world, especially China, India, and the USA.
The main cause of pungency of capsicums is a group of alkaloid components known as capsaicinoids (Figure 6). Capsicum contains 8-10% moisture, 12–15% protein, 9–17% lipid, 50–60% carbohydrate, and vitamins (A and C) as well as volatile oil. The yield of oleoresin varied from 11.5 to 16.5%, which is primarily red pepper (mainly for color and pungency), paprika (color), and capsicum (used for pungency) (Peter 2001). The most abundant compounds found in capsicum are shown in Table 6. The red capsicum has a significantly higher level of phenolics, β-carotene, capsanthin, quercetin, and luteolin followed by orange, yellow, and green capsicum (Sun et al. 2007). In addition, Kumar et al. (2010) determined the total phenolic contents of Capsicum annuum at three ripening stages (green, intermediate, and red) from nine cultivars and reported red stage had the highest amount of phenolics (110–266 mg GAE/100 g) compared to intermediate (85–220 mg GAE/100 g), and green stage (64–188 mg GAE/100 g) products.
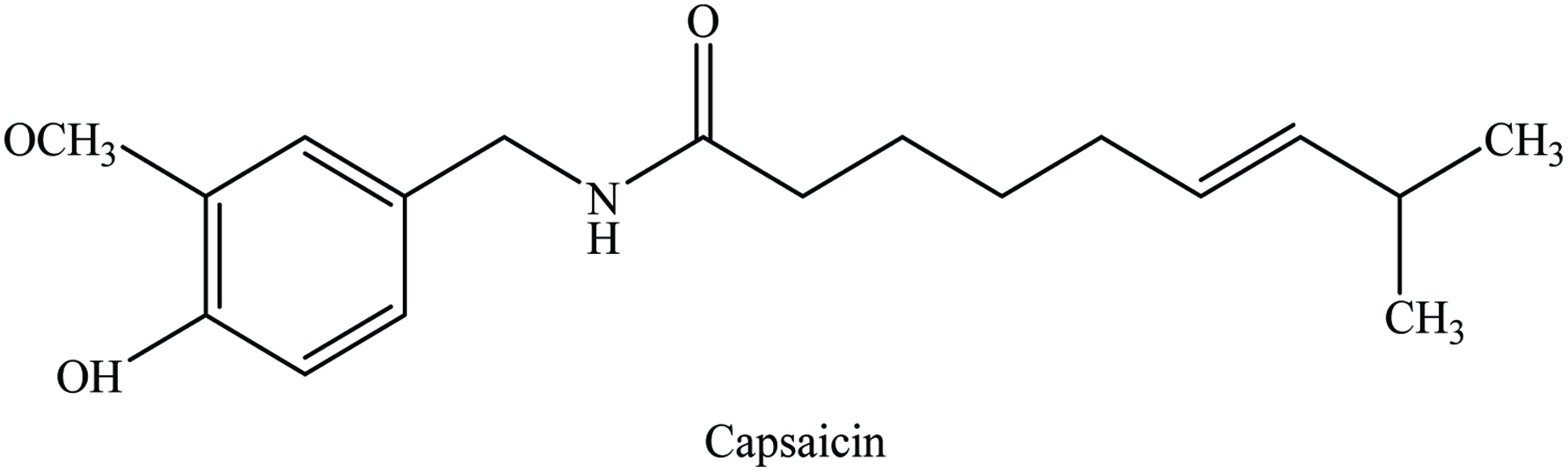 Click for large image | Figure 6. Chemical structure of capsaicin. |
 Click to view | Table 6. Phenolic composition of capsicum |
2.7. Cardamom
Cardamom (Elettaria cardamomum L.) belongs to the family of Zingiberaceae and is known as “Queen of Spices”. It is a perennial herbaceous plant and considered as the third most expensive spice after saffron and vanilla (Parthasarathy et al. 2008). India and Saudi Arabia are the main producer and exporter of cardamom; Sri Lanka, Bangladesh, Tanzania, Vietnam, Laos, Cambodia, El Salvador, and Papua New Guinea are also among the major exporting countries. It grows up to 16 ft high with orchid-like white flowers and lance-shaped leaves. The fruit is an ovoid containing oblong red-brown seeds which are covered by an aril. Fruit, essential oil, oleoresin, and seeds of cardamom have pungent, sweet and also warm and aromatic flavor (Charles 2013).
Cardamom contains around 30% husk, about 75% seed, nearly 11% volatile oil, and fixed oils (mainly oleic and palmitic acids). It has 8.28% moisture, 10.76% protein, 6.70% lipid, 68.47% carbohydrate, 28% dietary fiber, and vitamins (Charles 2013). The oleoresin of the cardamom is a colorless to pale yellow liquid which is used in food, pharmaceutical, perfumery, and liquor industries. The major constituents of cardamom oleoresin are identified by GC-MS, mainly α-terpinyl acetate (46.42%) and 1,8-cineole (30.23%) (Sardar and Singhal 2013). The major components of the volatile oil are 1,8-cineole (around 50%) and α-terpinyl acetate (about 50%) (Figure 7). Cardamom is a rich source of alkaloids, flavonoids, saponins, steroid glycosides, tannins, and terpenoids (De Soysa et al. 2016; Sibi et al. 2013). The major phytochemical components are 1,8-cineole, limonene, α-terpinyl acetate, α-pinene, β-pinene, sabinene, borneol, methyl eugenol, myrcene, γ-terpinene, p-cymene, terpinolene, aphellandrene, linalool, α-terpineol, α-terpinyl acetate, citronellol, linalyl acetate, nerd, trans-nerolido, and geraniol (Table 7) (Guenther 1975; Przygodzka et al. 2014; Yashin et al. 2017).
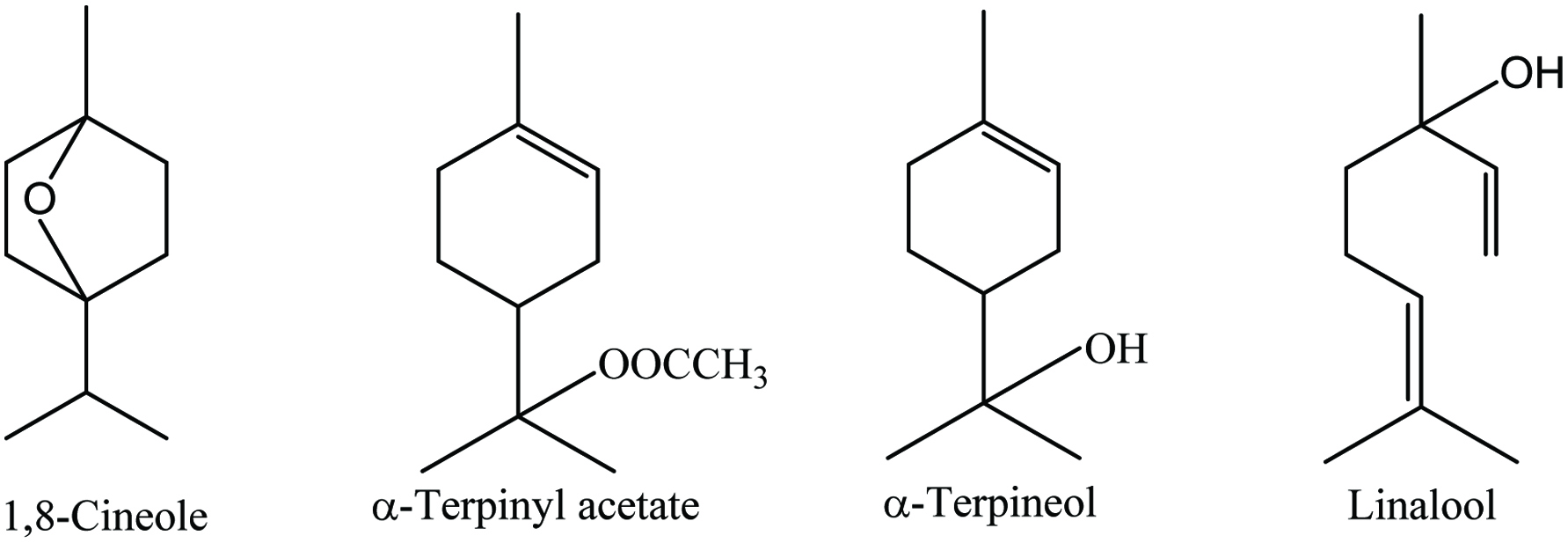 Click for large image | Figure 7. Major phytochemicals of cardamom. |
 Click to view | Table 7. Phenolic composition of cardamom seeds |
2.8. Cinnamon
Cinnamon (Cinnamomum verum J. Presl) is a member of the family Lauraceae and is known as Ceylon cinnamon, Cinnamomum zeylanicum Nees, and true cinnamon. It is a tropical evergreen plant and considered one of the finest sweet spices in the world. It is indigenous to China, Southeast Asia, and Saudi Arabia and also found in Australia, Indonesia, Thailand, South America, and the West Indies. This plant can grow up to 50 ft high with long, leathery, bright green leaves and yellow flowers. The fruit is ovoid purple containing a single seed. Bark powder, Bark, leaf essential oil, bark essential oil, and oleoresin of cinnamon are used as a spice (Nabavi et al. 2015).
Cinnamon contains 9.5–10.5% moisture, 3.89–4.65% protein, 59.55–80.59% carbohydrate, 53.1% dietary fiber, 3.55% ash, and vitamins (Charles 2013; Parthasarathy 2008; Peter 2001). Cinnamon oleoresin is a reddish brown powder which is obtained by solvent extraction such as ethanol (yield 10–12%) and benzene (yield 2.5–4.3%), the latter being a prohibited solvent. Cinnamon contains a wide range of phytochemicals including cinnamaldehyde, cinnamyl acetate, cinnamyl alcohol, eugenol, eugenol acetate, linalool, benzaldehyde, cinnamaldehyde, methyl eugenol, monoterpene, hydrocarbon, benzyl benzoate, caryophyllene, pinene, phellandrene, safrole, cymene, and cineol (Table 8) (Jayaprakasha et al. 2002; Kaefer and Milner 2008; Peter 2001; Vangalapati 2012; Yashin et al. 2017). The major constituents of cinnamon bark are essential oil (up to 2.8%), with cinnamaldehyde (60–90%) as a major component. Marongiu et al. (2007) isolated 19 compounds from cinnamon by supercritical CO2 extraction, reported the major constituents are trans-cinnamaldehyde (77.1%), trans-β-caryophyllene (6.0%), γ-terpineol (4.4%), as well as eugenol (3.0%) (Figure 8). Cinnamon leaves contain 0.24–3.0% volatile oil and the principal component of leaf oil is eugenol, varied from 65 to 97% (Senanayake et al. 1978). In addition, the major components of root bark, flower, and fruit oils are camphor, trans-cinnamyl acetate, and linalool, respectively (Parthasarathy et al. 2008). The biological properties of cinnamon are related to its higher polyphenol content and the major phenolics are vanillic acid, caffeic acid, and ferulic acid (Muchuweti et al. 2007). Abeysekera et al. (2013) reported that the ethanolic extracts of cinnamon leaf and bark had high phenolics (44.57 ± 0.5 and 33.43 ± 0.51 mg GAE/g, respectively) and flavonoids (12.0 ± 0.37 and 3.07 ± 0.24 mg QE/g, accordingly) contents which were more than those of dichloromethane/methanol extracts. Besides, phytochemicals such as glycosides, steroids, alkaloids, saponins, anthraquinones, tannins, terpenoids, and coumarins had also been identified in cinnamon extracts (De Soysa et al. 2016; Harsha et al. 2013; Sibi et al. 2013; Shreya et al. 2015; Tacouri et al. 2013).
 Click to view | Table 8. Phenolic composition of cinnamon |
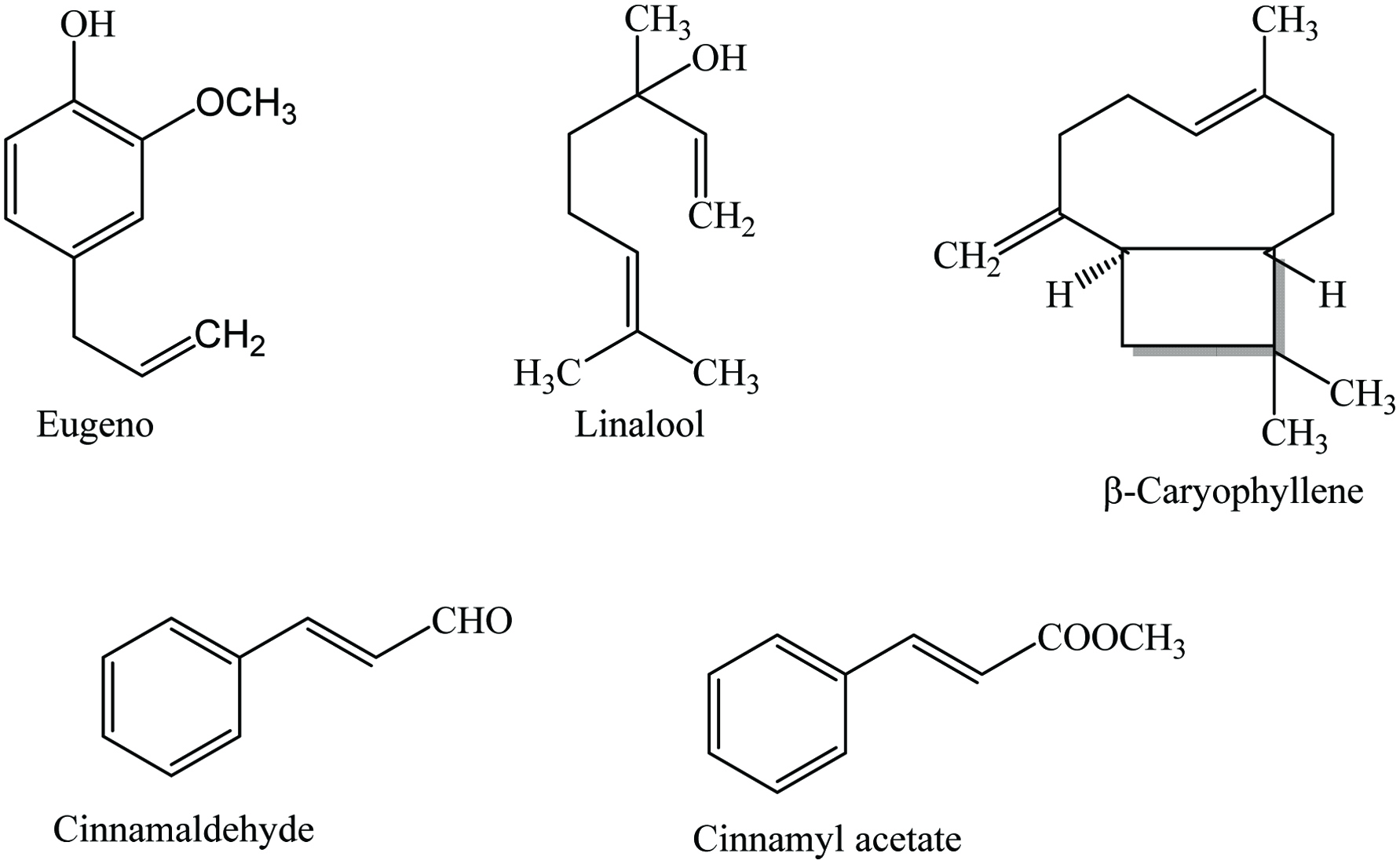 Click for large image | Figure 8. Major phytochemicals of cinnamon. |
2.9. Clove
Clove (Syzygium aromaticum L) belongs to the family Myrtaceae and its common synonyms are Caryophyllus aromaticus L, Eugenia caryophyllata Thunb, and Eugenia aromatic (L.) Baill. Clove is native to southeast Asia and is also cultivated worldwide, particularly in India, Sri Lanka, Indonesia, Madagascar, Zanzibar, Philippines, Tanzania, and Brazil. It is an evergreen tropical plant and grows up to 50 ft high with large glossy green leaves. It has white fragrant flowers with a fleshy hypanthium surrounded by sepals and a purple drupe fruit. Clove buds (whole or ground), clove bud essential oil, clove stem essential oil, clove leaf essential oil, and oleoresin are the main products of clove (Peter 2001).
The composition of clove includes 5.40–9.87% moisture, 5.97–6.0% protein, 13–20.06% lipid, 61.22–61.80% carbohydrate, and 5.0–5.88% ash, as well as vitamins (Charles 2013; Parthasarathy 2008; Peter 2001). Numerous phytochemicals have been identified in clove, mainly eugenol, acetyl eugenol, β-caryophyllene, α-cububene, isoeugenol, sesquiterpene, nerolidol, farnesol, vanillin, pinene, phenolic acids, flavonoids, and tannins (Table 9) (Jirovetz et al. 2006; Kaefer and Milner 2008; Leja and Czaczyk 2016; Nassar et al. 2007; Politeo et al. 2006; Przygodzka et al. 2014; Shahidi and Ambigaipalan 2015; Yashin et al. 2017). Clove buds contain 15–20% volatile oil, whereas leaf, fruit, and stem contain 3.0–4.8, 2.0, and 6% essential oil, respectively (Parthasarathy 2008). The major components of the essential oil are eugenol (70–95%), eugenol acetate (15–17%), and β-caryophyllene (8–15%) which are responsible for providing aroma (Figure 9) (Gopalakrishnan et al. 1988; Raina et al. 2001). Clove fruit has significantly higher phenolics (247.61 and 141.70 mg GAE/100 g) and flavonoids (209.48 and 126.50 mg QE/100 g) compared to clove stem (Al-Mashkor 2015). In addition, phytochemicals such as alkaloids, flavonoids, tannins, phenols, glycosides, steroids, saponins, phytosterols, and terpenoids also been identified in clove (De Soysa et al. 2016; Kumaravel and Alagusundaram 2014; Sibi et al. 2013). Besides, clove oleoresin (90–92% volatile compounds) prepared by alcohol extraction with a yield of 22–31% (Weiss 1997).
 Click to view | Table 9. Phenolic composition of clove |
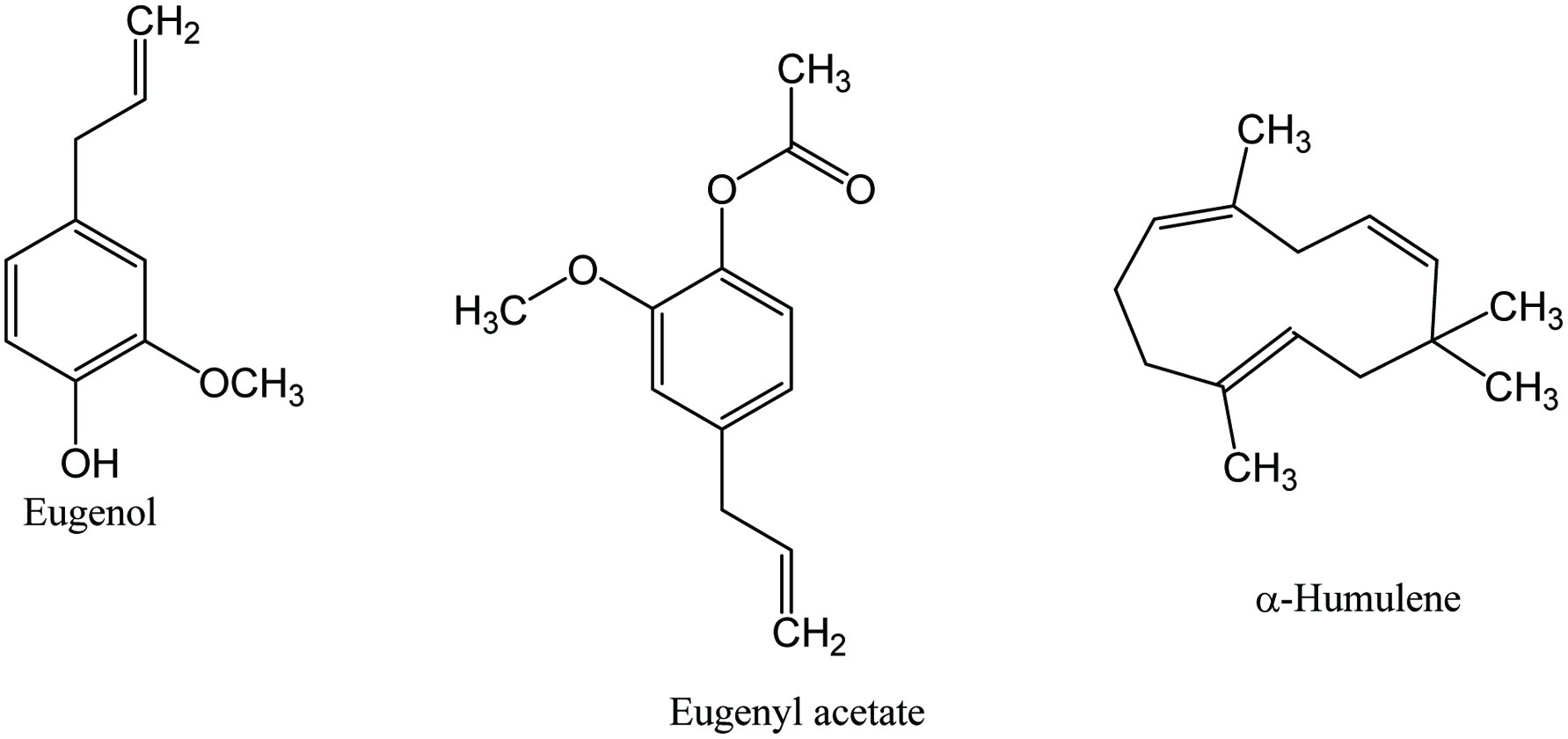 Click for large image | Figure 9. Major phytochemicals of clove. |
2.10. Coriander
Coriander (Coriandrum sativum L) is a member of the Apiaceae family which is native to the Mediterranean area and is extensively cultivated in Asia, central Europe, and North Africa as a culinary and medicinal plant. The common synonyms for coriander are Coriandrum majus Gouan, culantro, Chinese parsley, cilantro (leaf) and coriander (fruit). This annual herbaceous plant grows up to around 5 ft high with bright green strong-smelling leaves and pale-pink or white flowers. The globular coriander dried fruits (seeds) are widely used as a spice for a warm and pleasant odor. Ripe whole plant, fresh leaves (whole or chopped), stem and roots (fresh or dried), dry seeds, essential oil, and oleoresin are the main products of this plant (Iqbal et al. 2018; Laribi et al. 2015).
Coriander fruits contain 8–11% moisture, 11–17% proteins, 16–28% lipid, 18–20% carbohydrates, 3–5% ash, 23–36% fibre, and 1–3% essential oil. The oleoresin of coriander is a brownish-yellow liquid which is obtained by solvent extraction from coriander seeds. It contains a number of phytochemicals including linalool, borneol, cumene, geraniol, terpineol, quercetin, kaempferol, caffeic acid, p-coumaric acid, cinnamic acid, vanillic acid, ferulic acid, rutin, tocopherols, pyrogallol, quercetin, 1,8-cineole, α-terpinene, β-pinene, β-sitosterol, β-carotene, γ-terpinene, nerol, and limonene (Table 10) (Kaefer and Milner 2008; Leja and Czaczyk 2016; Przygodzka et al. 2014; Yashin et al. 2017). The major constituents of the coriander oil are linalool (60 to 90%) and monoterpene hydrocarbon (15–20%) mainly γ-terpene and geranyl acetate (Figure 10). Msaada et al. (2009) identified 41 compounds in different maturation stages of coriander fruits and reported that the yield of its essential oil increased during maturation stages, mainly linalool (36–78%). Sudanese coriander oils contained 78 compounds (mainly sabinene, camphor, and cis-β-ocimene), Korean coriander essential oils contained 39 components (mainly cyclododecanol, tetradecanal, and 2-dodecenal), whereas Bangladeshi coriander leaf oils had at least 44 compounds (mainly 2-decenoic acid, trans-11-tetradecenoic acid, and capric acid) (Al-Snafi 2016a). Coriander seed is a rich source of phenolics (594.780 ± 42.685 mg GAE/g) and flavonoids (10.560 ± 0.545 mg QE/g) (Tacouri et al. 2013). Moreover, Msaada et al. (2017) extracted the phenolics as gallic acid equivalents (GAE) and reported that Syrian coriander had the higher total phenolic content (1.09 mg GAE/g) than Tunisian (1.00 mg GAE/g) and Egyptian (0.94 mg GAE/g) coriander extracts. These included phenolic acids (38.66–81.47%), gallic acid (7.93–13.81%), chlorogenic acid (1.51–15.09%), vanillic acid (4–7.51%), ferulic acid (4.69–11.07%), and flavonoids (18.53–61.34%). Furthermore, numerous phenolics (dimethoxycinnamoyl hexoside, 5-O-caffeoylquinic acid, caffeoylquinic acid, ferulic acid glucoside, p-coumaroylquinic acid, quercetin-3-O-rutinoside, quercetin-3-O-glucoside, quercetin-3-O-glucuronide, and kaempferol-3-O-rutinoside) were reported in vegetative parts of coriander, while coriander fruit contained mainly caffeoyl N-tryptophan hexoside and p-coumaric acid (Barros et al. 2012). In addition, alkaloids, flavonoids, tannins, glycosides, saponins, sterols, and coumarins were reported in coriander extract (Bhat and Al-Daihan 2014; Kumar et al. 2014; Patel and Vakilwala 2016; Pathak et al. 2011; Ramkissoon et al. 2016; Sibi et al. 2013; Thangavel et al. 2015).
 Click to view | Table 10. Phenolic composition of coriander |
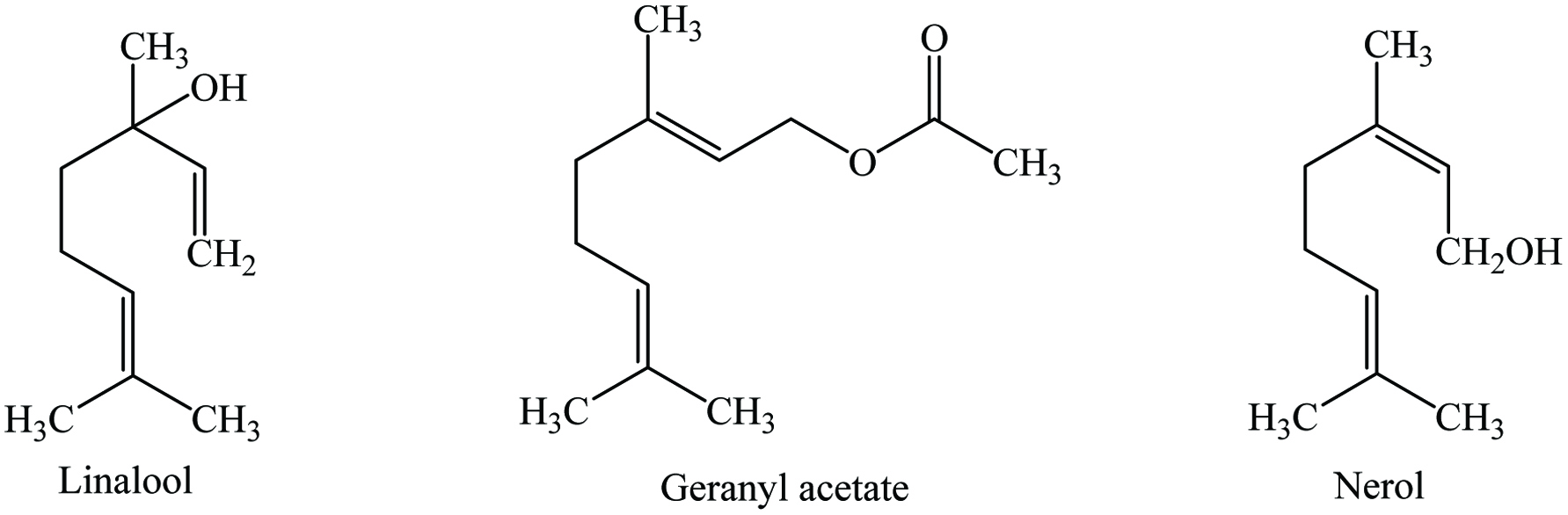 Click for large image | Figure 10. Major phytochemicals of coriander. |
2.11. Cumin
Cumin (Cuminum cyminum L) belongs to the family Apiaceae and is known as Cuminum odorum Salisb, Selinum cuminum L, Ligusticum cuminum L, Crantz, and Krause. Cumin is indigenous to the eastern Mediterranean region, northern Egypt, and India and also widely grown in Iran, Syria, Pakistan, and China. It is a glabrous annual plant and can grow up to 2 ft high with blue-green linear leaves. This slender herb has white or pink flowers and ovoid shaped brownish or yellow fruit containing a single seed. The seeds (ground or whole), essential oil, and oleoresin are the major products of this herb (Charles 2013).
Cumin contains 7–8.06% moisture, 15–18% protein, 4–22.27% lipid, 29–44.24% carbohydrates, 6% ash, 10.5–17% fiber, and 2.3–5% essential oil. Numerous phytochemicals such as α-pinene, β-pinene, cuminaldehyde, carvone, γ-terpinene, p-cymene, β-carotene, 1,8-cineole, β-sitosterol, carvaol, caffeic acid, carvacrol, geranial, p-coumaric acid, kaempferol, limonene, quercetin, thymol, and tannin have been reported in cumin (Table 11) (Embuscado 2015; Kaefer and Milner 2008; Leja and Czaczyk 2016). Chemical structures of most abundant compounds found in cumin are shown in Figure 11. The essential oil of cumin roots, stem and leaves, and flowers are 0.03, 0.1, and 1.7%, respectively, and the major compounds of these oils are bornyl acetate (around 23%), α-terpinene (about 34%), and γ-terpinene (nearly 51%) (Bettaieb et al. 2010). In addition, the maximum essential oils of cumin were obtained at the mature stage (4.3%), while the opposite scenario was seen at the immature stage (2.7%) (Moghaddam et al. 2015). Besides, the constituents of cumin essential oil vary from region to region, thus the major components in Turkish and Syrian cumin are cuminaldehyde, p-mentha-1,4-dien-7-al, and γ-terpinene, whereas Egyptian cultivars have aldehyde and tetradecene, Tunisian cumin contains cuminlaldehyde and γ-terpinene, Indian varieties have trans-dihydrocarvone and γ-terpinene, Chinese cumin contains cuminal and cuminic alcohol, and Iranian cumin has thymol and γ-terpinene (Al-Snafi 2016b). Moreover, Abdelfadel et al. (2016) reported that hot extracts of cumin seed showed significantly a higher content of phenolics compared to cold extracts (299.0 and 270.3 mg GAE/100 mL, respectively), whereas Ereifej et al. (2016) showed that methanolic extracts demonstrate the highest phenolics (43.8 mg GAE/100 g) than ethanol and acetone extracts at 20 °C. In addition, cumin oleoresin (2.5 to 4%), prepared by solvent extraction, is brownish to yellowish-green in color.
 Click to view | Table 11. Phenolic composition of cumin |
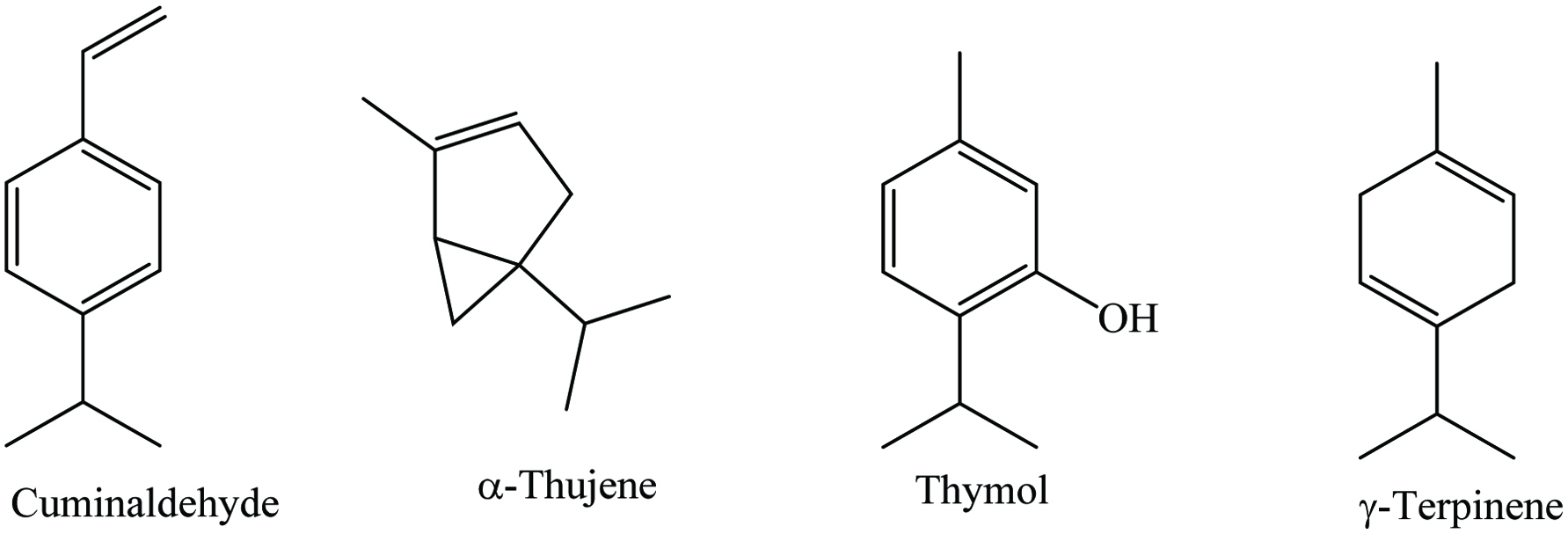 Click for large image | Figure 11. Major phytochemicals of cumin. |
2.12. Curry leaf
Curry leaf (Murraya koenigii Spreng) belongs to the family Rutaceae and is known as Bergera koenigii, Chaleos koenigii, Indian curry tree, and Indian bay. It is a perennial leaf vegetable indigenous to India, Bangladesh and Sri Lanka. This deciduous aromatic shrub grows up to 20 ft high with glossy dark leaves. The fresh and dried leaves are used whole, crushed, or chopped as spice ingredients.
Curry leaf contains 66.3% moisture, 1.0–6.1% protein, 1% lipid, 16–18.17% carbohydrate, 6.4% fibr, and 4.2% ash. The major components of the essential oil (nearly 2.5%) are β-caryophyllene, β-elemene, β-gurjunene, β-phellandrene, α-pinene, sabinene, and β-pinene (Table 12) (Jain et al. 2017; Nayak et al. 2010; Verma et al. 2013). Chemical structures of most abundant compounds found in curry leaf are shown in Figure 12. Numerous phytochemicals have been reported in curry leaf including flavonoids (7.43 ± 0.03 mg/100 g), alkaloids (1.90 ± 0.01 mg/100 g), saponins (2.50 ± 0.01 mg/100 g), tannins (0.86 ± 0.02 mg/100 g), and glycosides (0.11 ± 0.01 mg/100 g) (Igara et al. 2016; Ramkissoon et al. 2016; Vats et al. 2011). Sasidharan and Menon (2011) extracted total phenolics and evaluated antioxidant activity of curry leaf by using hexane, ethanol, chloroform, ethanol-water, and water at 25 °C and reported ethanol-water extract had the highest amount of phenolics (501 ± 4.6 mg GAE/g) and DPPH radical scavenging activity (82%) at 10 μg/mL level. Ghasemzadeh et al. (2014) collected curry leaves from three different places in Malaysia and reported that the maximum amount of phenolics and flavonoids were present in Kelantan extracts (3.771 and 14.371 mg/g), followed by Selangor (3.146 and 12.272 mg/g) and Johor (2.801 and 12.02 mg/g). In addition, among the three morphotypes of Murraya Koenig from Uttarakhand, gamthi type showed significantly higher phenolics (532.8 ± 2.81 mg/g) and flavonoids (6.01 ± 3.21 mg/g) followed by dwarf type (168.2 ± 2.58 and 4.82 ± 1.58 mg/g, respectively) and the regular type (111.6 ± 3.85 and 3.58 ± 2.33 mg/g, accordingly) (Sivakumar and Meera 2013). Besides, Singh et al. (2011) identified and quantified the major flavonols (5,752–11,187 µg/g) in curry leaf by LC-MS-MS, including myricetin-3-galactoside (325–391 µg/g), quercetin-O-pentohexoside (709–1,166 µg/g), quercetin-3-diglucoside (92–135 µg/g), quercetin-3-O-rutinoside (86–122 µg/g), quercetin-3-glucoside (1,435–2,863 µg/g), quercetin-3-acetylhexoside (13–51 µg/g), quercetin-O-xylo-pentoside (48–1,270 µg/g), kaempferol-O-glucoside (129–682 µg/g), kaempferol-aglucoside (669–5,559 µg/g), and quercetin (146–436 µg/g).
 Click to view | Table 12. Phenolic composition of curry leaf |
 Click for large image | Figure 12. Major phytochemicals of curry leaf. |
2.13. Dill
Dill (Anethum graveloens Linn.) belonging to the family of Apiaceae and is also known as Anethum arvense Salis, Peucedanum sowa Kurz, P. graveolens (L) Hiern, Selinum anethum Roth, and garden dill. It is an annual aromatic branched herb and native to south-east Europe and West Asia; it is also cultivated commercially in Egypt and the USA. It can grow up to 3 ft high with green leaves, yellow flowers, and small pungent fruit. Dill seed, dill weed, and oleoresin are the main products of this annual herb (Charles 2013).
The dill seed has been reported to contain 7–9% moisture, 16–18% protein, 14–20% lipid, 35–55% carbohydrate, 20–30% total dietary fiber, and vitamins as well as 1–8% essential oil. It contains a wide range of phytochemicals mainly quercetin, kaempferol, catechins, myricetin, isorhamnetin, limonene, carvone, α-pinene, α-phellandrene, limonene, linalool, myristicin, and α-thujene (Table 13) (Chahal et al. 2017b; Goodarzi et el. 2016; Kaefer and Milner 2008; Shekhawat and Jana 2010; Yashin et al. 2017). Chemical structures of most abundant compounds found in dill seed are shown in Figure 13. Recently, Chahal et al. (2017b) compared the dill seed oil with different geographic regions from different scientific articles and described the major components of Iranian, Egyptian, Pakistani, Canadian, Indian, Chinese, Uzbekistan’s, and Thailand’s dill seed oil are α-phellandrene (0.03–18.36%), p-cymene (0.10–2.34%), trans dihydrocarvone (0.30–14.70%), limonene (1.10–83.0%), and carvone (20.73–55.20%). Moreover, Vokk et al. (2011) identified 25 compounds from Estonian dill and dill seeds and reported the content of its principal components; α-phellandrene, and β-phellandrene, and also dill ether was higher in summer, whereas the percentage of carvone and α-bergaptene was maximum in winter. In addition, microwave heating changed the bioactive compounds of dill leaf. The highest amount of phenolics (1,287 mg GAE/100 g), antioxidants activity (DPPH, 50.71%), chlorophyll (34.6 mg/kg) and carotenoid (48.52 mg/kg) contents were observed upon one min of heating, then it started to decrease gradually (Kamel 2013). Furthermore, the highest content of phenolics, phenolic acids (mainly chlorogenic acid, p-coumaric acid, benzoic acid, salicylic acid, and ellagic acid) and flavonoids was observed in fresh dill extracted with 50% acetone compared to methanol (Świeca and Gawlik-Dziki 2008). Besides, dill seeds, roots, and leaves are also rich in tannins, terpenoids, saponins, steroids, flavonoids, phlobatannin, cardiac glycoside, and anthraquinone (Jana and Shekhawat 2010; Kerrouri et al. 2016; Shekhawat and Jana 2010).
 Click to view | Table 13. Phenolic composition of dill |
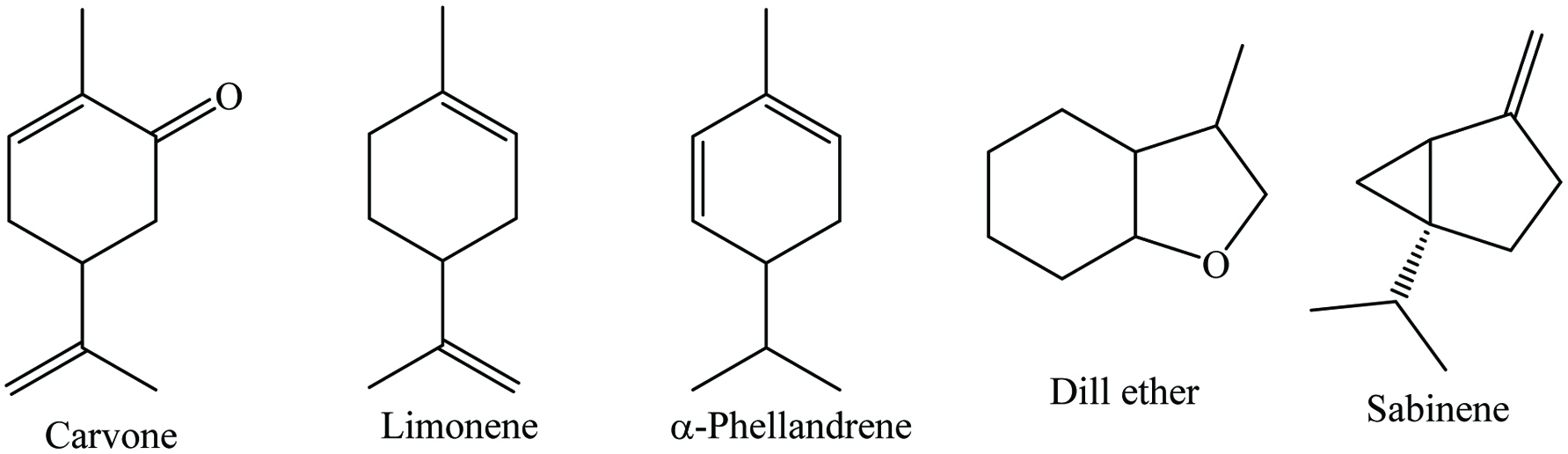 Click for large image | Figure 13. Major phytochemicals of dill. |
2.14. Fennel
Fennel (Foeniculum vulgare Mill) belongs to the family Apiaceae and its most common synonyms are Foeniculum capillaceum, Foeniculum officinale, Anethum foeniculum, fenkel, sweet fennel, and common fennel. It is indigenous to southern Europe and the Mediterranean region and is cultivated all over the world, particularly China, India, Egypt, Turkey, Central Europe, Argentina, and the USA. It is a perennial plant which can grow up to 6 ft high with green leaves and yellow flowers on short pedicels. The seeds are grooved, slightly curved, yellowish, and oval in shape. The seed, herb, and essential oil are used as spices.
It has been reported that fennel fruit contains 8.8% moisture, 15.8% protein, 14.9% lipid, 36.6% carbohydrates, 15.7% fiber, 8% ash (Ca, Na, Mg, Fe, K, P, and Zn), and 6% essential oil (Bernath et al. 1994). Moreover, the fruit contains a fixed oil (15–30%) and a 12% volatile essential oil; the principal component of the essential oil is trans-anethole (65–70%). Oleoresin of fennel is obtained by solvent extraction of whole seeds and contains a volatile oil around 60%. Numerous phytochemicals have been characterized in fennel, of which trans-anethole, estragole, fenchone, α-pinene, 1,8-cineole, myristicin, limonene, β-carotene, β-sitosterol, cinnamic acid, ferulic acid, fumaric acid, benzoic acid, caffeic acid, p-coumaric acid, vanillic acid, kaempferol, quercetin, rutin, and vanillin are important (Table 14) (Badgujar et al. 2014; Hossain et al. 2011a; Kaefer and Milner 2008; Kochhar 2008; Kunzemann and Herrmann 1977; M. Lu et al. 2011; Muckensturm et al. 1997; Rather et al. 2016). Other studies have shown that phytochemicals of the fennel extract contain alkaloids, flavonoids, saponins, glycosides, phenols, terpenoids, and tannins (Bano et al. 2016; Kaur and Arora 2009; Sibi et al. 2013; Tacouri et al. 2013). Moreover, Miraldi (1999) determined the essential oil contents of ten fennel samples (dry and ripe fruits) from different origins; 16 main components were identified by GC-MS mainly trans-anethole, limonene, estragole, and fenchone (Figure 14). Furthermore, essential oil, mainly monoterpenes, α-pinene, α-terpinene, β-myrcene, and limonene which their contents varied significantly during maturation stages (Telci et al. 2009). Similarly, Anwar et al. (2009b) observed maximum (3.5%) and minimum (2.8%) essential oil content in the mature and immature fruit, respectively. This includes the presence of trans-anethole (65.2, 69.7, and 72.6%), limonene (3.5, 4.7, and 7.8%), fenchone (8.8, 10.0, and 11.0%), and estragole (6.9, 6.9, and 7.2%), as the major constituents at immature, intermediate, and mature stages, respectively. Besides, cultivated fennel had a high content of terpenes compared to wild fennel (Conforti et al. 2006). Similarly, the cultivated fennel had the highest amount of phenolics (3.1%) and flavonoids (1.6%) than the wild fennel (Ghanem et al. 2012). Recently, Salami et al. (2016a) studied 23 fennel populations from different geographical regions of Iran to determine the effect of self-pollination on essential oil yield, antioxidant activity, total phenolic, and flavonoid contents. Results suggested that self-pollination increased the essential oil yield (25.61%), antioxidant activity (6.23%), TPC (21.66%), and TFC (49.40%). In another study, Salami et al. (2016b) determined the phenolic compounds of 23 fennel from different geographical places, Kh1 from Iran demonstrated the maximum TPC (262 mg/g), TFC (14.8 mg/g), and antioxidant activity (IC50=76µg/mL). In addition, they analyzed the major phenolic acid (caffeic, chlorogenic, and 1,5-dicaffeoylquinic acids) and flavonoid (apigenin, quercetin, and rutin) compounds by HPLC. Besides, Križman et al. (2007) determined the phenolic compounds of fennel by using a reversed-phase high-performance liquid chromatography (HPLC), the major components were 3-O-caffeoylquinic acid, 4-O-caffeoylquinic acid, chlorogenic acid, eriocitrin, miquelianin, rutin, 1,3-O-dicaffeoylquinic acid, 1,4-O-dicaffeoylquinic acid, 1,5-O-dicaffeoylquinic acid, and rosmarinic acid. In another study, Rawson et al. (2013) examined the influence of boiling (100 °C for 30 min) and roasting (16 °C for 15 min) on the levels of polyphenols; a significant decrease of gallic acid, caffeic acid, apigenin-7-O-glucoside, syringic acid, ferulic acid, isovitexin, and phloridzin was observed upon boiling and in the contents of falcarindiol-3-acetate, falcarindiol, and falcarinol upon roasting. In addition, vitamins such as ascorbic acid, α-tocopherol, β-tocopherol, γ-tocopherol, and δ-tocopherol were the most abundant in fennel shoots, leaves, stems, and inflorescences (Barros et al. 2009).
 Click to view | Table 14. Phenolic composition of fennel seed |
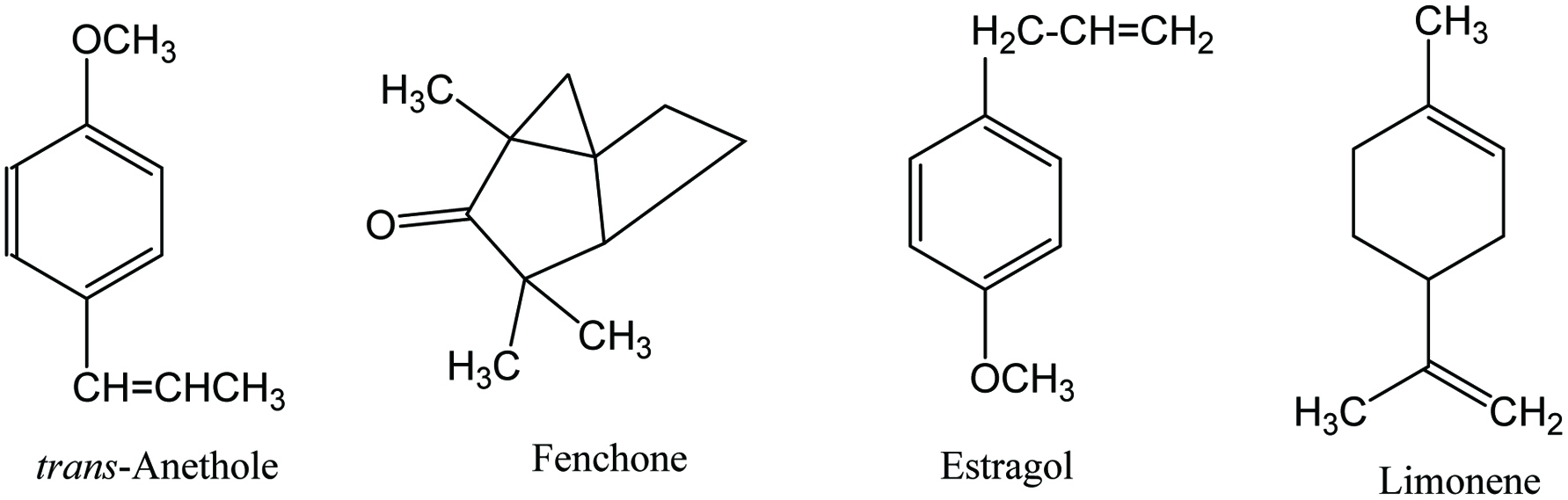 Click for large image | Figure 14. Major phytochemicals of fennel. |
2.15. Fenugreek
Fenugreek (Trigonella foenum-graecum L) belongs to the family Fabaceae (Leguminosae) and is also known as Greek hay, foenugreek, and methi. It is an aromatic herbaceous annual plant native to the Mediterranean region and south-eastern European countries and also widely cultivated in Africa, South America and Asia. It is a self-pollinated crop and grows up to 1.5 ft high with light-green leaves and white to whitish-yellow flowers. The fruit is ovoid-cylindrical, light green to yellow brown with smooth brownish seeds. Dried ripe seeds, leaves (fresh or dry), essential oil, and oleoresin are its major products (Parthasarathy et al. 2008).
The fenugreek fruit (seed) contains 8–10% moisture, 15–28% protein, 6–12% lipid, 35–45% carbohydrates, 4–8% ash, 8–16% fiber, and 0.2–0.3% essential oil. Fenugreek oleoresin is a brownish-yellow viscous liquid which is obtained from dried ripe seeds. The main components of the fenugreek seed essential oil are β-pinene, β-caryophyllene, camphor, and neryl acetate. Girardon et al. (1985) determined 39 components in the volatile oil of fenugreek seeds, including sesquiterpenes, n-alkanes, and some oxygenated components. Fenugreek contains a wide range of phytochemicals including phytosterols, terpenoids, flavonoids (naringenin, saponaretin, lilyn, kaempferol, isovitexin, orientin, vitexin, isoorientin, luteolin, and quercetin) (Figure 15), alkaloids (choline, trigonelline, and carpaine), and saponins (fenugrin, foenugracin, trigonoesides, glycoside, yamogenin, smilagenin, yuccagenin, gitogenin, sarsasapogenin, hederagin, tigonenin, diosgenin, and neotigogenin) (Table 15) (Ahmad et al. 2016; Anbumalarmathi et al. 2016; Kumaravel and Alagusundaram 2014; Kumari et al. 2016; Madhava Naidu et al. 2011; Mahmood and Yahya 2017; Mandegary et al. 2012; Nandagopal et al. 2012; Wani and Kumar 2016). In addition, Yashin et al. (2017) stated that the major phytochemicals in fenugreek were sesquiterpenes, terpenes, and aromatic aldehydes. Moreover, Kenny et al. (2013) quantified 18 phenolic components in fenugreek seeds by using UPLC-MS and the major flavonoids were listed as apigenin-7-O-glycoside (1955.55 ng/mg) and luteolin-7-O-glycoside (725.50 ng/mg). In another study, Pasricha and Gupta (2014) reported that fenugreek stem contained the highest amount of phenolics (40.19 µg gallic acid equivalents/mg) and flavonoids (7.51 µg catechin equivalents/mg) compared to fenugreek leaves. The same study identified the secondary metabolites by GC-MS; the major compounds in the leaves were n-hexadecanoic acid, 9,12,15-octadecatrien-1-ol, (Z,Z,Z), nonanoic acid, and octadecanoic acid.
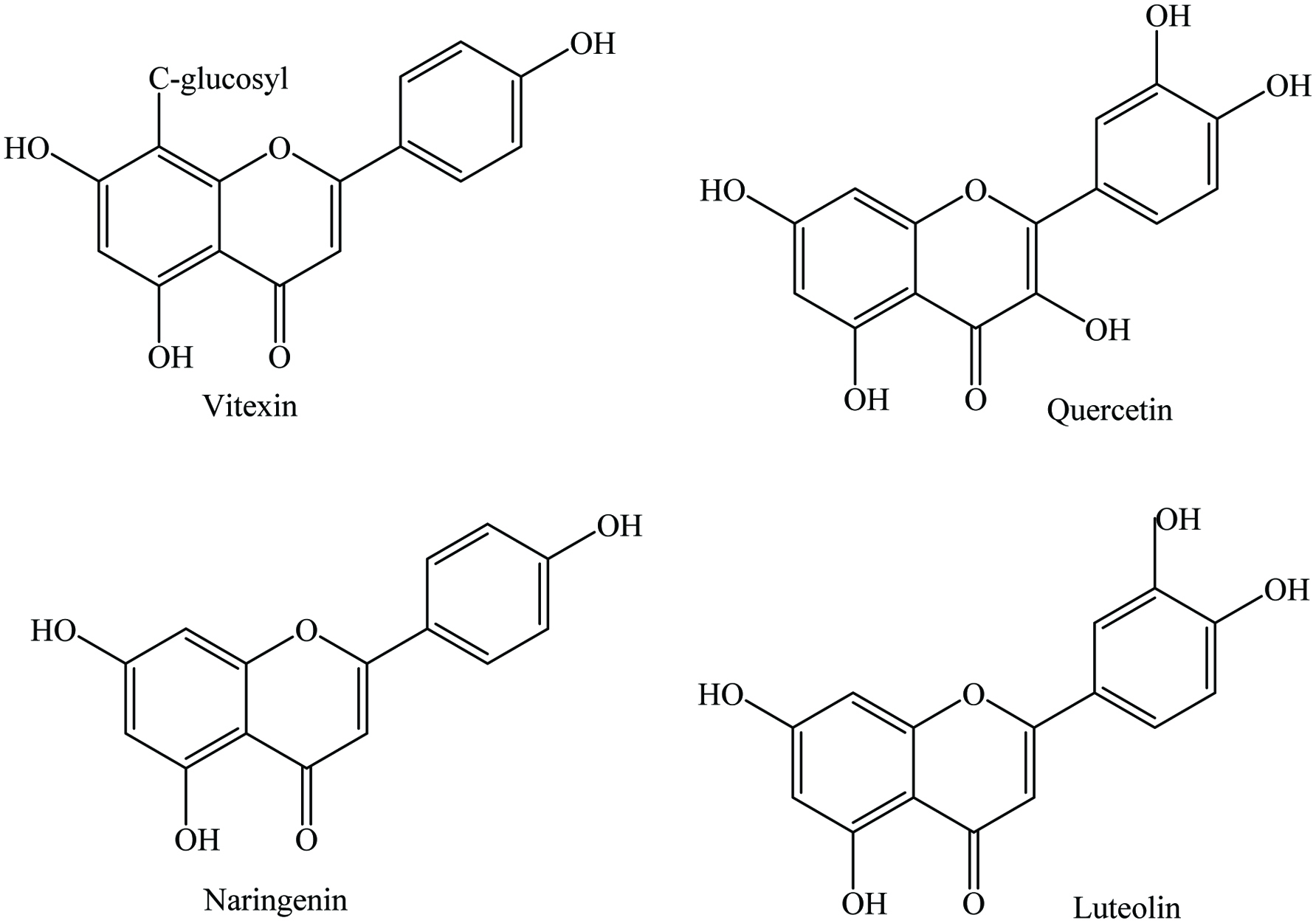 Click for large image | Figure 15. Major flavonoids of fenugreek. |
 Click to view | Table 15. Phenolic composition of fenugreek seed |
2.16. Garlic
Garlic (Allium sativum L) belongs to the family of Amaryllidaceae (Liliaceae and Alliaceae) and the second most broadly cultivated Allium after onion. It is native to Asia (mainly China, India, Korea, and Bangladesh) and also Spain, Argentina, Egypt, and the USA which are the major garlic producing countries. It is a perennial herb and grows up to 1 ft high with flat leaves and small white flowers and bulbils. The shape of garlic is round, smooth, and solid. Whole bulbs, dried as granules, cloves, flakes, garlic oil, and garlic juice are the main products of garlic.
Garlic powder contains 5–6.45% moisture, 16.5–17.5% protein, 71–73% carbohydrates, 1.8–2% fiber, ash, and vitamins. In addition, volatile oils of garlic comprise 0.1–0.4% of the fresh weight of garlic and the major constituents in the oil are mainly sulfur compounds (Figure 16). Numerous phytochemicals have been identified in garlic, including flavonoids, terpenoids, alkaloids, coumarins, saponins, and tannins (Table 16) (Boukeria et al. 2016; Huzaifa et al. 2014; Mikail 2010; Otunola et al. 2011; Ramkissoon et al. 2016; Siddhartha et al. 2017; Tacouri et al. 2013). In addition, the major chemical constituents of garlic are allicin, diallyl disulfide, diallyl sulfide, diallyl trisulfide, S-allyl cysteine, and allyl isothiocyanate (Kaefer and Milner 2008; Kim et al. 2017; Yashin et al. 2017). The bioactive compounds of garlic, particularly polyphenols, vary from cultivar to cultivar and geographical region (Beato et al. 2011; Szychowski et al. 2018). Chen et al. (2013) determined the phenolic and flavonoid contents from 43 (China-40, Ethiopia-1, Thailand-1, and Korea-1) garlic cultivars and reported that ‘74-x’ (Chinese cultivar) had the highest phenolics content and the strongest antioxidant activity. In addition, Bhandari et al. (2014) analyzed the phytochemical components (alliin, vitamin C, phenolics, and flavonoids), free sugars, and antioxidant capacity of 19 garlic cultivars; the yield was found to be related to the cultivar. However, thermal processing had an effect on both bioactive compounds and antioxidant activity; boiling was found to be less destructive than frying, whereas fresh garlic had the highest amount of bioactive compounds. For example, raw, boiled, and fried garlic had 121.82 ± 3.11, 75.37 ± 2.82, and 68.07 ± 1.81 mg GAE/100 g phenolics, respectively, whereas the amount of flavonoids was 123.3 ± 8.4, 112.4 ± 3.9, and 93.9 ± 5.4 mg QE/kg (de Queiroz et al. 2014). Besides, fresh garlic had the highest phenolic content than chopped without salt, chopped with salt, fried, and dehydrated garlic samples (Queiroz et al. 2009). Moreover, aged-garlic extract exhibited a significantly higher phenolic content (5.62 mg GAE/g) compared to raw or heated garlic extract (Park et al. 2009). Furthermore, Kallel et al. (2014) extracted the phenolic compounds from industrially disposed garlic husk by different solvents; the highest amount of phenolics was obtained by methanol-water (1:1, v/v) compared to other solvents. Recently, Fedosov et al. (2016) identified the phenolic profile of garlic extracts by HPLC, the compounds were luteolin-7-glucoside, apifenin-7-glucoside, luteolin, hyperoside, chlorogenic acid, caffeic acid, rosmarinic acid, and rutin.
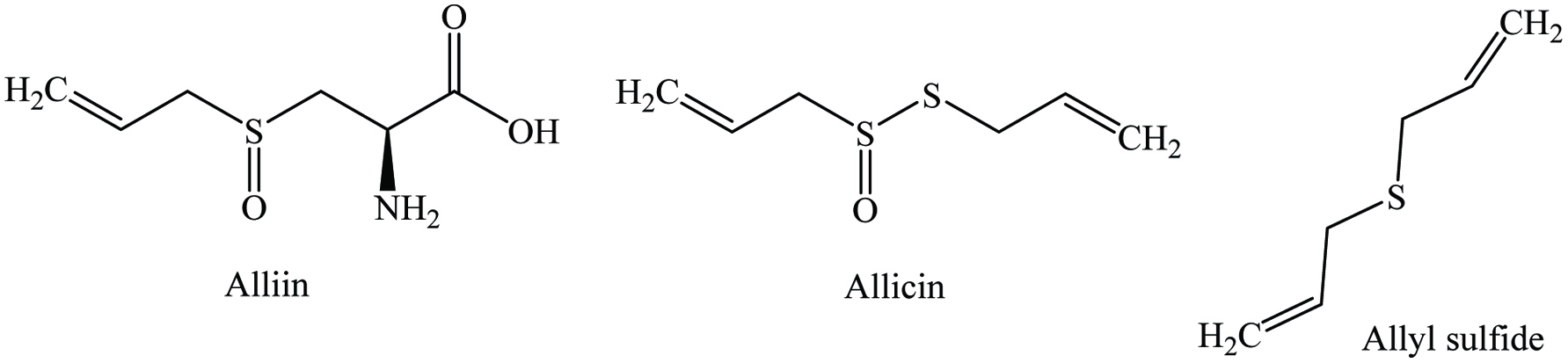 Click for large image | Figure 16. Major phytochemicals of garlic. |
 Click to view | Table 16. Phenolic composition of garlic |
2.17. Ginger
Ginger (Zingiber officinale Rosc) is underground stems or rhizomes belonging to the family Zingiberaceae. The common synonyms are Zingiber zingiber (L) Karst, Amomum zingiber L, Jamaican ginger, common ginger, shunthi, and ardraka. It is a monocotyledon herb indigenous to tropical and subtropical countries such as India, Bangladesh, Sri Lanka, China, Japan, Africa, Jamaica, and Australia. This perennial herb grows up to 4 ft high with leafy shoots and white or yellow flowers. Rhizome (fresh or dried), ground ginger (powder), essential oil, oleoresin are the main products of ginger (Bailey-Shaw et al. 2008; Varakumar 2017).
It has been reported that dried ginger contains 7.0–10.90% moisture, 8.50–12.4% protein, 70–72% carbohydrate, 5.70–6.64% ash, 7.17–14.1% fiber, and 1.8–4.0% volatile oil (Charles 2013; Peter and Kandiannan 1999). Due to the presence of more than 80 components in the volatile oil of stem, ginger provides a pleasant aroma. The main components of the essential oil are zingiberene (40%) as well as α-curcumene (20%). Ginger contains a number of phytochemicals, mainly gingerol, geraniol, curcumin, geranial, paradol, borneol, zingerol, zingiberon, zingiberene, linalool, camphene, shogoal, gamma-terpinene, terpinen-4-ol, ascorbic acid, β-carotene, p-coumaric-acid, and caffeic acid (Table 17) (Cheng et al. 2011; Embuscado 2015; Jiang et al. 2005; Kaefer and Milner 2008; Leja and Czaczyk 2016; Suhaj 2006; Yashin et al. 2017). Chemical structures of most abundant compounds found in ginger are shown in Figure 17. Moreover, Yeh et al. (2014) examined two Taiwanese ginger and identified the gingerol compounds which included 6-shogaol, 6-gingero, 8-gingerol, 10-gingerol, and curcumin. Furthermore, they identified 60 to 65 compounds from the essential oils, mainly camphene, α-curcumene, sabinene, zingiberene, β-sesquiphellandrene, afarnesene, neral, and geranial. In another study, Chari et al. (2013) stated that enzyme-assisted extraction (α-amylase or viscozyme) of ginger demonstrates higher yield of gingerol (12.2 ± 0.4%) and oleoresin (20 ± 0.5%) compared to the control sample. Similarly, Varakumar et al. (2017) stated that the samples treated with enzymes (accellerase) showed increased yield of 6-, 8-, and 10-gingerols as well as 6-shogaol. However, drying had an effect on gingerol content; Bartley and Jacobs (2000) reported that drying reduced the content of gingerol, increased the terpene hydrocarbons and also assisted in converting some of the monoterpene alcohols to their corresponding acetates. Similarly, Cheng et al. (2011) compared fresh, dried, and steamed ginger; results suggested that steaming (120 °C) reduced the level of gingerol and increased the concentration of shogaol which is ultimately improved the anticancer potential. In another study, Kou et al. (2018) claimed that ionic liquid-based ultrasonic-assisted extraction (ILUAE) notably increased the yield of total gingerols and reduced the extraction time compared to the traditional extraction methods. In addition, red ginger contained a higher amount of vitamin C (1.83 ± 0.09 mmol/100 g), phenolics (95.34 ± 8.44 mg/100 g), and flavonoids (53.67 ± 0.30 mg/100 g) than the white ginger (Oboh et al. 2012). Besides, Sanwal et al. (2010) identified 6-gingerol as a main pungent phenolic component in 18 different Indian ginger varieties, whereas 8- and 10-gingerols occurred in lower levels. In addition, gallic acid and quercetin were the most abundant phenolic acid and flavonoid in Malaysian young ginger cultivars, respectively (Ghasemzadeh et al. 2010b). Similarly, Ghasemzadeh et al. (2010a) described that the amount of phenolics, flavonoids, and antioxidant activity in Malaysian young ginger was higher in leaf followed by rhizome and stem. In addition, ginger oleoresin is a dark brown viscous liquid which contains 20–25% volatile oil, mainly monoterpenes and sesquiterpenes. Gingerol and shogaol were the major components of the oleoresin, which exhibited significant antioxidative and antimicrobial properties (Bailey-Shaw et al. 2008; Murthy et al. 2015; Varakumar et al. 2017).
 Click to view | Table 17. Phenolic composition of dried ginger |
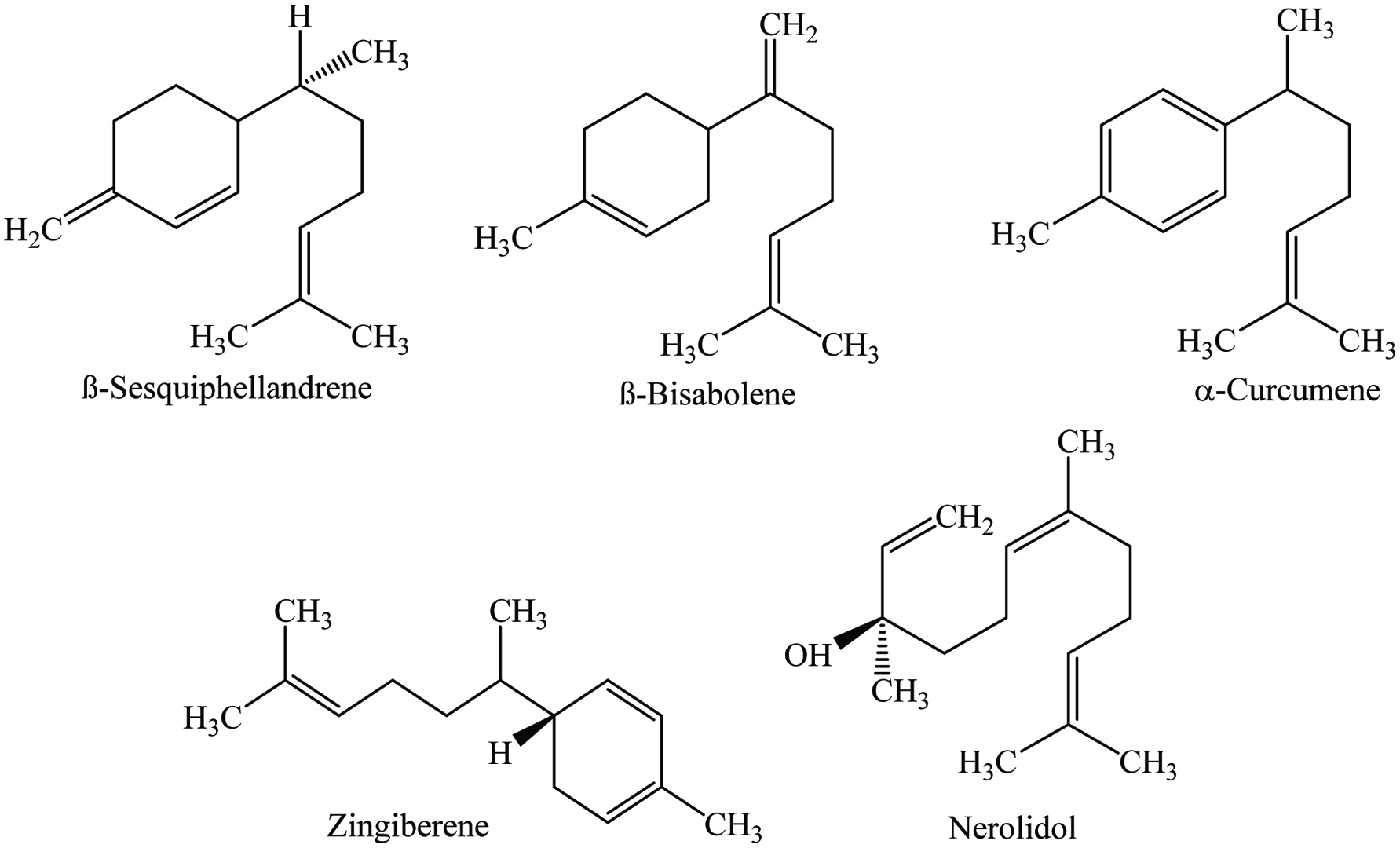 Click for large image | Figure 17. Major phytochemicals of ginger. |
2.18. Marjoram
Marjoram (Origanum majorana L) is in the family of Lamiaceae and is also known as Majorana hortensis and knotted marjoram. It is native to the Mediterranean countries and also UK, USA, North Africa, and Asia which are the major marjoram producing countries. It is a perennial herbaceous plant and grows up to 2 ft high with dark green oval leaves and red or white flowers. The dried herb is light green with highly aromatic flavor. The fresh leaves (whole or chopped), dried leaves (whole or ground), essential oils and oleoresins are the main products.
The components of marjoram include 6.5–8% moisture, 60.56–64.40% carbohydrate, 12.50–14% protein, 6–24% ash, 20–22% fiber, 5.50–5.60% fixed oil, and 1.8–3.5% essential oil (Peter 2001). The main components of the essential oils are γ-terpinene (20%), terpin-4-ol (20%), sabinene hydrate (12–15%), α-terpineol, linalool, and sabinene. Numerous phytochemicals have been identified in marjoram, of which the main ones are pinene, limonene, eugenol, 1,8-cineole, carvacrol, terpinene, farnesol, thymol, apigenin, β-carotene, β-sitosterol, myrcene, and p-cumene, as well as caffeic, syringic, ferulic, sinapinic, rosmarinic, vanillic, and 4-hydroxybenzoic acids (Table 18) (Bina and Rahimi 2017; Embuscado 2015; Kaefer and Milner 2008; Leja and Czaczyk 2016; Muchuweti et al. 2007; Suhaj 2006; Yashin et al. 2017). Vera and Chane-Ming (1999) identified 45 essential oil components from Reunion Island’s marjoram by GC-MS and GC-FTIR, mainly terpinen-4-ol (38.4%), cis-sabinene hydrate (15.0%), γ-terpinene (6.9%), and p-cymene (7.0%). Moreover, Beltrame et al. (2013) studied the photodegradation profile of marjoram essential oil and reported that chemical constituents like 2 undecanone and p-diisopropylbenzene as being its major compounds. Furthermore, Sellami et al. (2009) harvested sweet marjoram at four phenological stages and identified 38 essential oil constituents by GC and GC/MS; these were mainly oxygenated monoterpenes (64.01–71.4%), sesquiterpene hydrocarbons (1.47–4.05%), and monoterpene hydrocarbons (21.73–29.92%). The major essential oil components were terpinen-4-ol, trans-sabinene hydrate, γ-terpinene, cis-sabinene hydrate, bornyl acetate, and linalool (Figure 18), whereas the main phenolic compounds were trans-2-hydroxycinnamic, chlorogenic, rosmarinic, vanillic, gallic, cinnamic acid and also amentoflavone, luteolin, apigenin, quercetin, coumarin, and rutin. However, salinity had an effect on growth, essential oil yield, and the content of phenolic compounds of marjoram (Baâtour et al. 2013; Jelali et al. 2011). Therefore, the content of chlorophyll and rate of plant growth were significantly decreased with increasing of NaCl levels, whereas the yield of essential oil increased notably up to 55.5% with 5 ds/m NaCl and decreased at high salinity (Jelali et al. 2011). Besides, methanol was an important extraction solvent to extract bioactive compounds from marjoram and the major phenolic acids were methyl rosmarenate, rosmarenic acid, caffeic acid, chlorogenic acid, ferulic acid, cinnamic acid, and quinic acid, whereas luteolin, apigenin, and quercetin were the main flavonoids (Dhull et al. 2016; Roby et al. 2013b).
 Click to view | Table 18. Phenolic composition of dried marjoram |
 Click for large image | Figure 18. Major phytochemicals of marjoram. |
2.19. Nutmeg and mace
Nutmeg (Myristica fragrans Houtt) belongs to the family of Myristicaceae and is also known as Myristica moschata Thunb, Myristica aromatic Swartz, Myristica officinalis L, Myristica amboinensis Gand, and myristica. Nutmeg and mace are two different important parts of the same fruit of this plant. The dried kernel of the seed is known as nutmeg, whereas mace is the dried aril surrounding it. Nutmeg is indigenous to the Banda Islands in Indonesia and is also cultivated in India, Malaysia, Sri Lanka and West Indies. This dioecious evergreen plant can grow up to 70 ft high with yellowish flowers, green leaves and pendulous yellowish fruit. Nutmeg (ground or grated), mace, essential oil, and oleoresins are used as a spice (Morsy 2016).
Nutmeg contains 6–14% moisture, 5–7.5% protein, 28–49% carbohydrate, 11–21% fiber, and ash. It contains 25–50% lipids as fixed oil containing mainly petroselinic, myristic, and palmitic acids and also has around 16% essential oil comprising α-pinene, β-pinene, sabinene, and myristicin, whereas the main compound of oleoresins is elemicine. Nutmeg oleoresin is prepared by solvent extraction which is used as a coloring and flavoring agent in the food industry. Nutmeg oleoresin extracted with ethanol yielded 18 to 26% of oleoresins, but use of the prohibited solvent benzene afforded a yield of 31–37% (Peter 2001). Moreover, 53 components were identified in nutmeg oleoresin, mainly sabinene, α-pinene, β-pinene, myristicin, elemicin, terpinen-4-ol, limonene, and myristic acid (Morsy 2016). However, the major chemical constituents of nutmeg are trimyristin, myristicin, terpinen-4-ol, limonene, 1,8-cineole, safrole, eugenol, linalool, elemicin, catechins, lignans, myricetin, orgentin, and caffeic acid (Table 19) (Asgarpanah and Kazemivash 2012; De Soysa et al. 2016; Embuscado 2015; Kaefer and Milner 2008; Parthasarathy et al. 2008; Sibi et al. 2013; Suhaj 2006; Yashin et al. 2017). Chemical structures of most abundant compounds found in nutmeg are shown in Figure 19. In addition, Singh et al. (2005b) identified 49 phytochemicals from mace essential oil by GC-MS, mainly sabinene (20.22%), safrole (10.32%), terpinen-4-ol (12.08%), α-pinene (9.7%), γ-terpinene (5.93%), and β-phellandrene (6.56%). In another study, Hou et al. (2012) isolated six constituents from nutmeg, namely licarin B, malabaricone B, malabaricone C, dehydrodiisoeugenol, β-sitosterol, and daucosterol. Among them, malabaricone C exhibited a stronger antioxidant activity. Besides, nutmeg seed contained the maximum amount of phenolics followed by mace, skin, as well as pulp (Tan et al. 2013).
 Click to view | Table 19. Phenolic composition of nutmeg seed |
 Click for large image | Figure 19. Major phytochemicals of nutmeg. |
2.20. Onion
Onion (Allium cepa L) is a common spice commodity belonging to Liliaceae family. The synonyms are bulb onions, potato onion, multiplier onions, tree onion, shallots and palandu. It is native to Asia and the Mediterranean region but is grown all over the world. Generally, it is a biennial herb and can grow up to 3 ft high with erect stems and flowers. The leaves are cylindrical blue-green color, whereas flowers are white or pink. Bulb (fresh, dry or frozen), leaves, and essential oil are the main products.
Generally, fresh onion contains 88.6–92.8% moisture, 0.9–1.6% protein, 0.10–0.20% lipid, 5.2–9.0% carbohydrate, 0.5–0.6% ash, and vitamins. A number of phytochemicals have been identified in onion, including flavonoids, alkaloids, terpenoids, saponins, and tannins (Table 20) (Begum and Yassen 2015; Corea et al. 2005; Ramkissoon et al. 2016; Sani and Yakubu 2016). In particular, quercetin, kaempferol, apigenin, dipyridyl disulfide, rutin, quercetin-4-glucoside, cyanidin glucosides, peonidin glucosides, taxifolin, and allicin are present (Kaefer and Milner 2008; Leja and Czaczyk 2016; Yashin et al. 2017). The major flavonoids of onion are 3,4′-O-quercetin diglucoside (50–1900 mg/kg) and 4′-O- quercetin monoglucoside (36–850 mg/kg) of quercetin, which account for around 85–90% of the total flavonoids (Figure 20) (Caridi et al. 2007; Galdon et al. 2008; Lombard et al. 2005; Price and Rhodes 1997; Ren et al. 2017a). Moreover, Patil et al. (1995) analyzed the quercetin content of 75 onion cultivars by a reverse-phase high-performance liquid chromatography; the content of quercetin in red, yellow, and pink onions varied from 54 to 286 mg/kg of fresh weight. In addition, they suggested that the storage conditions and genetic factors affect the quercetin content. Furthermore, the total quercetin content decreased from the dry skin to inner rings when compared the quercetin level in dry skin, outer rings, and inner rings (Bilyk et al. 1984; Patil and Pike 1995). Red onion contained the highest amount of total phenolic content and antioxidant activity followed by yellow, white, and sweet onion bulb extracts (X. Lu et al. 2011; Bajaj et al. 1980; Prakash et al. 2007; Nile and Park 2013; Onyeoziri et al. 2016). Moreover, phenolic compounds and their antioxidant activity were determined from skin and flesh of different colored onions (pearl, yellow, red, and white); the highest content of phenolics was observed from pearl onion skins and the major compounds were quercetin, quercetin 3,4′-diglucoside, and kaempferol (Albishi et al. 2013a, 2013b). Recently, Ren et al. (2017b) analyzed the effect of conventional, organic, and mixed cultivation practices on biologically active constituents (total flavonoids, individual flavonols, total anthocyanins, individual anthocyanins, and antioxidant activity) of 6 years repeated sampling, reported that the content of bioactive compounds was higher under fully organic condition. In addition, quercetin, quercetin-4′-glucoside, total phenolic content, and radical-scavenging activity from onion skin increased dramatically after gamma irradiation at 10 kGy (Yang et al. 2012). Besides, Ren et al. (2017a) claimed that blanching (60–70 °C for 1–3 min) before drying of onions increased the retention of bioactive compounds and antioxidant activity. However, industrial wastes such as residues, surpluses, juice, paste, and bagasse of onion showed a high content of total phenolics, flavonoids, quercetin aglycone, and antioxidant activity (Benítez et al. 2011; Roldán et al. 2008). Yalcin and Kavuncuoglu (2014) analyzed the volatile compounds from onion seeds by GC and GC-MS; the amount of oil was 21.86 to 25.86 %; composed mainly of linoleic acid (49.42–60.66 %), oleic acid (27.05–31.52%), and palmitic acid (7.23–12.2%). In addition, Gawlik-Dziki et al. (2013) suggested that addition of 2–3% onion skin in bread results in a significant improvement of its antioxidant potential.
 Click to view | Table 20. Phenolic composition of onion |
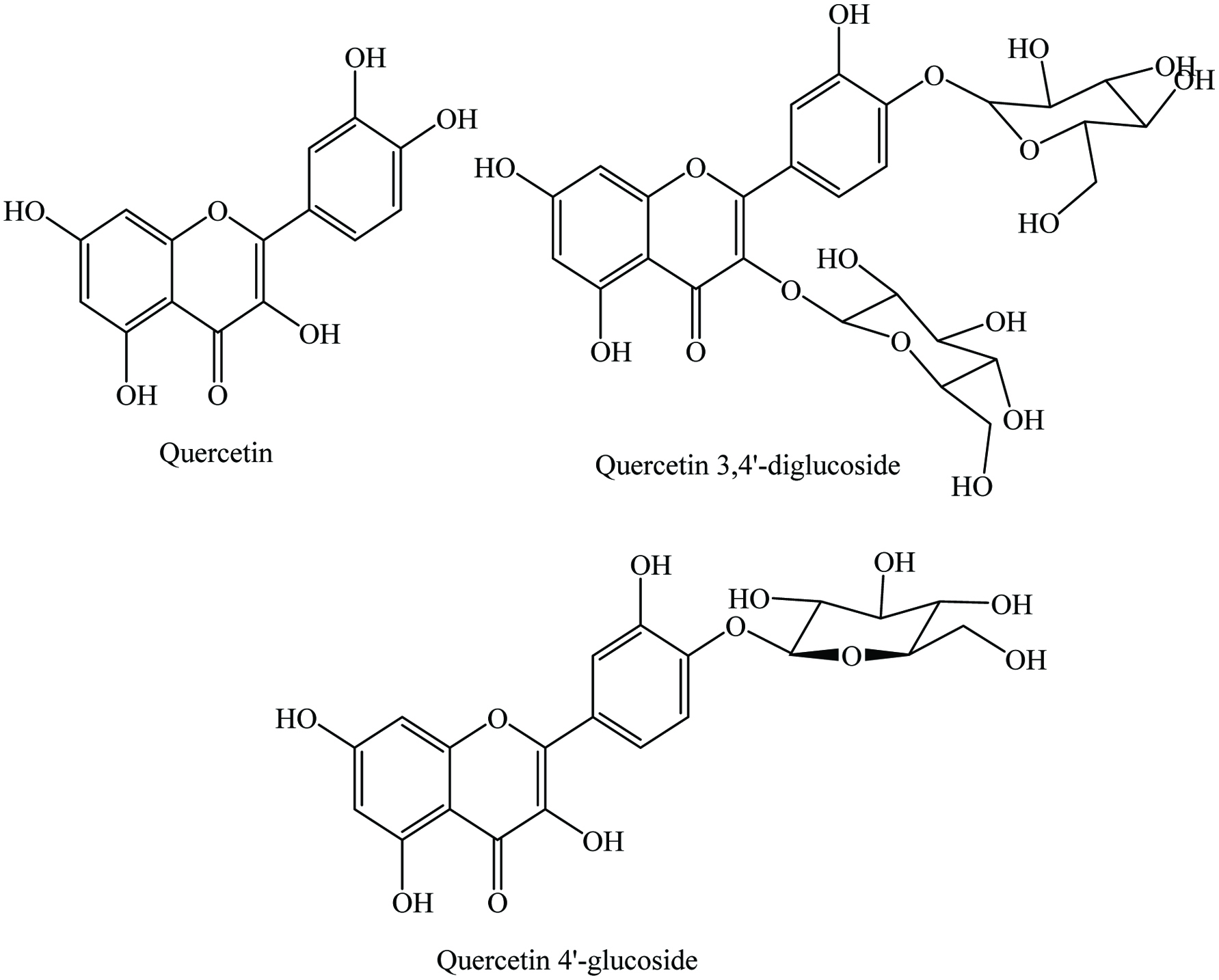 Click for large image | Figure 20. Major flavonoids of onion. |
2.21. Rosemary
Rosemary (Rosmarinus officinalis L) is one of the most common spices, broadly used in food processing. It belongs to the Lamiaceae family and is known as Rosmarinus coronarium. It is native to the Mediterranean region and is also widely cultivated in Spain, Portugal, France, Italy, Algeria, Morocco, Russia, Yugoslavia, China, Tunisia, and the USA. It is a perennial shrub and can grow up to 7 ft with bright green narrow leaves and pale purple or bluish flowers. Leaves (fresh or dried) and essential oil are the main products.
Rosemary contains 9.31% moisture, 4.88% protein, 15.22% lipid, 64.06% total carbohydrate, 42.06% total dietary fiber, ash, and vitamins. The active components include essential oil up to 2.5% which are mainly 1,8-cineole, camphor, and α-pinene. Numerous phytochemicals have been identified in rosemary including alkaloids, terpenoids, saponins, tannins, flavonoids, quinines, and steroids (Soundararajan et al. 2017). The major constituents of rosemay are rosmarinic acid, carnosol, carnosic acid, vanillic acid, p-coumaric acid, caffeic acid, ursolic acid, genkwanin, scutellarein, homoplantaginin, cirsimaritin, 12-O-methylcarnosic, rosmanol, rosmariquinone, rosmadial, caffeoyl derivatives, epirosmanol, cineole, α-pinene, geraniol, limonene, β-carotene, naringin, apigenin, luteolin, and rutin (Table 21) (Almela et al. 2006; Borras-Linares et al. 2014; Borrás Linares et al. 2011; Cuvelier et al. 1996; Embuscado 2015; Kaefer and Milner 2008; Kontogianni et al. 2013; Leja and Czaczyk 2016; Shahidi and Ambigaipalan 2015; Suhaj 2006; Vallverdú-Queralt et al. 2014; Yashin et al. 2017). Chemical structures of some phenolic compounds found in rosemary are shown in Figure 21. Recently, Mena et al. (2016) isolated 57 components from the rosemary extract; 24 flavonoids (mainly flavones, flavonols, and flavanones), 24 diterpenoids (particularly carnosol, carnosic acid, and rosmanol derivatives), five phenolic acids (mainly carnosic acid), three lignans (mostly medioresinol derivatives), one triterpenoid (betulinic acid). They also identified 63 volatile compounds; mainly terpenes, aldehydes, alcohols, esters, and ketones. Moreover, Rašković et al. (2014) identified 29 chemical components from rosemary essential oil; the major volatile compounds were 1,8-cineole (43.77%), α-pinene (11.51%), and camphor (12.53%). Similarly, Takayama et al. (2016) isolated the volatile constituents of rosemary by GC-MS and the major components were identified as cineole (28.5%), camphor (27.7%), and α-pinene (21.3%). In addition, the major compounds of rosemary leaves were 6″-O-trans-feruloylhomoplantaginin, 6″-O-trans-p-coumaroylnepitrin, 6-methoxyluteolin 7-glucopyranoside, 6″-O-trans-feruloylnepitrin, luteolin 3′-O-(3″-O-acetyl)-β-D-glucuronide, luteolin 3′-O-β-D-glucuronide, kaempferol, genkwanin, luteolin, ladanei, 1-O-(4-hydroxy-benzoyl)-β-D-glucopyranose, 1-O-feruloyl-β-D-glucopyranose, carnosic acid, rosmarinic acid, and carnosol (Bai et al. 2010).
 Click to view | Table 21. Phenolic composition of rosemary |
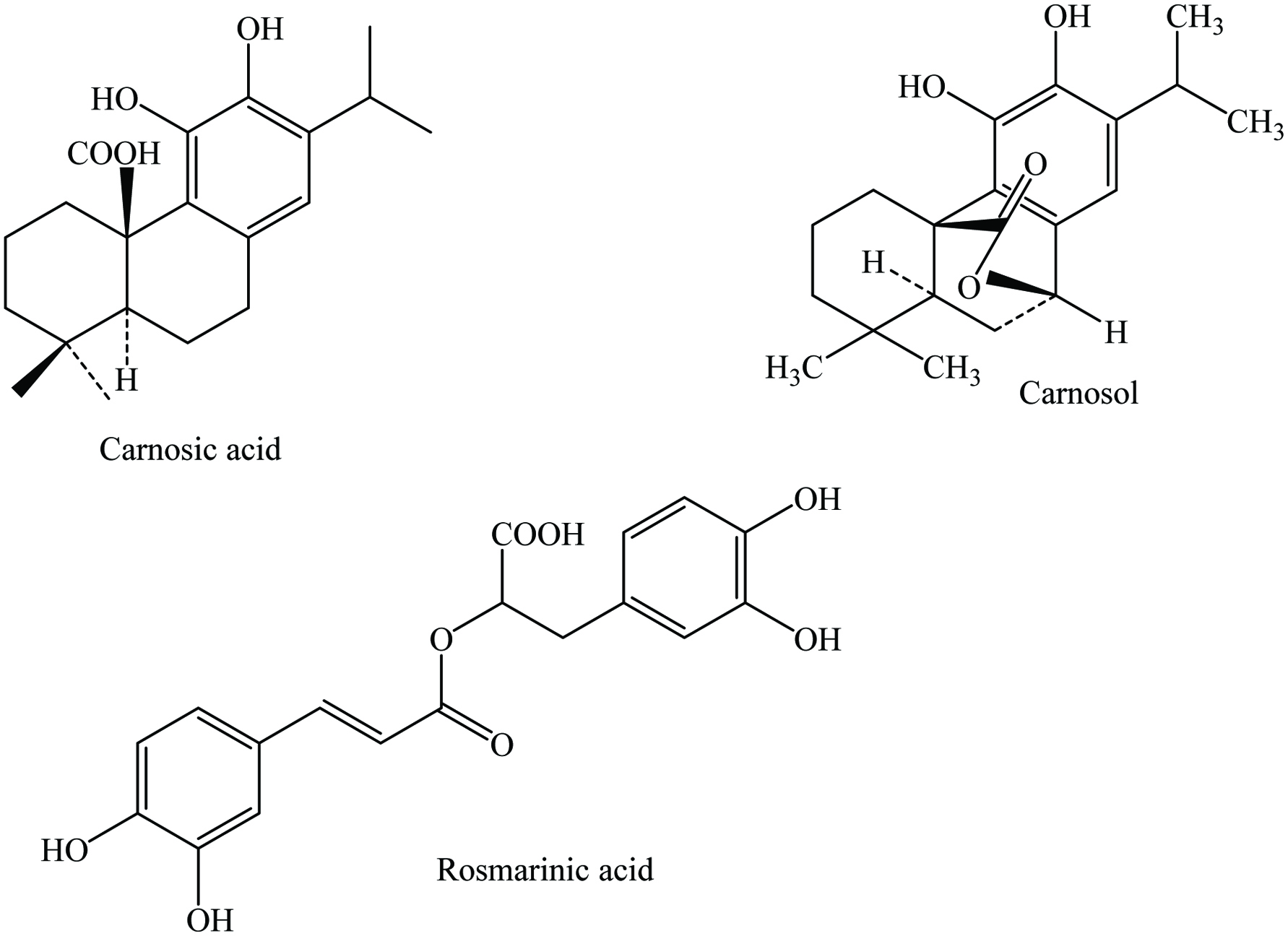 Click for large image | Figure 21. Major phenolic compounds of rosemary. |
2.22. Saffron
Saffron (Crocus sativus L) is the most expensive spice in the world, belonging to Iridaceae family. The most common synonyms are saffron crocus, Spanish saffron, Alicante saffron, true saffron, and autumn crocus. It is cultivated worldwide particularly in Iran which is the very main producing and exporting country as well as China, Japan, India France, Turkey, Spain, Greece, and Morocco. It is a perennial herb up to 1 ft high with gray-green leaves and funnel-shaped reddish-purple or whitish flowers. Dried flower stigmas are the major products.
Saffron contains 8.5–11.90% moisture, 10–12% protein, 5–6% lipid, 65–67% carbohydrate, 1.2% ash, 3–4% fiber, 8–13% fixed oil, and 0.4–1.5% essential oil (Charles 2013). The principal constituents which are responsible for the coloring of saffron are trans- and cis-crocins (water-soluble carotenoids). The major compound of saffron is a carboxaldehyde volatile compound (safranal), which constitutes70% of total volatile components (Himeno and Sano 1987). Numerous phytochemicals have been identified in saffron, mainly cardiac glycosides, tannins, saponins, steroids, flavonoids, anthraquinones, terpenoids, and anthocyanins (Table 22) (Khayyat 2017). Particularly, crocins, safranal, aglycone of picrocrocin, flavonoids, caffeic, ferulic, gallic, cinnamic, gentisic, salicylic, p-hydroxybenzoic, syringic, and vanillic acids are the abundant constituents in saffron (Abdi et al. 2011; Karimi et al. 2010; Khayyat 2017; Yashin et al. 2017). Chemical structures of most abundant compounds found in saffron are shown in Figure 22. Recently, Mounira et al. (2015) identified 57 volatile constituents from 19 different Moroccan saffrons and reported that the chemical classes were monoterpene hydrocarbons (6–42%), oxygenated monoterpenes (3–29%), and non-terpene components. They reported that the major constituents in all accessions were safranal, 1,8-cineole, isophorone, 4-keto-isophorone, and α-pinene. Moreover, Caballero-Ortega et al. (2007) collected saffron samples from 11 different countries and Sigma Chemical Company; the highest concentrations of water-soluble glycosidic carotenoids were found in Greek, Indian, New Zealand, and Spanish saffron extracts. In addition, the different flower tissues and corms of saffron are the main source of bioactive compounds with pharmacological properties. The highest concentration of phenolics was observed in waking corms compared to the dormant stage (Abdi et al. 2011).
 Click to view | Table 22. Phenolic composition of saffron |
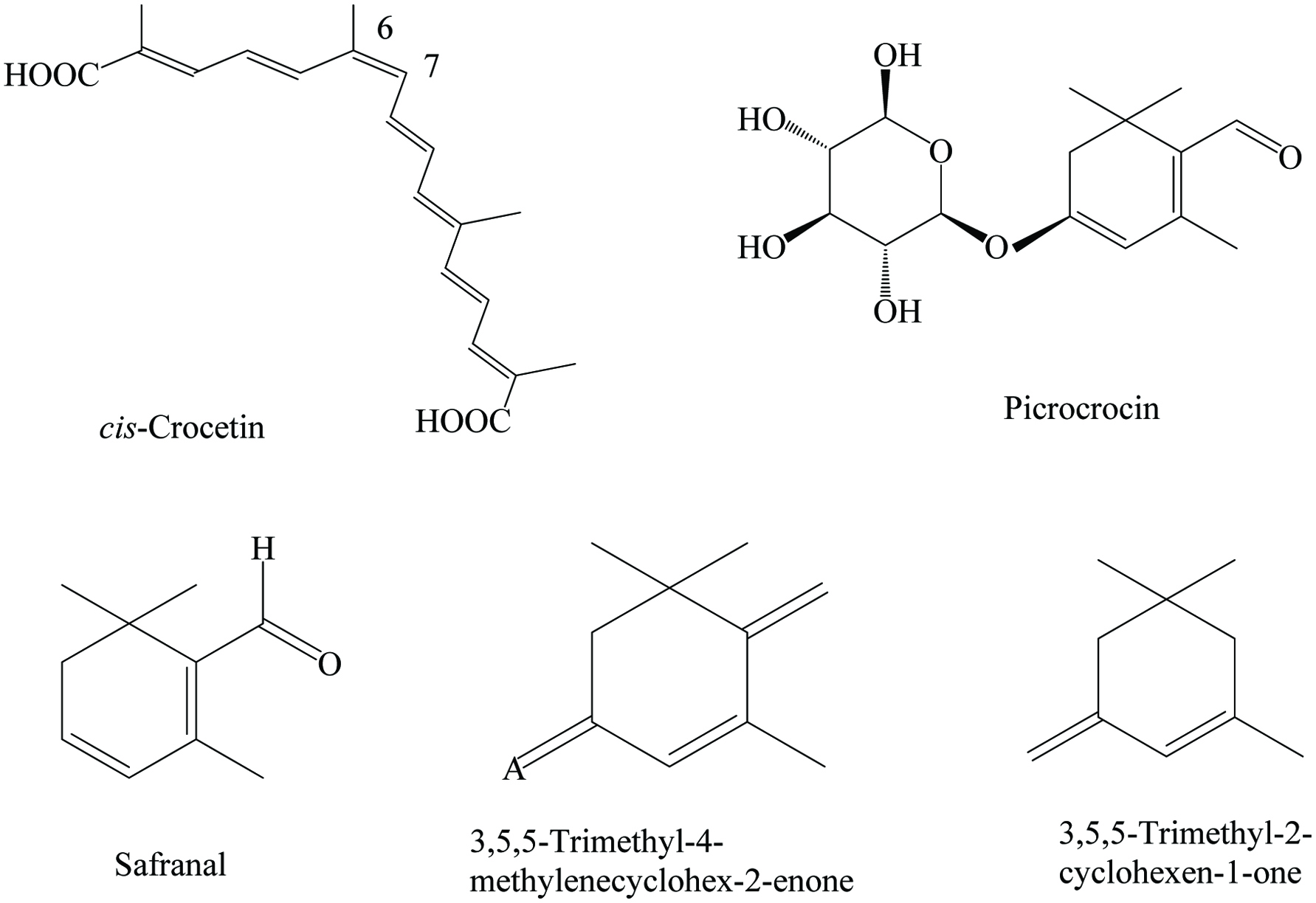 Click for large image | Figure 22. Major phytochemicals of saffron. |
2.23. Tamarind
Tamarind (Tamarindus indica L) belongs to the family of Leguminosae and is commonly known as tamarind tree. This evergreen tree is planted widely in Bangladesh, India, Sri Lanka, Myanmar, Malaysia, Thailand and several Australian, African, and American countries. The height of this tree around 80 ft with dark grey bark and yellow flowers. The most important part is the pod which contains many seeds. Fruit, kernel (powder) and pulp are the main products (Peter 2001).
The dried fruit contains 15–30% moisture, 2–8.79% protein, 0.50–2.53% lipid, 56.70–70.70% carbohydrates, 2.10–2.90% ash, and 2.20–18.30% fiber, whereas the seed contains 17.1–20.1% protein, 6.0–8.5% lipid, 65.1–72.2% carbohydrates, 2.3–3.2% ash, and 0.7–4.3% fiber (Parthasarathy et al. 2008; Peter 2001). Both pulp and seed are the rich source of mineral mainly magnesium, manganese, calcium, phosphorus, sodium, and iron (Gomathinayagam et al. 2017; Khanzada et al. 2008). A number of phytochemicals have been identified in tamarinds such as alkaloids, flavonoids, tannins, triterpenoids, saponins, and phytosterols (Table 23) (Gomathinayagam et al. 2017; Yusha’u et al. 2014). Chemical structures of most abundant compounds found in tamarind are shown in Figure 23. The major volatile constituents of tamarind are linalool anthranilate, phenyl acetaldehyde, benzyl benzoate, furfural, limonene, α-pinene, β-pinene, and nerol. The polyphenolics profile of tamarind pericarp and seed is dominated by (+)-catechin, procyanidin B2, (-)-epicatechin, procyanidin pentamer, procyanidin trimer, procyanidin tetramer, procyanidin hexamer, taxifolin, luteolin, apigenin, eriodictyol, and naringenin (Sudjaroen et al. 2005). Dry sample of tamarind extracted with methanol had the highest concentration of total phenolics (32.96 ± 3.08 g/100 g) and tannins (26.34 ± 2.20 g/100 g) compared to the raw sample (Siddhuraju 2007). Furthermore, Natukunda et al. (2016) incorporated tamarind seed powder (0–10%) into mango juice and cookies and found an increase of total content of phenolics, flavonoids, and condensed tannins, as well as total antioxidant activity which were around 13, 5, 90, and 5 times, compared to the control sample, respectively. Moreover, Tsuda et al. (1994) isolated 2-hydroxy-3′,4′-dihydroxyacetophenone, methyl 3,4-dihydroxybenzoate, 3,4-dihydroxyphenyl acetate, and (-)-epicatechin from tamarind seed extracts and suggested that the tamarind seed coat could be used as a safe and low-cost source of antioxidants. In addition, Pino et al. (2004) identified 81 volatile constituents from tamarind; mainly 2-furfural, 2-phenylacetaldehyde, and hexadecanoic acid.
 Click to view | Table 23. Phenolic composition of tamarind (fruit pulp) |
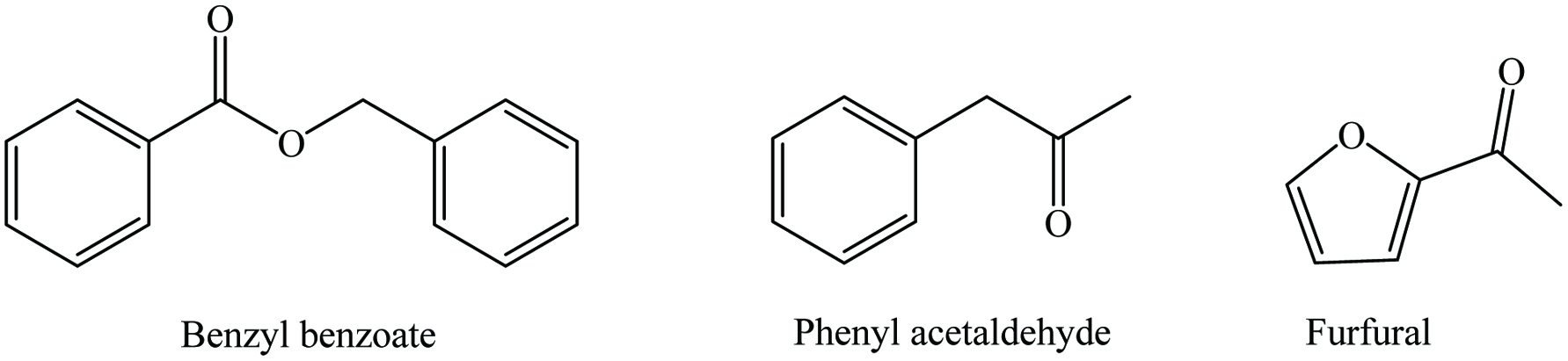 Click for large image | Figure 23. Major phytochemicals of tamarind. |
2.24. Thyme
Thyme (Thymus vulgaris L) belongs to the family of Lamiaceae and is known as Thymus aestivus, Thymus webbianus, Thymus valentianus, Thymus ilerdensis, French thyme, garden thyme, and common thyme. Thyme is native to the European countries and particularly Spain, Italy, France, Yugoslavia, Turkey, Greece, and North America which are also the major thyme exporting countries. It is a perennial shrub up to 2 ft high with gray-green oval leaves and pale purple hairy flowers. Herb (dried), leaves (dried), essential oil, and oleoresin are the major products.
The nutrient composition of dried thyme is 7.79% moisture, 9.11% protein, 7.43% lipid, 63.94% total carbohydrate, 37.0% fiber, ash, and vitamins. Various phytochemicals such as thymol, carvacrol, α-pinene, γ-terpinene, eugenol, limonene, cineole, p-cumene-2,3-diol, ascorbic acid, β-carotene, ursolic acid, caffeic acid, rosmarinic acid, gallic acid, carnosic acid, flavonoids, hispidulin, cismaritin, apigenin, luteolin, and naringenin have been reported in thyme (Table 24) (Aguilar and Hernández-Brenes 2015; Embuscado 2015; Jarić et al. 2015; Kaefer and Milner 2008; Lee et al. 2005; Leja and Czaczyk 2016; Miura and Nakatani 1989; Suhaj 2006; Vallverdú-Queralt et al. 2014; Venskutonis et al. 1996; Yashin et al. 2017). Moreover, Pirbalouti et al. (2013) identified 24 phytochemicals from wild and cultivated thyme; mainly thymol (35.5–44.4%), γ-terpinene (10.5–11.9%), carvacrol (4.4–16.1%), and p-cymene (8.5–16.1%) (Figure 24). They reported that the compositions of essential oil are affected by agronomic management practices as well as environmental conditions. Recently, Satyal et al. (2016) analyzed the essential oil composition of thyme from various geographical locations and suggested that the enantiomeric distribution may also have intense effect on its bioactivity, aroma, and flavor profile. They reported that samples collected from France (Nyons), France (Richerenches), Serbia (Jablanicki), Serbia (Pomoravje) were dominated by linalool chemotype, thymol chemotype, geraniol chemotype, and sabinene hydrate chemotype, respectively. Similarly, 42 and 46 essential oil components were also identified in Iranian thyme (mainly thymol, γ-terpinene, p-cymene, carvacrol, and terpinolene) and British thyme (mostly thymol, p-cymene, γ-terpinene, carvacrol, linalool, and trans-caryophyllene), respectively (Alizadeh 2013). In addition, the highest phenolics and radical scavenging activity were found in the Iranian thyme compared to its British counterpart. Furthermore, the total phenolic and flavonoid contents of Turkish thyme were 256.0 μg GAE/mg and 44.2 μg QE/mg dried extract, respectively (Köksal et al. 2017). However, Hudaib et al. (2002) analyzed the oil composition and variations during the vegetative cycle (young-2 years and old-5 years); the young plant harvested in June/July showed the best oil yield and the highest percentage of the monoterpene phenols. In addition, Wang et al. (1999) isolated four acetophenone glycosides from thyme extracts; namely 4-hydroxyacetophenone 4-O-[5-O-(4-hydroxybenzoyl)-β-D-apiofuranosyl]-(1→2)-β-D-glucopyranoside, 4-hydroxyacetophenone 4-O-[5-O-(3,5-dimethoxy-4-hydroxybenzoyl)-β-D-apiofuranosyl]-(1→2)- β-D-glucopyranoside, picein, and androsin.
 Click to view | Table 24. Phenolic composition of thyme |
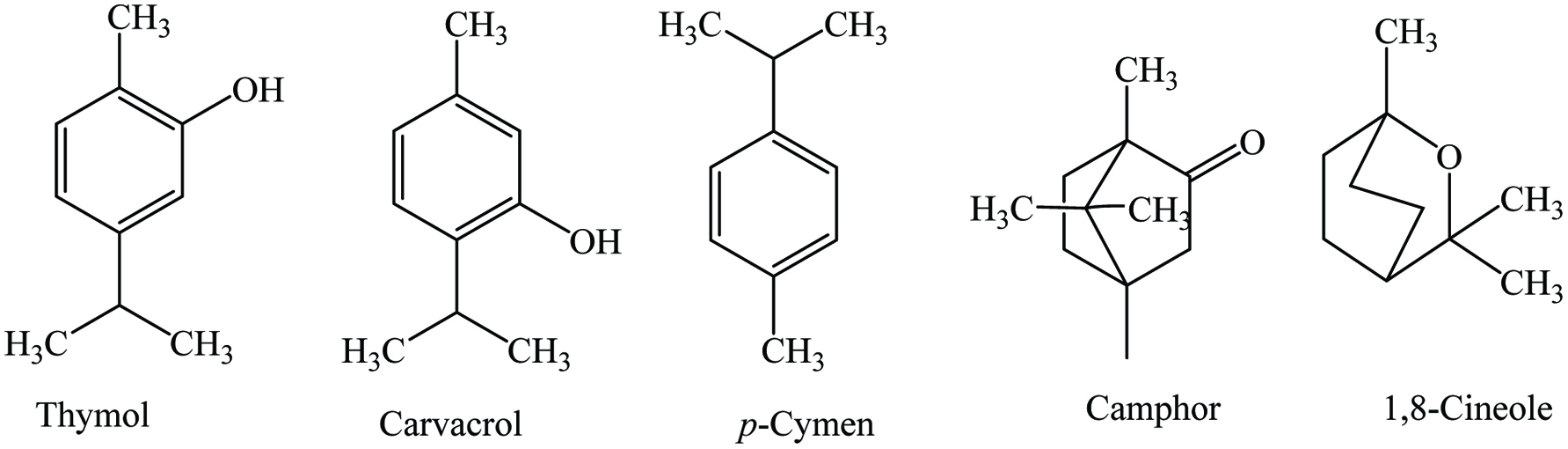 Click for large image | Figure 24. Major phytochemicals of thyme. |
2.25. Turmeric
Turmeric (Curcuma longa L) is in the family of Zingiberaceae and the most common synonyms for it are Curcuma domestica Vale, Curcuma domestica Loir, Amomum curcuma Jacq, curcuma, yellow root, Indian saffron, and yellow ginger. It is native to India, Bangladesh, and Sri Lanka and is also cultivated in Pakistan, Nepal, Thailand, Cambodia, China, Japan, Taiwan, Indonesia, Malaysia, Madagascar, Philippines, Peru, Jamaica, and Haiti. It is a tropical annual or perennial that its roots can grow up to 3 ft with dark green distichous leaves and yellow flowers. The underground rhizome is the main product which is used as fresh or dry, while essential oil and oleoresin are also used (Nisar et al. 2015).
The general composition of turmeric is 11–13% moisture, 6–9% protein, 5–10% lipid, 60–70% total carbohydrate, 3–7% ash, and 2–7% fiber (Charles 2013; Parthasarathy et al. 2008; Peter 2001). Turmeric oleoresin is orange-red in color; mainly volatile oil, non-volatile and other active ingredients which are used in the food industries for color and aroma. The yield of oleoresin is around 7–15% which is obtained by solvent extraction, mainly acetone. Turmeric contains 1.5–5% volatile oil, mainly 60% turmerone and 25% zingiberene. It contains curcuminoids (2.5–8%) which is responsible for the yellow color. Particularly, three curcuminoids, namely curcumin (diferuloylmethane) (71.50–94%), demethoxycurcumin (6–19.4%), and bisdemethoxycurcumin (0.30–9.10%) are responsible for the yellow color (Kumar and Sakhya 2013; Sabale et al. 2013; Schieffer 2002). Curcumin is a hydrophobic phenolic compound and its content depends on the extraction method employed. Sahne et el. (2016) demonstrated that the highest yield of curcumin was obtained by Soxhlet method (6.9%) followed by enzyme-assisted (4.1%), ultrasound-assisted (3.92%), and microwave-assisted (3.72%) extraction. To date, more than 250 phytochemicals have been identified in turmeric; primarily phenolic compounds and terpenoids. They are mainly curcumins, curcuminoids, turmerone, α-turmerone, β-sesquiphellandrene, eugenol, γ-terpinene, camphene, carotene, ascorbic acid, caffeic, protocatechuic, syringic, p-coumaric, ferulic, and vanillic acid (Table 25) (Jiang et al. 2006; Kimthet et al. 2017; Leja and Czaczyk 2016; Li et al. 2011; Nisar et al. 2015; Sahne et al. 2017; Shahidi and Ambigaipalan 2015; Suhaj 2006; Toda et al. 1985; Yashin et al. 2017). Chemical structures of most abundant compounds found in turmeric are shown in Figure 25. In addition, the contents of curcuminoids and essential oils in turmeric vary with varieties, geographical locations, sources, cultivation conditions, extraction methods, and storage conditions (Li et al. 2011; Pawar 2014; Sandeep et al. 2016). Turmeric oil was examined by GC and GC-MS; α-turmerone, turmerone, and curlone were the main components along with other oxygenated constituents (Negi et al. 1999). Moreover, Singh et al. (2010) compared the chemical constituents of fresh and dry rhizomes, the principal components in fresh rhizome were α-turmerone (20.5%) and β-turmerone (11.1%), whereas the dry rhizome oil was dominated by α-turmerone (21.4%), α-curcumene (6.6%), and α-santalene (7.2%). Besides, in oleoresins, α-turmerone (53.4%) and β-turmerone (18.1%) were abundant in the fresh rhizome, while dry rhizome was dominated by α-turmerone (9.6%) and α-santalene (7.8%). However, Tanvir et al. (2017) collected turmeric from different locations of Bangladesh; Chittagong’s turmeric contained the highest concentrations of phenolics (16.07 ± 0.30 g GAE/100 g), flavonoids (9.66 ± 0.04 g CE/100 g), and tannins (16.38 ± 0.07 g TE/100 g) compared to the other places. In addition, turmeric flower is also a rich source of curcumin (3.87 ± 0.5 µg/g) and phenolics (210.45 ± 1.32 mg GAE/100 g) ( Kumar et al. 2016).
 Click to view | Table 25. Phenolic composition of turmeric |
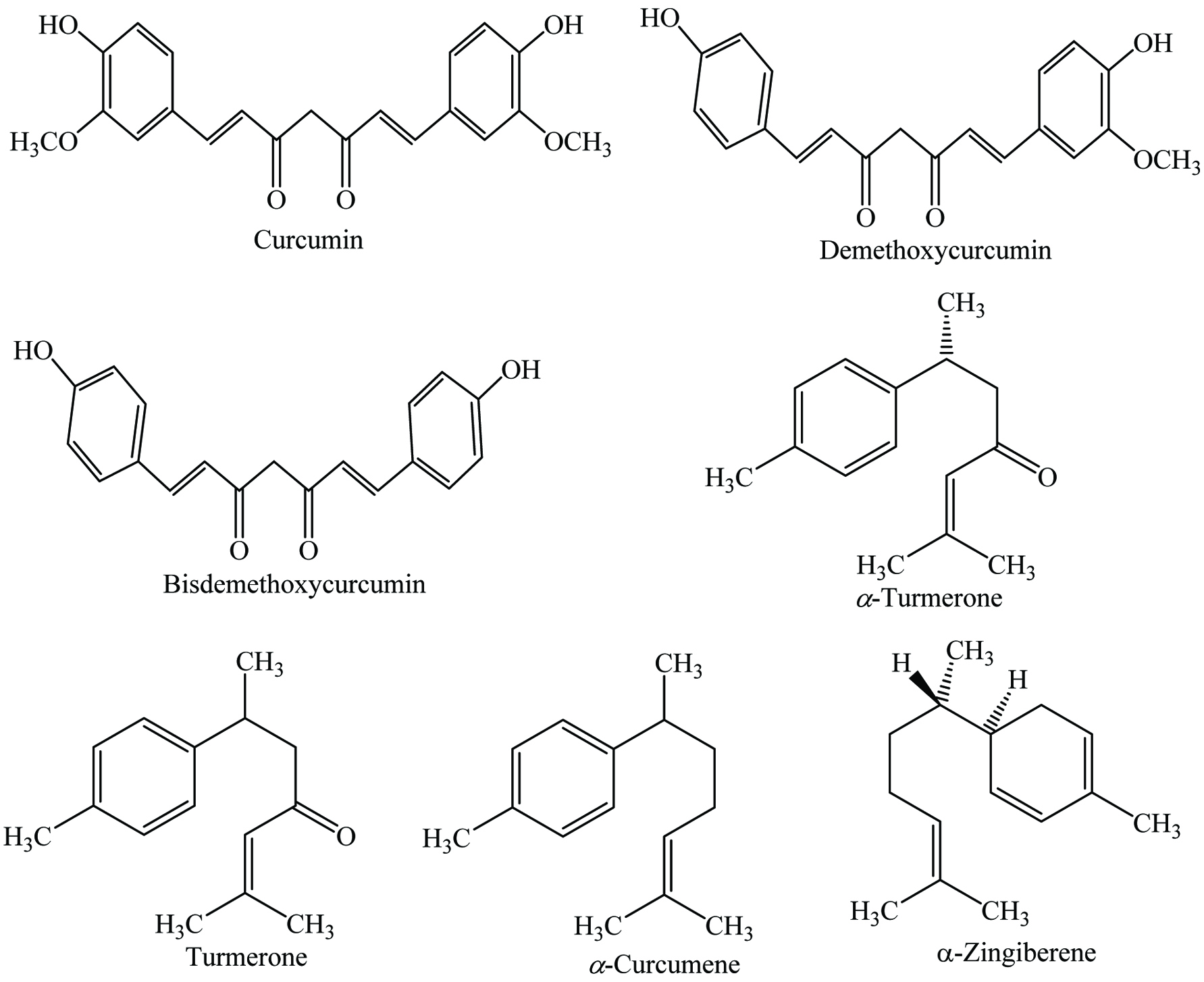 Click for large image | Figure 25. Major chemical constituents of turmeric. |
| 3. Antioxidant efficacy of spices | ▴Top |
Spices are rich sources of antioxidant phenolic and polyphenolic compounds. Due to the antioxidant activity of spices, they have potential health benefits by inhibiting lipid peroxidation in food and oxidative stress in the body. A summary of the antioxidant activity of common spices is given in Table 26. The antioxidant properties of different spices vary extensively based on the assay type. Every assay has a different mechanism of action and hence may exhibit varied antioxidant property trends among spices. Moreover, the antioxidant properties of spices are influenced by variety, geographical location, fertilization, weather condition, storage condition, extraction procedure, and calculation method, among others. Therefore, it may not be wise to rank spices based on their antioxidant activity due to various units used for even the same assay. Various methods have been used to determine the antioxidant properties of spices. These include DPPH (2,2-diphenyl-1-picrylhydrazyl) radical scavenging activity, ABTS (2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) method), FRAP (ferric reducing antioxidant power), ORAC (oxygen radical absorbance capacity), and reducing potential. For example, Hossain et al. (2008) compared the antioxidant activity of 30 common spices with five popular synthetic antioxidants; rosmarinic acid from Lamiaceae and eugenol from clove demonstrated a higher antioxidant activity compared to synthetic antioxidants. They also reported that kaempferol from Apiaceae spice, curcumin from turmeric, capsaicin from chili, gingerol from ginger, and thymol from thyme were also comparable to the synthetic antioxidants. In addition, Wojdyło et al. (2007) determined the total equivalent antioxidant capacities (TEAC) values of the 32 selected spices; these ranged from 1.76 to 346 µM trolox/100 g dw, which showed a positive correlation between TEAC values and total phenolic content.
 Click to view | Table 26. Reported antioxidant activity in common spices |
| 4. Antimicrobial activity of spices | ▴Top |
Generally, spices contain around 10% moisture which is considered as a quality indicator. The high moisture content is an indicator of its freshness, whereas low moisture content ensures longer shelf life due to unfavorable conditions for the growth of microorganism. Due to the presence of a wide range of phytochemical components, spices possess antimicrobial activity against bacteria, yeast, molds, and viruses. Phytochemicals such as phenols, alcohols, aldehydes, ketones, hydrocarbons, and ethers play an important role in extending the storage time and stability of the food. Particularly, flavonoids such as kaempferol, as well as tannins, and volatile oils play an important role in inhibiting microbial growth (Hossain et al. 2017). For example, essential oils containing mainly polyphenols, terpenes, monoterpenes, and sesquiterpenes are responsible for the antimicrobial activity of spices. The essential oils bring about structural changes and cause functional damage to the bacterial cell membrane. It has been reported that the mechanisms of antimicrobial activity are influenced by different pathways, such as disrupting enzyme systems, affecting the phospholipid bilayer of the cell membrane, compromising the genetic component of bacteria, and developing hydroperoxidase by oxygenation of unsaturated fatty acids (Arques et al. 2008; Lanciotti et al. 2004; Proestos et al. 2008; Skocibusic et al. 2006). In this context, natural extracts from spices have been incorporated into packaging materials in order to achieve antioxidant and antimicrobial activities. For example, tamarind seed starch was found to render antioxidant and antibacterial properties, which can be a good supplement matrix for the preparation of edible films (Mohan et al. 2016). A summary of the antimicrobial activity of common spices is given in Table 27.
 Click to view | Table 27. Antimicrobial properties of spice against test microorganism |
Ajowan essential oil showed a 100% inhibition against three Aspergillus species; thymol and eugenol were the main providers to the antimicrobial activates (Kim et al. 2016). Moreover, ajwain oil exhibited a dose-dependent antimicrobial activity against Streptococcus mutans and Enterococcus faecalis (Ganapathi and Roy 2017). However, anise extracts demonstrated antimycotic activity against Candida parapsilosis, C. albicans, C. tropicalis, C. krusei, and C. pseudotropicalis with MIC (minimum inhibitory concentrations) values between 17 and 20% (v/v) (Kosalec et al. 2005). They also reported that the essential oil of anise exhibited strong antifungal activity against dermatophytes with MIC of less than 0.78% (v/v) and yeasts with MIC of lower than 1.56% (v/v). In addition, methanol extracts of aniseed were effective against Helicobacter pylori at MIC of 100 µg/mL (Mahady et al. 2005). Beatovic et al. (2015) reported that basil essential oil was most effective against Micrococcus flavus bacterium, with MIC value of 0.009 µg/mL. The same study described that the essential oils exhibited a 10- to 100-fold higher ability to prevent fungal growth than the commercial antifungal agents. Moreover, the essential oil of basil completely inhibited the growth of 22 mycelial species of fungi at a dose of 1.5 mL/L, including Aspergillus flavus and Aspergillus parasiticus (Dube et al. 1989). However, Cherrat et al. (2014) found that the combination of bay leaf essential oils (0.2 µL/mL) with mild heat (54 °C for 10 min) and high hydrostatic pressure (175–400 MPa for 20 min) had a higher inactivation of foodborne pathogens. Furthermore, essential oils of bay leaf showed a very effective antibacterial activity against Staphylococcus aureus, Staphylococcus intermedius, and Klebsiella pneumonia with MIC ranging from 0.01 to 1 mg/mL (Derwich et al. 2009). On the other hand, Al-Shahwany (2014) reported that the alkaloids from Piper nigrum (fruits) showed a maximum and minimum zone of inhibition against Gram-negative bacteria Escherichia coli and Gram-positive bacteria Staphylococcus aureus, respectively. While the opposite scenario was seen in phenols. Similarly, black pepper chloroform extract (BPCE) showed a strong inhibitory effect on Escherichia coli and Staphylococcus aureus (Zou et al. 2015). The same study described that the antibacterial mechanism of BPCE was due to the destruction of the bacterial cell membrane by inhibition of tricarboxylic acid pathway and increased the pyruvic acid level in bacterial solutions as well as decreased ATP level in bacterial cells. Additionally, BPCE destroyed the cell membrane permeability, which consequently inhibited energy synthesis, caused metabolic dysfunction, and triggered cell death. Recently, Chen et al. (2018) stated that the BPCE destroyed the bacterial cell membranes and walls, which was followed by a breakdown of bacterial cell respiration.
The MIC values of cinnamon oil against food-borne pathogens (Escherichia coli, Candida albicans, Bacillus cereus, and Salmonella typhimurium) were determined as 0.5 μL/disc (Vazirian et al. 2015). Moreover, Naveed et al. (2013) compared the MIC values of cumin, cinnamon, cardamom, and clove; cinnamon bark oil showed the best antibacterial activities against pathogenic strains (mainly Salmonella typhi and Pseudomonas fluorescens) with 2.9 mg/mL concentration. Furthermore, Krishnan et al. (2014) analyzed the antimicrobial and antioxidant activity of spice extracts (clove, cinnamon, oregano, and mustard) in raw chicken meat during storage of 15 days. Results showed that spice extracts were very effective against microbial loads as well as lipid oxidation. In addition, the antibacterial mechanism of Cinnamomum cassia bark against Staphylococcus aureus and Escherichia coli may be due to the increase of cell membranes permeability as well as the leakage of intracellular constituents (Huang et al. 2014). Ranasinghe et al. (2002) suggested that the clove and cinnamon essential oil (0.03–0.11%, v/v) could be used as a natural antifungal agent to prevent post-harvest fungal diseases of banana. However, due to the presence of alcohols (2-trans-decenol, 1-decanol, hexenol, and 2-cis-dodecenol), essential oil of coriander showed a strong antifungal activity against Candida yeast (Begnami et al. 2010). In addition, Darughe et al. (2012) suggested that the essential oil of coriander could be used as a natural antifungal and an antioxidant in foodstuffs, particularly those containing a high amount of lipid. Recently, Saee et al. (2016) stated that the cumin essential oil can have a significant influence on Gram-negative bacteria which are responsible for urinary tract infection compared to the Gram-positive bacteria. Moreover, the combination of nisin and cumin essential oil can inhibit the growth of Staphylococcus aureus and Salmonella typhimurium bacteria, which can be used as an alternative for chemical food preservatives (Tavakoli et al. 2015).
On the other hand, Ksouri et al. (2015) tested the antimicrobial activity of wild dill extracts against four Gram-negative bacteria, two Gram-positive bacteria, and one yeast, the range of MIC value was 0.19 to 12.5 mg/mL. Recently, Bano et al. (2016) evaluated the antimicrobial activity of fennel extracts against Escherichia coli, Staphylococcus aureus, Bacillus pumilus, Listeria monocytogenes, and Enteropathogenic E. coli. The maximum antimicrobial activity was found against S. aureus at 20 mm inhibition zone. Moreover, fennel essential oil showed a strong antibacterial activity against food-borne pathogens such as Staphylococcus albus, Salmonella typhimurium, Shigella dysenteriae, Bacillus subtilis, and Escherichia coli (Diao et al. 2014). They concluded that the antimicrobial mechanisms of fennel essential oil against S. dysenteriae could be due to the leakage of electrolytes and losses of nutrients mainly reducing sugars and proteins. However, due to the presence of phytochemicals such as tannins and flavonoids, fenugreek seed extracts (aqueous and methanolic) showed a strong anti-bacterial activity against E. coli (Chalghoumi et al. 2016). On the other hand, the major organosulfur compound of garlic is allicin which is considered as a strong antimicrobial agent against bacteria (Pseudomonas, Streptococcus, and Staphylococcus), fungi (Candida albican), and parasite (Entamoeba histolytica and Giardia lamblia) (Ankri and Mirelman 1999; Reiter et al. 2017). Allicin can react with thiol groups of various enzymes such as alcohol dehydrogenase, RNA polymerase, and thioredoxin reductase, which can influence the metabolism of cysteine proteinase action involved in the virulence of parasites (Ankri and Mirelman 1999). In addition, Park et al. (2008) concluded that the highly alkylated gingerols (10-gingerol and 12-gingerol) obtained from ginger strongly inhibited the growth of oral pathogens (Porphyromonas gingivalis, Porphyromonas endodontalis, and Prevotella intermedia) at a MIC range of 6–30 μg/mL.
Recently, Walker et al. (2016) suggested that the marjoram essential oil has a strong antimicrobial activity against Salmonella enterica serovar Schwarzengrund and it can be used as a natural disinfectant during vegetable processing instead of sodium hypochlorite. However, fungi such as Alternaria alternata, Magnaporthe grisea, Colletotrichum coccodes, C. gloeosporioides, Acidovorax konjaci, Agrobacterium tumefaciens, and Burkholderia glumae were relatively sensitive to the three lignans (meso-dihydroguaiaretic acid, erythro-austrobailignan-6, and nectandrin-B) of nutmeg seed methanol extracts (Cho et al. 2007). Moreover, Valente et al. (2011) stated that the crude essential oil (0.1%) of the Brazilian nutmeg inhibited the radial growth of Aspergillus glaucus (60%), A. niger (71%), Fusarium oxysporum (75%), F. semitectum (78%), Colletotrichum musa (97%), and C. gloeosporoides (98%). On the other hand, Hannan et al. (2010) analyzed the antimicrobial activity of onion (purple and yellow) against 33 clinical isolates of Vibrio cholera and found that all strains of Vibrio cholera were sensitive to onion. The MIC range was 19.2–21.6 and 66–68.4 mg/mL for the purple and yellow type onion, respectively. Moreover, the major compound of onion such as quercetin and kaempferol showed a strong inhibitory effect against Gram-positive bacteria (Bacillus cereus, Microcroccus luteus, Staphylococcus aureus, and Listeria monocytogenes) compared to the Gram-negative bacteria (Pseudomonas aeruginosa and Escherichia coli) (Santas et al. 2010). However, the antimicrobial activity of rosemary extracts is related to their specific phenolic composition mainly carnosic acid and rosmarinic acid. Moreno et al. (2006) demonstrated that the methanol extracts of rosemary containing 5% of rosmarinic acid, 16% carnosol, and 30% of carnosic acid were the most sensitive against Gram-positive bacteria (MIC, 2 to15 mg/mL), Gram negative bacteria (MIC, 2 to 60 mg/mL), as well as yeast (MIC, 4 mg/mL). Similarly, Collins and Charles (1987) reported that the carnosol at the concentration of 50 µg/mL can decrease the bacterial and yeast growth significantly, while it can be inhibited microbial load completely at 150 µg/L. Moreover, the major constituents of rosemary oil such as cineole, borneol, camphor, and α-pinene had a strong antibacterial activity against Listeria monocytogenes strains (Pandit and Shelef 1994). In another study, the principal components of rosemary (borneol, camphor, verbenone, α-pinene, and 1,8-cineole) exhibited antimicrobial activity against Gram-positive bacteria (Bacillus subtilis and Staphylococcus aureus), Gram-negative bacteria (Pseudomonas aeruginosa and E. coli), fungus (Aspergillus niger), and yeast (Candida albicans) (Santoyo et al. 2005). The same study reported that borneol was the most sensitive followed by camphor and verbenone.
On the other hand, whole Pacific white shrimp treated with 1% of saffron tepal extracts (water, ethanol, and methanol) had the lowest microbial growth, lipid oxidation, and melanosis and also superior sensory properties during storage (Abbasvali et al. 2016). However, Escalona-Arranz et al. (2010) investigated the antimicrobial activity of tamarind leaves against Bacillus subtilis, Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Enterococcus faecalis, Salmonella typhimurium, and Candida albicans and reported that the phenols as effective compounds against Bacillus subtilis cultures among others. In addition, due to the inhibitory activity of tamarind seed coat against Staphylococcus aureus and Escherichia coli, it can be used as a natural mordant for dyeing cotton, silk, and wool fabrics with natural dyes (Prabhu and Teli 2014). However, Fani and Kohanteb (2017) tested the antimicrobial activity of thyme oil against 30 clinical isolates, Streptococcus mutans and Streptococcus pyogenes were the most sensitive isolates with a MIC of 3.6 and 1.9 mg/mL, accordingly. Besides, Klarić et al. (2007) reported that the thymol showed nearly three-times higher inhibition compared to the essential oil of thyme and suggested that thymol and essential oils could be used for the disinfection of moldy walls. On the other hand, ethyl acetate extracts of turmeric exhibited a stronger antibacterial activity compared to the water or methanol extracts against methicillin-resistant Staphylococcus aureus (Kim et al. 2005). In addition, curcuminoids and essential oils of turmeric exhibited inhibitory activity against Bacillus subtilis, Bacillus licheniformis, Bacillus macerans, Azotobacter, Candida tropicalis, Candida albicans, Trichophyton rubrum, and Trichophyton mentagrophytes (Apisariyakul et al. 1995; Naz et al. 2010).
| 5. Beneficial effects of spices in health promotion | ▴Top |
Traditionally, spices are used for curing cold, cough, intermittent fever, dyspnea, diseases of the throat, wounds, colic, vomiting, indigestion, nosebleed, allergies, digestive problems, bronchitis, tuberculosis, worms, dysentery, piles, and so on. However, numerous human intervention and animal studies have been assessed the potential health benefits of spices. So far spices have been studied for their anti-arthritic, antidiabetic, anticancer, anti-inflammatory, antihypertension, cardiovascular, neuroprotective, wound healing, body weight management, and antioxidant properties etc. A summary of biological and medicinal benefits of common spices is given in Table 28.
 Click to view | Table 28. Health promoting properties and mechanisms of actions demonstrated by common spices determined in human and animal studies |
5.1. Anticancer activity
A number of in vitro and in vivo studies have demonstrated the potential cancer-preventing activities of spices. For example, curcumin is a phenolic compound from turmeric has the ability to inhibit various types of cancer, including cervical, oral epithelial, gastric, hepatic, leukemia, ovarian, pancreatic, prostate, and colorectal (Villegas et al. 2008). Curcumin shows anti-cancer properties via various biological pathways including mutagenesis, apoptosis, oncogene expression, cell cycle regulation, tumorigenesis, and metastasis (Wilken et al. 2011). Moreover, Ryan et al. (2013) reported that the oral curcumin (6.0 g/day) decreased the severity of radiation dermatitis in breast cancer patients during radiotherapy. However, ginger shows strong inhibitory activities against breast cancer, colorectal cancer, prostate cancer, and lung cancer; mainly due to the presence of gingerols, paradols and shogaols. For example, shogaols (6-, 8-, and 10-shogaol) had much stronger growth inhibitory effects against HCT-116 human colon cancer and H-1299 human lung cancer cells compared to gingerols (6-, 8-, and 10-gingerol), particularly 6-shogaol had much stronger inhibitory effects on nitric oxide (NO) synthesis and arachidonic acid release than 6-gingerol (Sang et al. 2009). Recently, Akimoto et al. (2015) stated that the ginger extract exhibited potential anticancer activity against pancreatic cancer cells (panc-1 cells) by inducing reactive oxygen species-mediated autosis and warrants. In addition, 6-gingerol enhanced tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) viability reduction of gastric cancer cells by inhibiting TRAIL-induced NF-κB stimulation, whereas 6-shogaol alone minimized viability by damaging microtubule (Ishiguro et al. 2007). However, garlic possesses cancer-preventive potential due to the presence of natural organosulfur compounds (mainly diallyl tetrasulfide, diallyl trisulfide, diallyl disulfide, diallyl sulfide, S-allyl mercaptocysteine, and allicin) and organoselenium compounds (se-methyl-L-selenocysteine and selenomethionine). It has been reported that the garlic compound mainly diallyl disulfide improved the activity of eicosapentaenoic acid (EPA), a breast cancer suppressor while decreased the activity of linoleic acid (Tsubura et al. 2011).
On the other hand, onion, mainly quercetin and fisetin, exhibited potential anticancer properties against human oral cancer, ovarian cancer, and colon cancer. Lai et al. (2013) reported that the quercetin inhibited invasion and migration of SAS oral cancer cells via suppression of NF-κB as well as matrix metalloproteinase-2/-9 signaling ways. Furthermore, the major anticancer inhibiting agents of saffron are crocin and crocetin, which are responsible for inhibiting lung cancer, digestive system cancer, and reproductive system cancer. Particularly, crocin inhibited the growth of N-nitroso-N-methylurea- induced breast cancer in rats via inducing apoptosis and inhibiting cell cycle progression by downregulation of cyclin D1 (Ashrafi et al. 2015). Moreover, piperine is a major alkaloid constituent of black pepper shows potential inhibitory activity against breast cancer, prostate cancer, colorectal cancer, and osteosarcoma. Krishnakumar et al. (2009) suggested that piperine may exert its chemopreventive effect due to it increased the content of lipid peroxidation and minimized the levels of nucleic acid and proteins content. Furthermore, black pepper (oleoresin) displayed impressive anticancer effect against PC-3 and HEP-2 cancer cells (IC50 values 5.57 and 5.31 μg/mL, accordingly) (Morsy and Abd El-Salam 2017). However, capsaicin, the major pungent compound of capsicum demonstrates potential anticancer activity in lung cancer, breast cancer, gastric cancer, prostate cancer, colorectal cancer, pancreatic cancer, and bladder cancer. For example, capsaicin was analyzed against prostate and breast carcinoma cell lines in vitro, results indicated that sweet pepper had a higher anticancer effect against prostate cancer, whereas chili pepper had a higher activity against breast cancer cells (Shaimaa et al. 2016). Moreover, rosmanol, carnosic acid, and carnosol are the potential anticancer agents found in rosemary, which are responsible for inhibiting colorectal cancer, breast cancer, colon cancer, and pancreatic cancer. González-Vallinas et al. (2014a) reported that the carnosic acid-rich rosemary extract had a potential inhibitory effect against colon and pancreatic cancer and the possible mechanism was metabolic-related gene GCNT3 expression. In addition, rosemary extract and carnosic acid improved apoptosis and minimized viability in colon cancer cell lines by inhibiting an HCT116 xenograft tumor formation and expression of Nrf2 in colon cells (Yan et al. 2015).
Bennett et al. (2013) reported that the bay leaf inhibited colorectal cancer cell growth in HCT-116, Caco-2, HT-29, and SW-480 human cancer cell line in vitro. Besides, bay leaf exerted a moderate inhibition of cycloxygenase 2 and 5 lipoxygenase enzymatic activity. On the other hand, Qiblawi et al. (2012) suggested that cardamom acts as a chemopreventive agent against two-stage skin cancer. Interestingly, ethyl acetate extract of coriander root inhibited DNA damage in fibroblasts and prevented MCF-7 breast cancer cell migration induced by H2O2, signifying its possible anticancer effect and inhibition of metastasis (Tang et al. 2013). However, curry leaves can be a potential source of proteasome inhibitors for cancer therapy. Noolu et al. (2013) stated that the hydro-methanolic extract of curry leaves minimized cell viability and changed the growth kinetics in breast cancer cells in a dose-dependent manner. Moreover, fennel essential oil is rich in aldehyde content, which interferes with molecular targets in cells and causes a low cytotoxicity. Sharopov et al. (2017) summarized that the fennel essential oil exhibited potent cytotoxic activity against human cervical cancer, colorectal adenocarcinoma, breast adenocarcinoma, T lymphoblast leukemia, and adriamycin-resistant leukemia cancer cell lines. Furthermore, fenugreek also exerted anticancer effects due to its bioactive constituents such as saponins and flavonoids. Khoja et al. (2011) described that fenugreek extracts effectively killed MCF-7 human immortalized breast cancer cells through induction of apoptosis. However, Abaza et al. (2015) analyzed the growth inhibitory and chemo-sensitization properties of thyme (naringenin and flavanone) in human breast and colorectal cancer. Results suggested that naringenin induced apoptosis and showed potent inhibitory activity against colorectal and breast cancer cells via cell cycle arrest, down-regulation of anti-apoptotic genes, up-regulation of the expression of pro-apoptotic genes, and inhibition of pro-survival signaling pathways.
5.2. Anti-diabetic activity
Bay leaf decreased total cholesterol, LDL cholesterol, serum glucose, and triacylglycerols, and improved the level of HDL cholesterol in humans with type 2 diabetes (Khan et al. 2009). They suggested that consumption of bay leaf (1 to 3 g/d for 30 days) reduced the risk factors of cardiovascular diseases as well as diabetes. However, Jagtap and Patil (2010) reported that cumin was able to reduce oxidative stress, hyperglycemia, and formation of advanced glycated end products, which might help as an adjunct to conventional antidiabetic treatment. On the other hand, the effects of ginger on type 2 diabetes mellitus and metabolic syndrome were studied by Zhu et al. (2018) who summarized that the ginger may be a promising adjuvant therapy for type 2 diabetes mellitus and metabolic syndrome. Moreover, Ojewole (2006) stated that ginger ethanolic extract (800 mg/kg) notably lowered the fasting blood glucose in the STZ-diabetic rat after 1 h. In addition, raw ginger (500 mg/kg) has significant potential as a phytomedicine for the treatment of diabetes via glycolytic enzymes (Abdulrazaq et al. 2012). Besides, ginger exhibited antidiabetic properties by increasing insulin sensitivity, protecting β-cells of pancreatic islets, increasing glucose uptake, lowering oxidative stress, and reducing fat accumulation (Akash et al. 2015). Similarly, ginger aqueous extract (500 mg/kg, for 7 weeks) significantly lowered cholesterol, serum glucose, and triacylglycerol contents in the ginger-treated diabetic rats compared with the control diabetic rat (Al-Amin et al. 2006). However, Shukri et al. (2010) claimed that the clove notably reduced the blood sugar increases and lipid peroxidation in diabetic rats; it also inhibited hyperglycemia-induced oxidative tissue damage. Selvi et al. (2015) stated that turmeric had a synergistic effect with an oral anti-diabetic drug (metformin) in diabetics by lowering fasting blood glucose. Moreover, Rajeshwari et al. (2011) suggested that aniseeds could significantly decrease fasting blood glucose, protein and lipid oxidation in diabetic patients. Furthermore, thyme showed reducing effect in body weight, triacylglycerols, and blood pressure, whereas anise seeds were more effective in fasting blood sugar and increasing in body weight (Anwar 2017). However, the hypoglycemic effect of coriander seeds has been reported. Eidi et al. (2009) observed that the ethanol extract of coriander (200 and 250 mg/kg, i.p.) lowered the serum glucose in streptozotocin-induced diabetic rats and improved insulin release from the number of β cells of the pancreas. Besides, dill is another promising spice which has beneficial effects on the control and management of cardiovascular disorders and diabetes. Goodarzi et al. (2016) summarized that dill showed hypolipidemic and hypoglycemic effects by affecting antioxidant activity and modification of few genes in glucose and lipid pathways. Moreover, fenugreek showed potential hypocholesterolemic properties on type 1 and type 2 diabetes. Xue et al. (2007) stated that fenugreek extract can reduce kidney/body weight ratio, blood lipid levels, blood glucose, and increase hemorheological effects in diabetic rats for 6 weeks. Furthermore, garlic exhibited potential antidiabetic activity due to the presence of volatile sulfur compounds like allicin, diallyl disulfide, diallyl sulfide, diallyl trisulfide, and S-allyl cysteine. For example, S-allyl cysteine treatment can restore erectile mechanism in diabetic rats by preventing reactive oxygen species (ROS) formation via modulation of NADPH oxidase subunit expression (Yang et al. 2013). On the other hand, Dehghan et al. (2016) recommended that saffron consuming alongside exercise might progress diabetic parameters via redox-mediated functions as well as a GLUT4/AMPK pathway to entrap glucose uptake.
5.3. Cardioprotective activity
Cumin extract intake at 200 mg/kg (body weight) for a period of 9 weeks improved plasma nitric oxide as well as reduced the systolic blood pressure in hypertensive rats (Kalaivani et al. 2013). Moreover, ginger oleoresin lowered serum cholesterol level when rats were fed 10% ginger in a 1% cholesterol diet for 24 days (Giri et al. 1984). The black pepper and its constituents, mainly piperine, also showed a significant effect in reducing the extent of cardiovascular diseases. Taqvi et al. (2008) observed that piperine (1 to 10 mg/kg) possessed a Ca2+ channel blockade effect that is responsible for the cardio-depressant and vasodilator properties, and exhibited a blood pressure lowering effect. Moreover, piperine (20 mg/kg/day) lowered the blood pressure caused by chronic L-NAME (N(G)-Nitro-L-arginine methyl ester) administration (Hlavackova et al. 2010). Furhermore, the methanol extract of fennel showed hypolipidemic and anti-atherosclerotic properties by reducing the lipid level of serum and liver, and also facilitated the blood flow rate in the coronary arteries (Oulmouden et al. 2014). However, garlic, mainly allicin, plays a role in delaying or preventing cardioprotective activity by reducing cholesterol level, blood pressure, and inhibiting platelet aggregation, and increasing antioxidant status (Brace 2002; Chan et al. 2013; Rahman and Lowe 2006). Besides, Durak et al. (2004) analyzed the effects of garlic extract on blood lipid profile and antioxidant status. They found that low-density lipoprotein, very-low density lipoprotein cholesterols, serum total cholesterol, and triacylglycerol levels were notably lower after the treatment of extract, while the opposite scenario was seen in high-density lipoprotein cholesterol. Similarly, garlic (allicin) showed a significant effect in reducing blood cholesterol, glucose level, triacylglycerol, and caused a favorable decrease in minimizing hepatic cholesterol storage (Lu et al. 2012). However, quercetin is the major component of onion which may prevent cardiovascular diseases due to its vasorelaxant and antihypertensive properties. Brüll et al. (2015) suggested that supplementation with quercetin (162 mg/d) from onion skin extract lowered ambulatory blood pressure in patients with hypertension, thus proposing cardioprotective activity of quercetin.
5.4. Anti-inflammatory activity
Ginger extract (255 mg twice a day for 6 week) reduced the symptoms of osteoarthritis (knee) of 247 osteoarthritic patients (Altman and Marcussen 2001). Moreover, Güez et al. (2017) stated that basil extracts possessed potential anti-inflammatory activities via inhibition of pro-inflammatory mediator and the stimulation of anti-inflammatory cytokines. However, the major compound of black pepper is piperin which has potent anti-inflammatory effects. Bang et al. (2009) suggested that piperine (20 and 100 mg/kg/day) has anti-inflammatory activity on IL1β-stimulated FLSs and antirheumatic properties in animal models. Essential oils of cardamom show biological activity in human dermal fibroblasts. Han and Parker (2017) reported that cardamom essential oil has potential anti-inflammatory and immunomodulatory properties. Recently, Kazemi et al. (2017) claimed that cardamom improved inflammatory indices and oxidative stress in pre-diabetic women. However, eugenol and acetyleugenol are the major compounds of clove which displayed potential immunomodulatory/anti-inflammatory effects. Bachiega et al. (2012) found that clove exerted anti-inflammatory properties by inhibiting lipopolysaccharide, and the mechanism of action could be the nuclear factor-κB pathway by eugenol. Moreover, cumin essential oil was examined to analyze the anti-inflammatory activity in lipopolysaccharide-stimulated RAW 264.7 cells (Wei et al. 2015). Results showed that essential oils suppressed anti-inflammatory activity in lipopolysaccharide-stimulated RAW 264.7 cells through inhibition of nuclear factor-kappa B activation and mitogen-activated protein kinases extracellular signal-regulated kinase, and c-Jun N-terminal kinase signaling. However, Payahoo et al. (2014) suggested that inflammatory response improved via an effective reduction in inflammatory biomarkers of type 2 diabetes patients when supplemented with dill seed for 8 weeks. Moreover, fenugreek seeds have been used as antinociceptive, hypoglycemic, and anti-inflammatory agents. Mandegary et al. (2012) concluded that fenugreek seeds can be responsible for anti-inflammatory and antinociception effects due to the presence of flavonoid and alkaloid, respectively. In addition, the methanol extract of fenugreek inhibited the synthesis of phorbol-12-myristate-13-acetate-induced inflammatory cytokines, including tumor necrosis factor-α in cultured THP-1 cells, and production of melanin in murine melanoma B16F1 cells (Kawabata et al. 2011).
| 6. Application of spices | ▴Top |
Spices have been used as colorant, flavoring, antioxidant, and antimicrobial agents in food, pharmaceutical, nutraceutical, perfumery, and cosmeceutical industries. Generally, spice extracts and essential oils are used in foods due to its inhibitory effects against lipid oxidation and microbial growth. Spices are not only used for minimizing oxidative rancidity but also used for reducing the formation of harmful substances such as heterocyclic amines (Rounds et al. 2012). Moreover, spices as a hydrocolloid, which gives textural, thickening, emulsifying, appeal, gelling, stabilizing, and encapsulating properties in foods. For example, fenugreek is used as a food adhesive, stabilizer, and emulsifying agent in bakery products (Wani and Kumar 2016). It can be also used in bakery products by fortification of flour with soluble dietary fiber. In addition, spices can be used to formulate capsules or tablets along with the other nutrients and vitamins. There are many pharmaceutical products that are currently marketed, such as pain relief products VorwerkTM and ZostrixTM; which are made by 3% capsicum oleoresin and 0.75% capsaicin, respectively (Peter 2001).
6.1. Coloring agent
The most common spices such as turmeric, saffron, ginger, red pepper, mustard, and paprika are used as natural sources of colorants. The major component responsible for the color are carotenoids; mainly β-carotene, neoxanthin, and lutein. Besides, flavonoids are responsible for yellow color, chlorophyll provides green, whereas curcumin is responsible for orange color. For example, curcumin, a common bioactive compound found in turmeric used for fabric and food colorations due to its antimicrobial properties. Han and Yang (2005) suggested that curcumin can be treated in wool fabric due to its inhibitory activity against Staphylococcus aureus and Escherichia coli. Moreover, Arimboor et al. (2015) reported that red pepper carotenoids as a source of natural food colors due to their nutritional characteristics and the potential role in prevention and protection against degenerative diseases. In addition, crocin, a water-soluble compound found in saffron, is the major pigment responsible for the coloring strength of the spice. Addition of saffron in pasteurized ewe milk pressed cheeses improved the microbiological, color, textural, and sensory characteristic of the cheese (Licón et al. 2012). Particularly, color was the main aspect altered by the use of saffron. Besides, the addition of 0.2% (w/w) of rosemary and marjoram could decrease the degradation of pigment degradation in tomato products during storage (Biacs and Wissgott 1997).
6.2. Flavoring agent
Spices provide pleasant flavor due to the presence of monoterpenes, phenylpropanoids, and other phenolic compounds. The major flavor components of spices are linalool/methyl chavicol (basil), camphor/1,8-cineole/borneol (rosemary), thymol/carvacrol (thyme), cinnamic aldehyde/eugenol (cinnamon), eugenol (clove), and myristicin/α-pinene/sabinene (nutmeg) (Tajkarimi et al. 2010). For example, cumin seeds are used in cooking, while its volatile oil is used for food flavoring (mainly brown rice and sauteed vegetables), perfumery, and cosmetics industries (Sowbhagya 2013). Particularly, spice oil is used as a fragrant component of lotions, creams, and perfumes and also in the food processing industries. For example, nutmeg and mace oils are used in men’s perfume, cosmetics, and toiletries due to their aromatic properties. Moreover, Supartono et al. (2015) suggested that spices can be used as a flavoring agent in green tea due to their high consumer acceptance. In addition, spices such as pepper, capsicum, ginger, nutmeg, and turmeric are used for making the seasoning mix for fresh sausages due to their flavor and keeping quality to the products. However, the addition of spices (mainly cinnamon) improved the sensory quality (taste and smell), antimicrobial, and antioxidant activities of honey (Wilczyáska et al. 2017). In addition, application of ultrasound treatment during cooking of spiced beef significantly increased the content of sodium chloride, essential amino acid, TBARS values, and volatile flavor substances (aldehydes, alcohols, and ketones) (Zou et al. 2018).
6.3. Natural antioxidants
Spices are used as natural antioxidants in foods with the aim of preserving lipid components from deterioration. Antioxidant activity of ground ginger, oregano, clove, rosemary, thyme, and sage was investigated in a comminuted pork system (Shahidi et al. 1995). Results suggested that spices (200–2000 ppm) inhibited the formation of the 2-thiobarbituric acid reactive substances (TBARS) up to 96% for 21 days of storage at 4 °C. Similarly, the addition of spice extracts (rosemary, cloves, black mustard, oregano, and cinnamon) inhibited microbial growth, improved sensory attributes, and reduced lipid oxidation of raw chicken meat during refrigerated storage (Radha Krishnan et al. 2014; Zhang et al. 2016). Moreover, antioxidant potential and consumer acceptability of probiotic-yogurt with spice oleoresins (cardamom, nutmeg, and cinnamon) have been studied (Illupapalayam et al. 2014). Results suggested that spice oleoresins provided health-promoting properties based upon antioxidant potential without affecting the fermentation kinetics. Furthermore, Armellini et al. (2018) found that addition of saffron in wheat flour pasta significantly influenced antioxidant activity and quality parameters, such as color, aroma, and taste.
6.4. Antimicrobial agents
Species are effective against inoculated microorganisms on foods (meat and fish), especially against Gram-positive and Gram-negative bacteria. The presence of salt, carbohydrates, proteins, fat, and a low water activity of essential oils cause a decrease of the microbial load (Klein et al. 2013). Due to their antimicrobial and flavoring properties, spices have been used traditionally in processed meats, fish, vegetables, soups, sauces, chutneys, dressings, dairy products, confectionery, and beverages. For example, Kong et al. (2007) reported that spice extracts (honeysuckle, skullcap, cinnamon, rosemary, and clove) produced strong antimicrobial effects in vacuum-packaged fresh pork, therefore spices can be used as a natural antimicrobial agent for the inhibition of pathogenic and spoilage microorganisms in foods. Moreover, Abou-taleb and Kawai (2008) analyzed the shelf life of semi-fried tuna slices coated with 1% essential oil (0.5% eugenol and 0.5% linalool) in order to evaluate sensory acceptability and microbiological activity. Treatment with 1% essential oil significantly extended the shelf life of coated semi-fried slices and suppressed lipid oxidation during storage. Furthermore, commercial cinnamon essential oils inhibited the growth of microorganisms more firmly than methylparaben in a cosmetic emulsion (Herman et al. 2013). In addition, DL-lactide-co-glycolide, a biocompatible polymer can be prepared from cinnamon bark essential oils by using an emulsion-solvent evaporation method. Hill et al. (2013) suggested that poly DL-lactide-co-glycolide nanoparticles commonly used in the pharmaceutical industry for drug delivery and which could be used in food industry as well due to its antimicrobial activities. In addition, Sahu et al. (2017) analyzed the potential biogas production of kitchen waste in the presence of spices (garlic, red chili, clove, cinnamon, coriander, turmeric, black pepper, and cardamom) and suggested that the spices can effectively decrease methane production and population of methanogenic and fermentative bacteria. The inhibitory effect of the spices on biomethanation could be due to the high concentration of heavy metals along with other bioactive compounds of the spices. Besides, incorporation of turmeric extracts in chitosan effectively enhanced the antimicrobial activity and improved the ultraviolet-visible light barrier and tensile strength of the film. Therefore, the addition of turmeric extract in chitosan films can be suggested as potent coating agents in food packaging (Kalaycıoğlu et al. 2017). Recently, Vidanagamagea et al. (2016) suggested that 3% cinnamon extract can be used to formulate an antioxidant-rich butter and as a natural preservative for butter preparation due to its low levels of free fatty acids value, peroxide value, and low microbial count.
| 7. Toxicity and allergy | ▴Top |
Spices are generally considered as safe, but sometimes can cause carcinogenicity when very high doses are consumed. Apart from their flavoring and coloring effects, some spices contain active and irritant constituents that can produce carcinogenicity and toxicity under specific circumstances. For example, curcumin is considered as a potential source for the prevention and therapy of cancer, but it can exert toxic and carcinogenic properties under particular conditions (Lopez-Lazaro 2008). Moreover, ingestion of cinnamon oil may cause central nervous system depression (Barceloux 2009). In addition, some compounds of fennel and aniseed have come under scrutiny due to their possible toxic effects, particularly estragole and anethole which are chemically related to safrole, a known carcinogen and hepatotoxin. It was found that estragole possessed the possible development of malignant tumors in rodents and induced potential carcinogenesis in humans (Rather et al. 2016). Besides, allicin, a sulfur-containing compound of garlic has the ability to interact with –SH groups of enzymes and may be responsible for toxic action. It has been reported that the di (prop-2-enyl) disulfide of garlic may cause contact dermatitis and occupational allergy (Peter 2001). However, spices may cause allergic reaction; thus it is suggested that the use of spice essential oil should be avoided in any inflammatory, dermatitis, or allergic skin conditions. For example, due to the presence of one or more sesquiterpene lactone, bay leaves may cause allergic contact dermatitis (Peter 2001). Moreover, saffron may act as a “hidden” food allergen in a curry mix and may cause life-threatening anaphylaxis (Wüthrich et al. 1997).
| 8. Conclusions | ▴Top |
The major bioactive compounds of spices are phenolic compounds, phenolic diterpenes, tannins, sulfur-containing compounds, alkaloids, and vitamins. However, understanding their detailed chemical structures and biological function are still a major challenge. Based on the literature reviewed here, we can conclude that spices not only enhance the flavor, color, and aroma of foods, but they can also protect the human body from various chronic diseases due to their strong antioxidant properties. Both in vitro and in vivo studies have shown that spices act as anti-inflammatory, antioxidant, anticarcinogenic, antihyperglycemic, digestive stimulant, cardioprotective, and antimicrobial activity. However, it is important to characterize the bioactive constituents and their pharmacological effects towards clinical human models. Moreover, spices can inhibit both human pathogens (food safety) as well as food spoilage microorganisms (food preservation) due to the antimicrobial property of their phytochemicals. Furthermore, due to their antimicrobial activity, spices and their essential oils can be used as additives in edible or biodegradable films and also coatings for active food packaging in order to prolong shelf-life and functionality of the products. Currently, spices are used in food, nutraceutical, pharmaceutical, perfumery, and cosmeceutical industries as coloring, flavoring, and preservative agents, nevertheless, these applications are still under limitation. Therefore, utilization of spices for developing functional foods such as ready-to-eat food products may help to develop the quality of life by inhibiting the occurrence of diseases. In addition, although spices are generally recognized as safe, yet they may cause toxicity and allergy under specific circumstances.
| References | ▴Top |