| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Original Research
Volume 21, March 2023, pages 44-54
Quantitation of key odorants in arginine/cysteine-glucose maillard reactions
Robert J. Cannona, b, *, Chi-Tang Hob
aMcCormick & Company, 204 Wight Avenue, Hunt Valley, MD 21031, USA
bDepartment of Food Science, Rutgers University, 65 Dudley Road, New Brunswick, NJ 08901, USA
*Corresponding author: Robert J. Cannon, McCormick & Company, 204 Wight Avenue, Hunt Valley, MD 21031, USA. E-mail: robert_cannon@mccormick.com
DOI: 10.31665/JFB.2023.18338
Received: February 20, 2023
Revised received & accepted: March 19, 2023
| Abstract | ▴Top |
Unlike many other named reactions in organic chemistry, the Maillard reaction is not a clearly defined single pathway. Instead, it is one of the most complex reactions in food chemistry. Maillard reactions, studied in the literature over the last fifty years, have focused either on simple model systems or complex culinary processes. The next step in our understanding of Maillard reactions is to bridge the gap between these simple and complex systems. By adding more than one amino acid to a model system, we aim to better understand the different mechanisms for which certain aroma compounds are formed. In this study, L-arginine and L-cysteine were selected as the two amino acids in the thermal reaction with glucose. Under different conditions, the key odorants from several arginine-cysteine/glucose model studies were identified and quantified by gas chromatography-mass spectrometry and gas chromatography-olfactometry.
Keywords: Maillard reaction; Cysteine; Arginine; Key odorant
| 1. Introduction | ▴Top |
The Maillard reaction (Hodge, 1953) has garnered immense research due to the complex mechanisms and products formed that generate flavor, color, and physiological consequences. The Maillard reaction comprises several reactions including rearrangement, fragmentation, degradation, and dehydration.
The initial stage of the reaction is driven by the condensation of a reducing sugar and an amino compound. This condensation product can then generate a cascade of reactive intermediates such as Amadori compounds which ultimately produce aroma, flavor, and color compounds in foods. The formation of final products from the Amadori intermediates depend mainly on the pH of the system. At low pH, the route of 1,2-enolisation via 3-deoxy-1,2-carbonyls is favored. At high pH, 2,3-enolisation via 1-deoxy-2,3-carbonyls is favored (Nursten, 2005; Cui et al., 2021).
For simple Maillard model systems, the mechanisms for which volatiles are generated depend principally on the pH, temperature, amount of oxygen, and the concentration of amino acid and reducing sugar. One could imagine how complicated the reactions and mechanisms for which volatiles are generated in more complex Maillard systems, where there could be multiple amino acids and reducing sugars, as well as proteins, lipids, polyphenols, and other chemical constituents in natural foodstuffs. The more we continue to learn and document the analytical and sensory data from complex Maillard reactions, the better chance of guiding product development to deliver high impact ingredients in high purity from these processed reactions (Lund and Ray, 2017).
Arginine has been studied for its ability to be reversibly modified with α-dicarbonyl compounds including, glyoxal (Schwarzenbolz et al.,1997, Glomb and Lang, 2001), methylglyoxal (Klopfer et al., 2011), and 3-deoxyglucosone (Hayase et al., 1995). Glyoxal and methylglyoxal, both degradation products of reducing sugars like glucose, are highly reactive carbonyl compounds that are responsible for color and flavor generation in the Maillard reaction (Wang and Ho, 2012). The condensation products of these α-dicarbonyl compounds with the guanidine group have been identified and often characterized for their ability to limit the hydrolysis by trypsin. More recently, research by Zhu and Yaylayan (2017) investigated the reactivity of guanidine and arginine with glucose. Results showed that free guanidine will condense with one, two, or three glucose molecules. However, the guanidine group of arginine could not be released during the Maillard reaction, so these condensation Amadori rearrangement products would not form. For the reaction of arginine and glucose, the authors identified Amadori products at the α- and ε-amino groups. At the ε-amino group, arginine reacted with methylglyoxal and 3-deoxyglucosone to form imidazolone derivatives.
The volatile compounds generated from the reaction of L-cysteine with different reducing sugars have been detailed in numerous papers over the years, including cysteine-xylose (Tressl et al., 1989; de Roos et al., 2005), cysteine-glucose (Tressl et al., 1989; Kato et al., 1973), cysteine-arabinose (Tressl et al., 1994), cysteine-rhamnose (Hofmann and Schieberle, 1997; 1998), cysteine-ribose (Mulders, 1973; Zhang and Ho, 1991; Mottram and Nobrega, 2002; Cerny and Davidek, 2003). In the thermal reaction (145 °C) of cysteine and ribose, 29 odor-active volatiles were identified by gas chromatography-olfactometry (GC-O) and Aroma Extract Dilution Analysis (AEDA) through the work of Hofmann and Schieberle (1995). The same authors continued to study these reactions with different carbohydrates in both aqueous and dry heated systems (Hofmann and Schieberle, 1997; 1998). The intensity of the odorants using AEDA varied significantly. For example, pyrazines were not detected by GC-O in the aqueous systems for all three carbohydrate systems. However, in the dry heated treatments, pyrazines contributed significantly to the odor of the reaction. In addition, the dry heated conditions of cysteine-ribose produced a flavor dilution factor of furfuryl mercaptan four times that of the aqueous cysteine-ribose reaction.
The objectives for this research include the identification and quantitation of the key odorants from arginine/cysteine-glucose model studies by gas chromatography-mass spectrometry (GC-MS) and GC-O. Established literature are used to discuss the mechanisms for which several key odorants are formed.
| 2. Materials and methods | ▴Top |
2.1. Materials
The following reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA): L-arginine, L-cysteine, glucose, 3-mercaptopropionic acid, and 2-mercaptopropionic acid. Three molecules were purchased from Enamine (Kyiv, Ukraine): 2-(5-methylpyrazin-2-yl)ethan-1-ol; 2-(pyrazin-2-yl)ethan-1-ol; and 2-(6-methylpyrazin-2-yl)ethan-1-ol. Diethyl ether was purchased form Fisher Scientific (Waltham, MA, USA).
2.2. Preparation of maillard reactions
Equimolar ratios of glucose (0.90 g; 5 mmol) and L-arginine (0.87 g; 5 mmol) were added to distilled water (50 mL). An initial pH of 10.4 was measured for the solution. The pH of the arginine solution was reduced to 7.4 by dropwise addition of 1 M HCl and added to 75 mL reaction vessels. The vessels were heated in an oil bath for 1 h at three different reaction temperatures (100 °C; 130 °C; 160 °C). The same reaction was also carried out at the initial pH of 10.4 with a reaction temperature of 160 °C. Upon completion of the reaction, the vessels were added to an ice bath to cool to room temperature and a second pH measurement was made.
Equimolar ratios of glucose (0.90 g; 5 mmol) and L-cysteine hydrochloride (0.79 g; 5 mmol) were added to distilled water (50 mL). An initial pH of 1.7 was measured for the solution. The pH of the cysteine solution was increased to pH 7.4 by dropwise addition of 2 M NaOH and added to 75 mL reaction vessels. The vessels were heated in an oil bath for 1 h at three different reaction temperatures (100 °C; 130 °C; 160 °C). Upon completion of the reaction, the vessels were added to an ice bath to cool to room temperature and a second pH measurement was made.
Equimolar ratios of glucose (0.90 g; 5 mmol), L-cysteine hydrochloride (0.79 g; 5 mmol), and L-arginine (0.90 g; 5 mmol) were added to distilled water (50 mL). An initial pH of 5.9 was measured for the solution. The pH of the solution was increased to pH 7.4 by dropwise addition of 1 M NaOH and added to 75 mL reaction vessels. The vessels were heated in an oil bath for 1 h at three different reaction temperatures (100 °C; 130 °C; 160 °C). Upon completion of the reaction, the vessels were added to an ice bath to cool to room temperature and a second pH measurement was made.
2.3. Liquid-liquid extraction and solvent assisted flavor evaporation (SAFE) of Maillard reaction
The above resulted reaction mixture was allowed to cool to room temperature, transferred to a separatory funnel, and extracted twice with 25 mL of diethyl ether using an internal standard of octanol at 1 ppm. The extracts were combined, dried over anhydrous sodium sulfate, filtered, and added to a SAFE apparatus for isolation of the volatile compounds. The sample was added dropwise to the system while the vacuum was kept below 5 × 10−4 Torr. The frozen extract was allowed to thaw and finally reduced under a gentle stream of nitrogen to 250 µL.
2.4. Gas chromatography
The extract was analyzed on apolar and polar phase columns using an Agilent 7890A gas chromatograph (Santa Clara, CA, USA). The apolar capillary column had dimensions of 50 m × 320 µm × 0.52 mm (Restek RTX-1 F&F), and the polar capillary column had dimensions of 50 m × 320 µm × 0.5 mm (Varian CP-Wax 58 FFAP CB column). Samples were introduced to the gas chromatograph (GC) using an autosampler at a volume of 1 µL with a split ratio of 5:1. The hydrogen carrier gas flow rate was held constant at 2 mL/min, and the temperature program started at an initial temperature of 40 °C, then increased 2 °C/min up to 250 °C with a 10 min hold at 250 °C. The end of each apolar and polar column was affixed to a flame ionization detector (FID). The GC was calibrated using a homologous series of C1-C18 ethyl esters to generate retention index values for the observed peaks. The retention index values were calculated based on previous work by van den Dool and Kratz (1963), who considered the GC oven temperature program and non-alkane based calibrants, specifically ethyl esters. While this is an industry standard, it is commonly expected to report Kovats retention indices (RI), which are based off calibrations with a homologous series of alkanes. A linear relationship between the ethyl ester values and Kovats RI is found when plotting the measured values for the homologous series of the ethyl esters versus the Kovats values. Based on this relationship the following equation is derived:
It is with this equation that the Kovats values reported here have been calculated.
2.5. Gas chromatography-olfactometry
Each extract was analyzed on an apolar 50 m × 320 µm × 0.5 µm column (Restek) using an Agilent 6890A GC. All samples were introduced to the GC inlet using an autosampler at a volume of 1 µL with a 5:1 split ratio. The following parameters remained constant for all samples. The hydrogen carrier gas flow rate was 2 mL/min, and the temperature program started at an initial oven temperature of 40 °C, then increased 6 °C/min up to 80 °C, 4 °C/min up to 150 °C, 2 °C/min up to 200 °C, and finally 10 °C/min up to 310 °C and held for 5 min. In addition, an Olfactory Detection Port (ODP; GERSTEL, Inc., Linthicum, MD, USA) was equipped on the GC. The effluent was split 6:1 (ODP:FID), and the ODP transfer line heated to 225 °C. Olfactory comments were recorded using Dragon Naturally Speaking, Speech Recognition Software 12.0 (Nuance Communications, Inc., Burlington, MA, USA) in conjunction with the ODP software. The GC was calibrated using a homologous series of C1-C18 ethyl esters to generate index values for the observed peaks and then converted to Kovats values using the aforementioned equation.
2.6. Gas chromatography-mass spectrometry
The chromatographic conditions were the same as described for the GC analysis. All data was acquired using a Waters GCT-Premier orthogonal acceleration time-of-flight (TOF) mass spectrometer (Milford, MA, USA) in electron ionization (EI) mode. The ion source was operated at 150 °C with an electron energy of 70 eV and a trap current of 50 µA. The temperature of the transfer line was 250 °C. Spectra were acquired between 27 and 400 Da in a time of 0.05 s and a delay of 0.01 s (approximately 20 spectra/s). Exact mass spectra were obtained using a single-point lock mass (m/z 218.9856 from perfluorotri-n-butylamine) infused into the ion source continuously during the run. Mass spectral library identification was achieved using the data acquired from synthesized or purchased authentic standards. Standard relative retention data was used for confirmation, which was obtained by calibrating the instrument with a homologous series of ethyl esters.
| 3. Results and discussion | ▴Top |
3.1. Maillard reaction of arginine-glucose
L-Arginine, a basic amino acid with a positively charged guanidine group, is found in many animal (meats, fish, eggs) and plant (nuts, grains, seeds) sources. This amino acid (Figure 1a) contains both an α- and ε-amino groups and has an isoelectric point at pH 10.8. Only a few references in the literature discuss the chemistry of arginine in the Maillard reaction. In fact, no reference has yet to identify the Strecker aldehyde of arginine. Strecker aldehydes are an important class of flavor chemicals that provide common aromas in foods. Strecker aldehydes are formed from the reaction of free amino acids and α-dicarbonyl compounds, which are typically Maillard reaction intermediates from thermal processing.
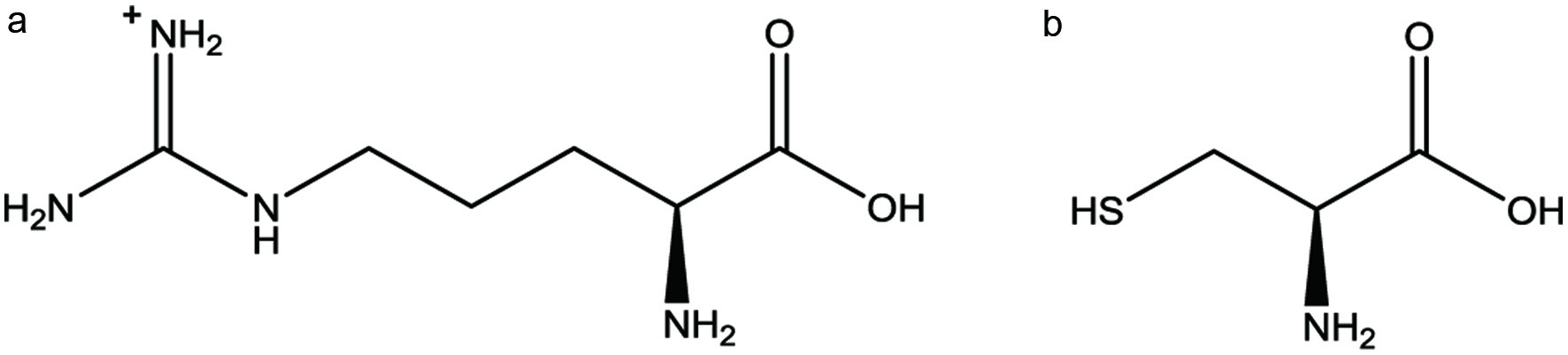 Click for large image | Figure 1. The chemical structure of (a) L-arginine and (b) L-cysteine. |
The focus of this research was to measure the volatile compounds produced from several arginine reactions, including changes in reaction temperature and pH. First, the analytical and sensory data for three reaction temperatures (100 °C, 130 °C, 160 °C) were acquired at the same adjusted pH of 7.4 (initial pH 10.4). There were clear differences in the color at the three different reaction temperatures. At 100 °C, very little aroma was perceived, and no color change was observed. This linked to the analytical data where only a few trace compounds could be identified. At 130 °C, a weak sweet brown, pretzel-like aroma was detected with a light-yellow color. This temperature yielded slightly higher concentrations of low molecular weight pyrazines and sugar degradation products. At 160 °C, the pH dropped to 5.2 by the end of the reaction and a more pronounced sweet brown, pretzel-like, nutty aroma was perceived, along with a deeper reddish, brown color. The chromatogram for this reaction yielded a series of alkylpyrazines as well as several furans.
In addition, two arginine-glucose (Arg-Glu) reactions were compared under the same reaction temperature (160 °C) at both pH 7.4 and pH 10.4. The initial pH of the solution of Arg-Glu (before heat treatment) measured 10.4. After the Maillard reaction, the pH measured 6.8, which represents a significant drop in pH. When the initial pH was adjusted to 7.4 before heat treatment, the pH after heat treatment measured 5.2. The differences between initial and final pH of both model systems are significant and will undoubtedly impact sugar fragmentation and the conditions suitable to generate Maillard chemistry. This was observed in both the color changes and the concentration of volatile constituents. As mentioned earlier, a deep reddish, brown color is observed in for the reaction at pH 7.4. For the reaction at pH 10.4, the color is significantly darker and appears like balsamic vinegar.
The analytical data for each of the arginine-glucose reactions can be found in Table 1. The total concentration of volatile compounds increased as the temperature and pH of the reaction increased. At pH 7.4, a total of 7.1 mg/mol of pyrazines was measured. At pH 10.4, a total of 207.9 mg/mol of pyrazines was measured. The largest differences were found in the concentration of pyrazine, methylpyrazine, 2,5-dimethylpyrazine and 2,6-dimethylpyrazine. At the higher initial pH, sugar fragmentation is catalyzed more readily under these basic conditions. Sugar degradation products, including glyoxal and methylglyoxal, react readily with the amino group of arginine to form two and three-carbon α-amino fragments, which are the building blocks of substituted alkylpyrazines. For example, the condensation of aminoacetaldehyde and aminoacetone will form methylpyrazine. This compound measured 0.4 mg/mol at pH 7.4 but increases to 105.4 mg/mol at pH 10.4, which is the highest concentration pyrazine derivative in the reaction. This data aligns with previous studies under similar conditions (Huang et al., 1989) and suggests a significant increase in the sugar fragmentation and the readily available condensation reactions that occur at higher pH. Due to 2,5-dimethylpyrazine and 2,6-dimethylpyrazine having similar chemistry, neither the polar nor apolar columns could resolve both peaks on a one-dimensional GC column. Therefore, the coeluting peaks are reported as a combination of both compounds in Table 1. Similar challenges to resolve these pyrazines have also been noted by other authors (Adams et al., 2008). Nonetheless, similar concentration differences to methylpyrazine were observed for the combination of 2,5-dimethylpyrazine and 2,6-dimethylpyrazine at different pH. At pH 7.4, both compounds measured 0.2 mg/mol, while at pH 10.4, both compounds measured 50.1 mg/mol.
 Click to view | Table 1. The volatile compounds from the Arg-Glu reactions |
Additional quantitative differences were observed for the furan-type compounds at each pH. An example includes furaneol (2,5-dimethyl-4-hydroxy-3(2H)-furanone), which is an important aroma compound found in many foods (Wang and Ho, 2008; Xiao et al., 2021). At pH 10.4, the concentration of furaneol measured 8.3 mg/mol. At pH 7.4, the concentration of furaneol measured 0.5 mg/mol, which shows a pH dependence on furaneol formation. Numerous studies have investigated the formation pathways of furaneol depending on the reducing sugar and addition of amino acid (Hofmann and Schieberle, 1997; Haleva-Toledo et al., 1999; Wang et al., 2009). Glucose, a hexose sugar, generates lower concentrations of furaneol compared to rhamnose, a 6-deoxyhexose sugar (Hofmann and Schieberle, 1997). The data from the Arg-Glu reaction suggests that the major formation pathway of furaneol is through the Cannizzaro reaction of methylglyoxal, which requires basic conditions.
A compound with high toxicological risk is 5-hydroxymethyl-2-furfural (5-HMF). This compound is formed from the intermediate 3-deoxyglucosone, which is derived from the 1,2-enolization and dehydration of glucose (Lee and Nagy, 1990). 5-HMF was only detected at pH 7.4 and measured 18.6 mg/mol, which aligns with other research in the literature (Gökmen et al., 2007) and demonstrates the preferential 1,2-enolization pathway at lower pH. Haleva-Toledo et al. (1999) studied the effect of the addition of arginine on the production of 5-HMF from glucose under acidic conditions. The concentration of 5-HMF increased in the presence of arginine. The opposite trend was found with 2-furfuryl alcohol (no toxicological concern) where high concentrations were found at pH 10.4 (34.7 mg/mol) compared to the reaction at pH 7.4 (1.6 mg/mol). This shows that the major precursors to 2-furfuryl alcohol, likely 3-deoxyaldoketose or 2-deoxyribose (Brands and van Boekel, 2001), are generated at higher pH.
GC-O was performed to identify the aroma-active compounds from the Arg-Glu reactions. Odor comments were recorded at the end of the ODP using the Dragon software to record each odor comment. Even though there were similar odorants perceived in both reactions at pH 7.4 and 10.4, the intensity of those odorants (“nutty, burnt, chocolate, pyrazinic”) was significantly higher in the reaction at pH 10.4 (data not shown). There were two separate odor comments from this reaction that were not immediately linked to a known MS library spectrum. Both peaks in the chromatogram were low in concentration (<0.1 mg/mol) but were described as “chocolate, pyrazinic, nutty” and “nutty, popcorn”.
Based on exact mass measurements from a TOF mass spectrometer and elemental composition of the molecular ion and associated fragments, two structures, 2-(pyrazin-2-yl)ethan-1-ol and 2-(5-methylpyrazin-2-yl)ethan-1-ol, were postulated. Along with 2-(6-methylpyrazin-2-yl)ethan-1-ol, the vendor standards were confirmed as the unknowns found in the Arg-Glu reaction. These three compounds are scarcely reported in the literature and still yet to be reported in a natural product. Shu tentatively identified 2-(pyrazin-2-yl)ethan-1-ol in the thermal reaction of glucosamine (Shu, 1998a), and later identified 2-(6-methylpyrazin-2-yl)ethan-1-ol in the reaction of inulin/asparagine and fructose/asparagine (Shu, 1998b). The structure, Kovats indices, and odor descriptors for each of the three hydroxyethyl pyrazines are listed in Table 2.
 Click to view | Table 2. Three hydroxyethyl pyrazines identified in the Arg-Glu reactions. |
A couple of mechanisms are possible to describe the formation of hydroxyethyl pyrazines. Shown in Figure 2, one proposal starts with two equivalents of aminoacetaldehyde to form dihydropyrazine. Glycoaldehyde, one of the most reactive sugar fragmentation products (Nursten, 2005), would then react with the dihydropyrazine intermediate to form an unstable diol. Dehydration followed by the migration of the double bond into the ring forms a more stable hydroxyethyl pyrazine. All three hydroxyethyl pyrazines were identified from an alkaline extraction of a thermal reaction of asparagine-glucose by Bohnenstengel and Baltes (1992). The authors proposed a second possible mechanism for the generation of hydroxyethyl pyrazines whereby the reaction of glyoxal (or methylglyoxal) with the Amadori product of glucose would form 4-(2-pyrazinyl)-1,2,3-trihydroxybutane. Additional oxidation and retro-aldol steps would lead to the formation of the respective hydroxyethyl pyrazines.
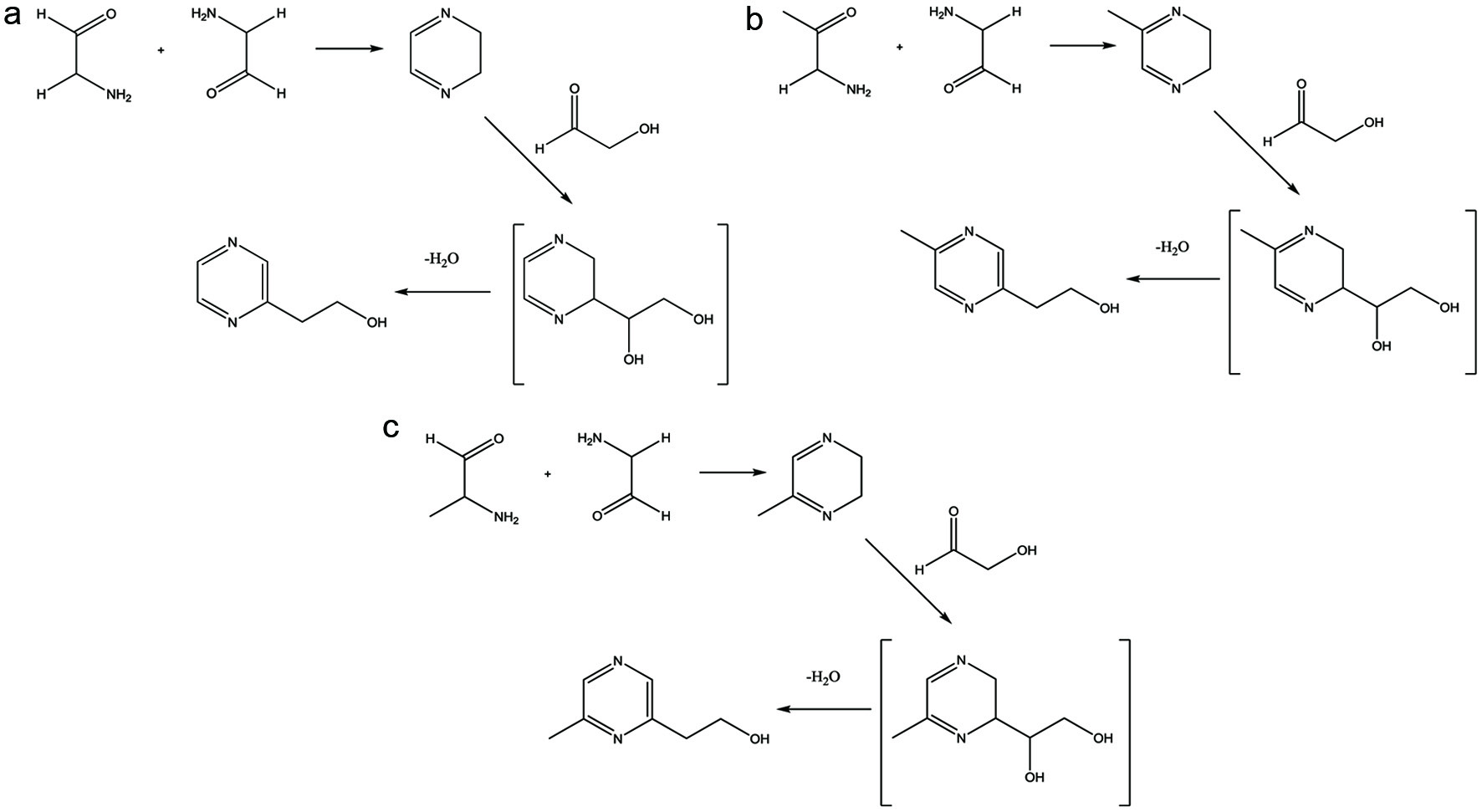 Click for large image | Figure 2. Proposed formation of (a) 2-(pyrazin-2-yl)ethan-1-ol; (b) 2-(5-methylpyrazin-2-yl)ethan-1-ol; and (c) 2-(6-methylpyrazin-2-yl)ethan-1-ol. |
Vinylpyrazines are also prevalent in Maillard reactions and often described as green, burnt, nutty, and coffee-like in aroma. It is plausible that hydroxyethylpyrazines, based on the mechanism described above, act as the intermediates by which water elimination leads to the formation of vinylpyrazines as shown in Figure 3.
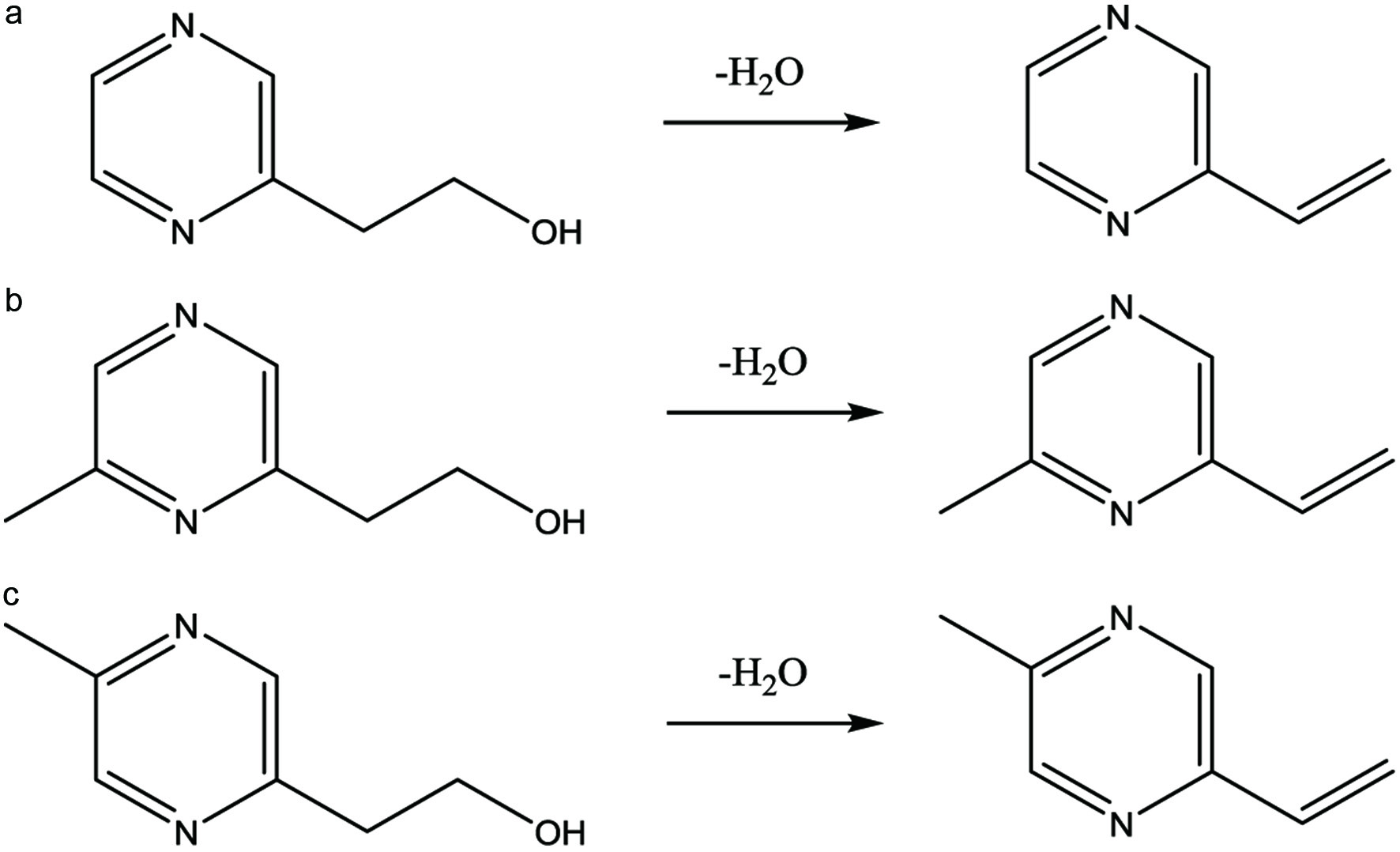 Click for large image | Figure 3. Proposed formation of (a) 2-vinylpyrazine; (b) 2-methyl-6-vinylpyrazine; and (c) 2-methyl-5-vinylpyrazine by water elimination of the respective hydroxyethyl pyrazine. |
3.2. Maillard reaction of cysteine-glucose
In comparison to L-arginine, L-cysteine has been well-studied in the Maillard reaction. L-cysteine is a sulfur-containing amino acid (Figure 1b) with an isoelectric point at pH 5.1. Essential to meat flavors, L-cysteine can generate a significant amount of both hydrogen sulfide and ammonia through several pathways (Kobayasi and Fujimaki, 1965). Hydrogen sulfide and ammonia can then react with many secondary volatiles including carbonyl compounds to produce high impact odorants including thiols, pyrazines, thiazolines, and thiazoles.
Initially, data on the Maillard reaction of Cys-Glu was acquired mainly as a baseline point of reference to compare to the arginine/cysteine-glucose (Arg/Cys-Glu) reactions in the next section. The analytical and sensory data of the Cys-Glu model system were acquired at two reaction temperatures (130 °C; 160 °C) at an adjusted pH of 7.4 (initial pH 1.7). At 130 °C, there was very little color change (slight yellow hue) to the final solution. There was a medium intensity aroma of roasted, meaty notes which was attributed to low concentrations of thiol compounds, including 1-mercaptopropan-2-one and 3-mercaptobutan-2-one. At 160 °C, the pH had dropped to 4.4 by the end of the reaction and the solution was orange in color with a strong aroma of roasted meat and savory notes. By comparing data from the literature, it is evident that the reaction at pH greater than seven is less vigorous compared to reactions closer to the isoelectric point (pH 5.1) of cysteine (Shu et al., 1985a).
The data for the Cys-Glu reactions are presented in Table 3. In contrast to the Arg-Glu reaction, the generation of pyrazines in the Cys-Glu reaction at pH 7.4 was considerably lower by SAFE: 1.6 mg/mol in Cys-Glu compared to 7.1 mg/mol in Arg-Glu. This data differed from similar reactions in the literature. For example, Zhang and Ho (1991) measured 117.6 mg/mol of total pyrazines from the reaction of Cys-Glu at pH 7.5. The temperature of the reaction was slightly higher (180 °C), but the authors incorporated SDE for the isolation of the volatiles. The different extraction methodologies may lead to inconsistencies in data.
 Click to view | Table 3. The volatile compounds from the Cys-Glu reactions at two different reaction temperatures |
The aroma contribution of each pyrazine of the Cys-Glu reaction was studied by GC-O and compared to the data generated from the Arg-Glu reaction. Results from the Cys-Glu extracts produced very few aromas that were described as ‘nutty, burnt, pyrazinic’ (data not shown). Instead, the dominant odorants from the GC-O smell out of the Cys-Glu extract were ‘sulfurous, coffee, roasted, and tropical’. The major constituents linked to these aromas were thiols, including many reported in Table 3. Two mercapto acids, 2-mercaptopropionic acid (185.2 mg/mol) and 3-mercaptopropionic acid (97.4 mg/mol), were found at the highest concentratoins in the reaction. Few references in the literature discuss 2-MPA and 3-MPA in Maillard reactions. Hofmann and Schieberle (1995) identified both 2-MPA and 3-MPA in the reaction of cysteine and ribose at pH 5.0. Tressl et al. (1994) studied the formation of both 2-MPA and 3-MPA through carbon labeling of cysteine in the thermal reaction with glucose. Both reactions started with the initial condensation of cysteine and glucose to form a Schiff base. Strecker degradation is led by the transamination of 2-oxo-3-mercaptopropionic acid and reduction of this α-keto acid to form 3-MPA. Strecker degradation is instead dominated by beta-elimination of hydrogen sulfide and the formation of pyruvate, followed by the addition of hydrogen sulfide to form 2-MPA. Based on the carbon labeling, Tressl et al. concluded that both 2-MPA and 3-MPA formed from the intact carbon skeleton of cysteine. From a similar model system of Cys-Glu, 2-MPA and 3-MPA measured low odor activity values in an AEDA study by Hofmann and Schieberle (1997). This agrees with previously published GC-O data from Maillard reactions involving cysteine where the most intense odorants were linked to sulfur compounds or furans (Hofmann and Schieberle, 1998). These compounds, along with other thiol compounds, are extremely potent odorants in foods. For example, 2-methylfuran-3-thiol, a key odorant found in meat (Kerscher and Grosch, 1998) and chicken (Farkas et al., 1997), measured one of the lowest odor thresholds recorded, between 0.0000025–0.00001 parts per billion (ppb) in air (Gasser and Grosch, 1990). Even at low concentration of 0.4 mg/mol in the Cys-Glu model reaction, 2-methylfuran-3-thiol can significantly affect the aroma profile as also shown in the literature (Hofmann and Schieberle, 1997). In 2005, de Roos et al. quantified several high impact thiols from the reaction of cysteine with different carbonyl compounds (de Roos et al., 2005). Albeit under different reaction conditions (pH 5.0; open system), the authors found that the Cys-Glu reaction produced significantly lower concentrations of 2-methylfuran-3-thiol, fufuryl mercaptan, 3-mercaptopentan-2-one, and 2-mercaptopentan-3-one, compared to the cysteine-ribose, cysteine-xylose, and cysteine-norfuraneol model systems.
Two less common thiophenones, 2,5-dimethyl-4-hydroxy-3(2H)-thiophenone (DMHT; 7.5 mg/mol) and 2,5-dimethyl-2,4-dihydroxy-3(2H)-thiophenone (DMDHT; 5.7 mg/mol), were also identified and quantified in the Cys-Glu reaction at 160 °C (Figure 4). Previously identified in yeasts extracts (Munch et al., 1997), DMHT has been also identified in Maillard reactions of cysteine-furaneol (Shu et al., 1986; Shu and Ho, 1988; Zheng et al., 1997), cysteine-glucose (Hofmann and Schieberle, 1997), and cystine-furaneol (Shu et al., 1985b). DMDHT has been identified in soy sauce (Satoh et al., 2011) and garlic (Hwang et al., 2007; Molina-Calle et al., 2017), as well as in Maillard reactions of cysteine-furaneol (Shu et al., 1986; Shu and Ho, 1988), cysteine-glucose (Tressl et al., 1989), and cystine-furaneol (Shu et al, 1985b). Described as possessing a pot roast-like aroma, DMHT and DMDHT were reported in the Maillard reactions referenced above at low pH (2–6) and higher temperatures (130–160 °C). The model systems suggest that water elimination from glucose at pH 5–7 will yield 3,4-dihydroxy-3-hexene-2,5-dione, which would further react with hydrogen sulfide to form DMHT and DMDHT in a ratio of 10:1 (Tressl et al., 1989). Another possible mechanism for the formation of DMHT could be through the hydrolysis of furaneol and subsequent addition of hydrogen sulfide and loss of water.
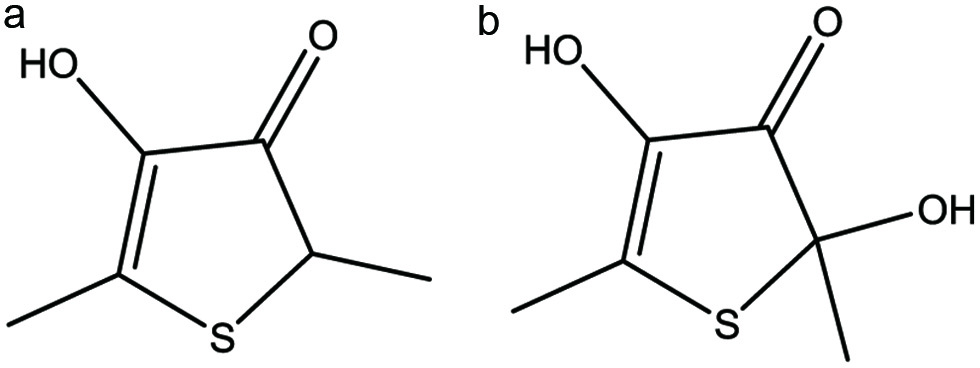 Click for large image | Figure 4. The structures of (a) 2,5-dimethyl-4-hydroxy-3(2H)-thiophenone (DMHT) and (b) 2,5-dimethyl-2,4-dihydroxy-3(2H)-thiophenone (DMDHT). |
As discussed in the previous section, 5-HMF was identified at high concentrations in the Arg-Glu reaction at pH 7.4. However, in the Cys-Glu reaction, there was no detected levels of 5-HMF at this pH. This aligns with previous reports by Haleva-Toledo et al. who demonstrated that the addition of cysteine in the reaction with glucose (with and without arginine) significantly reduced the concentration of 5-HMF (Haleva-Toledo, et al, 1999).
3.3. Maillard reaction of arginine/cysteine-glucose
Only a few articles in the literature discussed the reaction of more than one amino acid in the Maillard system. Samsudin et al. (1996) studied the reaction of leucine and valine with glucose. More recently, Hou et al. (2017) synthesized the Amadori intermediates for both cysteine and glycine to study its reaction with a free amino acid and xylose for the enhancement of meat flavors.
The initial data from the single amino acid (arginine or cysteine) with glucose was established in the previous sections. The final measurements in this study focused on the identification of the volatiles formed from the reaction of two amino acids (arginine and cysteine) with glucose. Knowing how each individual amino acid reacts with glucose will help to determine the preferential and competitive reactions that take place when two amino acids are reacted together. In this reaction, both arginine and cysteine were added at equal molar concentrations and reacted with glucose. Like the Arg-Glu reaction, the analytical and sensory data for three different reaction temperatures (100 °C, 130 °C, 160 °C) were acquired at an adjusted pH of 7.4 (initial pH 5.9). There were clear differences in the aroma, color, and chemistry at the three different reaction temperatures. At 100 °C, there was a slight eggy aroma with no color change observed. Only a couple trace compounds were identified at this reaction temperature. At 130 °C, a medium intensity roasted, meaty aroma was observed with a light-yellow color. This temperature yielded low concentrations of furans and sulfur-containing compounds. Finally, at 160 °C, the pH had dropped to 4.2 by the end of the reaction and a stronger roasted, meat aroma was detected along with a deeper orange color. The chromatogram for this reaction yielded a series of furans/pyrans and sulfur compounds.
The analytical data for these reactions are listed in Table 4. The total concentration of volatile compounds clearly increases as the temperature of the reaction increases. There were unfavorable conditions for pyrazine formation, which could be due to several reasons. As shown in the Arg-Glu reaction, pyrazine formation is favored by more basic conditions (pH 10.4 instead of pH 7.4), whereby increased sugar fragmentation yields secondary intermediates (glyoxal and methylglyoxal) that readily react with free ammonia or the amino group of arginine. Secondly, it appears that the thiol group of cysteine inhibits sugar fragmentation and blocks arginine interaction in the formation of pyrazines as discussed in previous sections. Schubert showed that the thiol group of cysteine was highly reactive with glyoxals (Schubert, 1935)
 Click to view | Table 4. The volatile compounds from the Arg/Cys-Glu reactions |
Both DMHT and DMDHT increased significantly in the Arg/Cys-Glu reaction compared to the Cys-Glu reaction. This seems to suggest that the addition of arginine helps to increase the degradation of glucose and release of hydrogen sulfide from cysteine, as both are requirements for DMHT and DMDHT generation. Like the Cys-Glu reaction, 5-HMF was not detected in the Arg/Cys-Glu model system. The thiol group appears to competitively bind with amines and block certain secondary intermediates that would generate 5-HMF, as also shown by Haleva-Toledo et al. (1999).
| 4. Conclusion | ▴Top |
The goal of this research was to increase our understanding of the Maillard reaction by measuring the volatile compounds formed from the addition of one or two amino acids with glucose. Therefore, L-arginine and L-cysteine were selected as the two amino acids in the thermal reaction with glucose. Data has shown that temperature and pH can significantly influence the final concentration of odorants.
| References | ▴Top |