| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Review
Volume 18, June 2022, pages 67-76
Ginger–a potential source of therapeutic and pharmaceutical compounds
Ravindra Verma*, Prakash S. Bisen
School of Studies in Biotechnology, Jiwaji University, Gwalior 474001, India
*Corresponding author: Ravindra Verma, School of Studies in Biotechnology, Jiwaji University, Gwalior 474001, India. Tel: 91-11-7999212685; E-mail: vermaravindra917@gmail.com
DOI: 10.31665/JFB.2022.18309
Received: March 30, 2022
Revised received & accepted: June 24, 2022
| Abstract | ▴Top |
Ginger is traditionally known for its therapeutic and pharmaceutical properties. It has been used widely to treat various health problems such as high blood pressure, coughs, colds, swelling, nausea, rheumatic disorders, vomiting, bronchitis, indigestion, gastric ulcers, and behavioral problems. Shogaol and Gingerol are anti-inflammatory, anti-fever, anti-pain, and anti-cough compounds that may help treat a cold. This review provides an up-to-date understanding of the impact of ginger and its active compounds on human health. Various ginger compounds such as gingerol, shogaols, zingiberene, zingerone, paradols and zingerone are receiving attention for their clinical applications and pharmaceutical properties. Studies indicate that ginger is anti-inflammatory, anti-tumor, antimicrobial, antiemetic, hepatoprotective, and neuroprotective. During the inflammatory response, ginger inhibits (NF-κB) and immune system activation in addition to many other cellular processes. Ginger has shown benefits in preclinical and clinical studies for neurology, cardiovascular disease, and cancer. These findings indicate the necessity for further in vivo and clinical studies.
Keywords: Ginger; Immunity; Pharmaceutical; Phytocompounds; Therapeutic
| 1. Introduction | ▴Top |
Ginger is a spice widely used in traditional medicine and as an ingredient in food. Zingiber officinale is a plant belonging to the Zingiberaceae family. Originally, it was mentioned in Confucius’s Analects (475-221 BC) as a spice for medicinal purposes (Pickersgill, 2005). Scientists have examined the effects of ginger on diseases such as asthma, stroke, diabetes, constipation, nervous disorders, etc., due to its antiviral and antioxidant properties (Butt and Sultan, 2011; Karna, et al., 2012; Mashhadi, et al., 2013). It is an underground rhizome that grows up to 75 cm in height. Ginger grows mainly in tropical and subtropical regions (Vijayan et al., 2020). China is the largest exporter, followed by the Netherlands, Thailand, Peru, and India, while the US is the major importer, followed by Japan, the Netherlands, Pakistan, and Germany. It contains mainly dietary fiber, vitamin E, vitamin B6, iron, magnesium, manganese, potassium, and selenium. In South Asian countries, it is used in many traditional formulations, especially in Ayurveda, a traditional Indian medical system. Therapeutically, it has been used in many Ayurvedic formulations from the ancient period and is also known as “Maha Aushadhi-A Great Medicine” (Narayana, et al., 2000; Abdulwase, et al., 2020). The United States and European countries registered and sold it as a nutraceutical against nausea, motion sickness, and migraine. It is an approved herb by the German Commission E Monographs for dyspepsia and prevents motion sickness (Vasala, 2012; www.herbalgram.org). Ginger is known by different brand names, like African ginger, black ginger, cochin ginger, Imber, Jamaica ginger, race ginger, rhizoma zingerberis, rhizome, sheng jiang, shokyo, zingibain, Zingiber officinale, and Zingiberis. A controlled trial on abdominal distention in post-cesarean women found that the quality of life and the number of patients who could eat was higher in the ginger group than the others. This study confirms that ginger is efficacious in abdominal distention, low-priced herbal folk medicine with minimum side effects (Tianthong and Phupong, 2018). A trial on postoperative nausea and vomiting observed that 1 g of ginger prevents vomiting and nausea. A minor side effect of abdominal discomfort was seen (Chaiyakunapruk et al., 2006). Its anti-inflammatory and antioxidant properties proved its worth in the pharmaceutical industry. Its phytocompounds like gingerols modulate the biochemical pathways and specific biomarkers activated in chronic inflammation (Yusof, 2016). The antioxidant properties due to 6-gingerol protect cell membrane lipids during oxidation and scavenging free radicals (Grzanna, et al., 2005; Han et al., 2022). The most important constituents are 6-gingerol, 6-paradol, and shogaol and zingerone, which are mainly responsible for the anticancer properties of ginger (Shukla and Singh, 2007; Zhang et al., 2021b). The scientific literature supports the use of ginger in the treatment of a variety of ailments, including osteoarthritis, neurological disorders, rheumatoid arthritis, diabetes, respiratory distress, liver diseases, and primary dysmenorrhea (Mahomoodally et al., 2021; Kiyama, 2020; Huang, et al., 2019; Khan et al., 2016; Jafarzadeh and Nemati, 2018; Bhaskar et al., 2020).
| 2. Pharmaceutical potential of ginger | ▴Top |
The ginger constituents vary depending on origin and whether the rhizomes are fresh or dry. A ginger root is largely composed of carbohydrates, fats, proteins, fiber, vitamins, minerals, water, ash, and volatile oils. The phytocompounds observed in fresh ginger are zingerone, gingerol, zingiberene, β-sesquiphellandrene, shogaol, bisabolene (α-form), β-phellandrene, farnesene, 1, 4-cineole, citral, camphene, 6-paradol, curcumene, terpineol (α-form), borneol, β-elemene, zingiberenol, limonene, geraniol, linalool (Jolad, et al., 2005; Mbaveng and Kuete, 2017; Ali et al., 2008). Ginger is receiving attention for its clinical applications and pharmaceutical properties due to gingerol, shogaols, zingiberene, zingerone, paradols, etc. Among these phytocompounds, gingerols are the primary components of several bioactivities (Kubra and Rao, 2012). The structure of selected phytocompounds is shown in Figure 1.
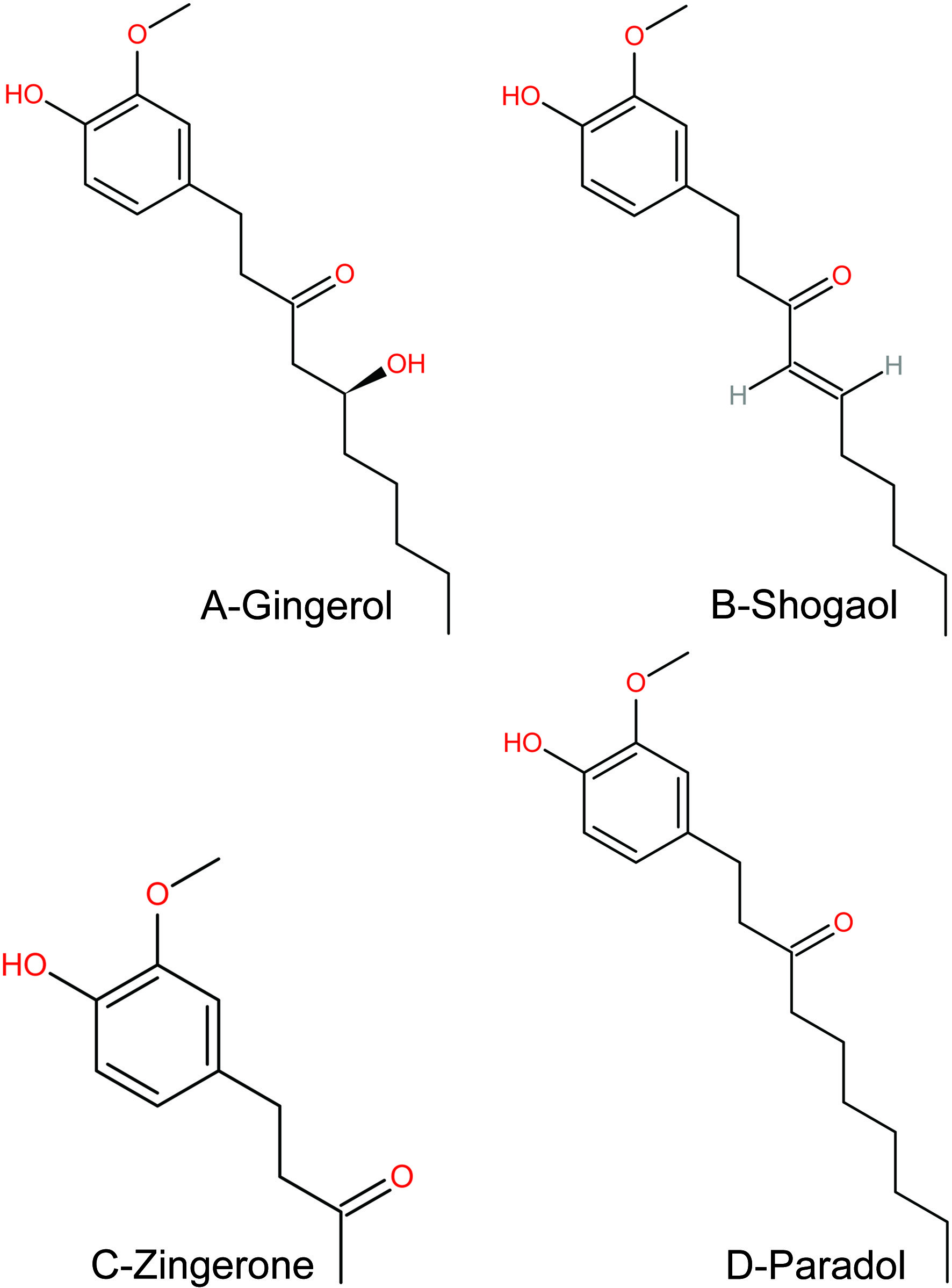 Click for large image | Figure 1. Chemical structure of important phytocompounds. |
The potential pharmacological activities of phytocompounds increase the significant investigations and clinical trials on the health benefits and ginger efficacy. In dry ginger, 6-gingerol transformed to 6-shogaol, which is more stable and has more powerful pharmacological effects. Further, 6-shogaol biotransformed to 6-Paradol (Jafarzadeh et al., 2021). The fiery taste of ginger is due to zingerone, the major phytochemical used for medicinal purposes. Ginger is also used as a primary food additive due to its fragrant and spicy characteristics (Srinivasan, 2017).
2.1. Gingerols
The gingerols are an important group of polyphenols isolated from the fresh root of ginger. Ginger rhizomes are one of the richest sources of ginger-derived bioactives (6-gingerol, 8-gingerol, and 10-gingerol). 6-Gingerol contains a 5-hydroxydecan-3-one moiety that is substituted by a 4-hydroxy-3-methoxyphenyl group at position 1; thought to inhibit adipogenesis. This beta-hydroxy ketone belongs to the group of plant metabolites known as guaiacols. It is an antineoplastic agent. It is rich in antioxidants, cardiotonics, and cardioprotective agents, including [8]-gingerol (Xue, et al., 2021b). Inhibition of leukotriene A (4) hydrolase expression and induction of G2/M arrest have been shown to be biochemical mechanisms by which 6-gingerol plays an anti-colorectal cancer role (Kumara et al., 2017).
2.2. Shogaol
Shogaols are bioactive compounds found in ginger that have gastroprotective and neuroprotective properties. [6]-Shogaol is a monomethoxybenzene, which belongs to the phenols and enones. Using a Mannich reaction, Mase and his colleagues synthesized shogaol using dimethylammonium dimethylcarbamate (DIMCARB), an ionic liquid (Mase et al., 2010). 6-Shogaol inhibits the infiltration of leukocytes into inflamed tissue, reducing edema swelling. 6-Shogaol affects pathways such as NFκB and MAPK, and regulates the cytoprotective HO-1 in both vitro and in vivo (Bischoff-Kont and Fürst, 2021). In addition to scavenging free radicals, the 6-, 8-, and 10-shogaols also exhibit strong anti-proliferative activity against human lung cancer cell lines (Sang et al., 2009). Previous studies suggest that 6-shogaol inhibits glial cell activation and has anti-oxidant properties against neurological disorders (Moon et al., 2014).
2.3. Paradols
[6]-paradol belongs to the class of phenols, a monomethoxybenzene, and ketone. It controls several obesity-related genes without triggering AMPK. 6-paradol decreases body weight gain and visceral and subcutaneous fat in mice after 2 weeks. It decreased liver cholesterol, triglyceride production, fatty acid synthesis, and lipid transport, as well as adipocyte differentiation, both in liver and adipose tissue (Hattori, et al., 2021). 6-shogaol is almost completely metabolized into 6-paradol, despite the fact that 6-paradol is only a minor component of ginger, mainly derived from 6-gingerol (Chen et al, 2012). According to a study evaluating the concentration-dependent effects of 6-paradol on glucose utilization, 6-paradol inhibits the synthesis of lipid in 3T3-L1 cells, reducing cellular lipid accumulation in a concentration-dependent manner, in addition to reducing insulin-induced lipid accumulation (Wei et al, 2017).
2.4. Zingerone
Zignerone is a 4-phenylbutan-2-one that contains a methoxy and hydroxy group at position 3 and 4 of the phenyl ring, respectively. It is responsible for the pungent flavor in ginger and may be used as an antioxidant, as an anti-inflammatory agent, as an antiemetic, as a flavouring, as a fragrance, and as a plant metabolite (Baliga, et al., 2011). Zingerone reduced oxidative stress and ameliorated inflammation, and levels of antioxidant enzymes. Oral administration was suggested as a treatment for rheumatoid arthritis. It significantly lowered levels of NF-κB, TGF-β, TNF-α, IL-1β, IL-6 and Hs-CRP while significantly increasing levels of IL-10 (Bashir et al., 2021). By regulating AMPK, Gingerone prevents hepatic inflammation, oxidative stress, and apoptosis (Mohammed, 2022).
2.5. Zingiberene
It is a sesquiterpene and a cyclohexadiene that contains a hydrogen at the 5 position that is substituted by a 6-methyl-hept-5-en-2-yl group (R configuration). It is found in dried ginger rhizomes, Zingiber officinale (indonesian ginger). Researchers observed that ginger rhizomes contained terpenes like zingiberene that inhibited MAO-A enzyme activity (Kukula-Koch et al., 2018). Experimental observations indicate that zingiberene can be exploited as a natural and novel therapeutic in preventing oxidative damage in neurodegenerative disorders (Togar et al., 2015a). Researchers have examined the cytotoxic, genotoxic, and antioxidant properties of zingiberene in an in vitro culture of rat brain cells and discovered that this compound could be potentially used as a natural anticancer agent (Togar et al., 2015b).
2.6. Zerumbone
Zerumbone is a sesquiterpene and cyclic ketone derived from (1E,4E,8E)-alpha-humulene, which can be obtained by steam distillation from ginger grown in Southeast Asia. As a plant metabolite, it is also useful as an anti-inflammatory drug for gliomas, as well as being an inhibitor of certain oncogenes (Rahman, et al., 2013). Researchers have found that zerumbone can prevent Zearalenone-induced liver injury and have presented the molecular basis for potential uses of zerumbone in treating liver injuries from Zearalenone (AbuZahra et al., 2021).
Recent studies have also shown that Zerumbone inhibits inflammation by inhibiting NF-κB and TLR. Zerumbone could potentially be used for the treatment and prevention of diabetes and its complications (Kim et al., 2022). Researchers found that zerumbone increased BAX, caspase-7, and caspase-9 expression and decreased BCL-2 expression, leading to apoptotic cell death induced by paclitaxel and proapoptotic proteins. By enhancing intracellular ROS-mediated oxidative stress, zerumbone promotes resensitization of breast cancer cells to PTX (Li et al., 2022).
| 3. Therapeutic potential | ▴Top |
A potential source of functional food additives for cancer prevention and treatment is ginger (Köngül and Şeker 2021). Gingerol is a phytocompound that is capable of resisting the proteases required for Coronavirus entry and replication (Oso, et al., 2020). Ginger extract reduces nausea and vomiting through inhibition of the cholinergic M3 receptors and the serotonergic 5-HT3 receptors associated with breast cancer (Pertz, et al., 2011). The effects of ginger on lipid profiles are noteworthy. Studies have shown that ginger has favorable effects on triacylglycerol (TAG) and low-density lipoprotein cholesterol (LDL-C) (Pourmasoumi et al., 2018; Jafarnejad, et al., 2017; Mazidi, et al., 2016). The phytocompounds in ginger extract and their associate diseases and disorders with molecular target are mentioned in Table 1.
 Click to view | Table 1. The phytocompounds in ginger extract and their associate diseases and disorders with molecular target |
3.1. In cardiovascular
The world’s leading cause of death is cardiovascular disease. Diabetes, high cholesterol, triglycerides, and high blood pressure causes these problems. Rat serum samples tested for the effects of ginger doses (500 mg/kg) revealed a significant reduction in cholesterol levels, but no change in triglycerides (Thomson et al., 2002). A study found a dose of 5 grams or more is required to show significant antiplatelet activity (Nicoll and Henein, 2009). Ginger inhibits reactive oxygen species (ROS), inducible nitric oxide synthase (iNOS), superoxide dismutase (SODs), glutathione, and heme oxygenase (Roudsari et al., 2021). 6-gingerol (20 μM) is a novel phytocompound from ginger that improves cardiac function and alleviates pressure overload-induced cardiac changes in a p38-dependent manner (Ma et al., 2021). 6-gingerol is a promising drug for the treatment of pathobiological cardiac remodeling (Xu et al., 2018; Lv et al., 2018; Ren et al., 2019). CVD inflammation triggers proinflammatory cytokines such as tumor necrosis factor-α (TNF-α), interleukin-1β (IL-1β), and interleukin-6 (IL-6). Studies have shown that ginger reduces TNF-α values, while 6-gingerol reduces inflammatory factors and nitric oxide synthase enzyme levels (Morvaridzadeh et al., 2020). The increase in evidence for Nrf2 and its downstream targets is likely responsible for preventing CVD development, including the development of endothelial dysfunction and atherosclerosis induced by oxidative stress. In the early stages of atherosclerosis, endothelium dysfunction is marked by an increase in blood flow, cellular permeability, LDL oxidation, monocyte adherence, platelet activation, vascular inflammation, and the proliferation and infiltration of smooth muscle cells from the media to the arteries (da Costa et al., 2019; Zhang et al., 2021a). The reduction of antioxidant enzyme protein expression was found in LDLR−/− mice models transplanted with NRF2-deficient bone marrow cells. Migratory macrophages, inflammatory cytokines, and atherosclerotic lesions were also observed after transplant of Nrf2-deficient bone marrow cells (Mimura and Itoh, 2015; Ooi et al., 2018). In the meantime, silencing Nrf2 in U937 monocytic cells led to elevations in proinflammatory cytokines such as interleukin-1β (IL-1β ), interleukin-6 (IL-6), tumour necrosis factor alpha (TNF-α), monocyte chemotactic protein-1 (MCP-1) and endoplasmic reticulum (ER) stress markers. Nrf2 deficiency also increased the production of pro-inflammatory cytokines such as (MCP-1, IL-6, and TNF-α) within macrophages, which led to foam cell formation (Ooi et al., 2018). A study found that bioactive molecules in ginger compounds inhibited the expression of Keap1, which resulted in a reduction in Nrf2, which subsequently elevated the levels of Nrf2 and other downstream antioxidant enzymes (Gao et al., 2022). Several other aspects of cardiovascular disease can also be treated with ginger. A study indicates that aqueous ginger extract lowers blood pressure and reduces heart palpitations by stimulating muscarinic receptors and blocking Ca++ channels (Ghayur, et al., 2005). A daily supplement of ginger in specific amounts has been found beneficial for heart health. Ginger’s extract exhibits moderate inhibitory properties against the Monoamine oxidase-A (MAO-A) enzyme, which is an effective antidepressant, as well as a panic disorder agent (Kukula-Koch et al., 2018). An investigation examined the cardioprotective effects of 8-Gingerol in isoproterenol (ISO)-induced MI. in rats ISO (85 mg/kg/d) was subcutaneously injected over 2 consecutive days to induce an acute MI model. There was an association between 8-Gin and relaxation of oxidative stress, cardiomyocyte apoptosis inhibition, and regulation of Ca2+ homeostasis through modulation of ICa-L (Xue et al., 2021a). Another study investigated the possible mechanisms by which 8-Gin (10 and 20 mg/kg/d) reduced J-point elevation and heart rate in mice induced with isoproterenol (ISO-10mg/kg/d) for 2 weeks. It was found that 8-Gin exerted cardioprotective effects on ISO-induced MF, which is likely associated with its inhibition of ROS generation, apoptosis, and autophagy by modulating the PI3K/Akt/mTOR signaling pathway (Xue et al., 2021b).
3.2. Anticancer agent
The term “cancer” refers to a group of disorders characterized by abnormal cell growth that may invade and spread. The cancer death rate is 15.7% (eighteen million deaths a year) (Wang, et al., 2016). A study demonstrated that 6-gingerol had anti-tumorigenic properties by affecting cell cycle regulation and apoptosis by increasing caspase activity during the G0/G1 phase of the cell cycle (Park et al., 2006; Wang et al., 2014). 8-gingerol is a derivative classified according to its ability to inhibit the growth of cancerous cells and blood vessel formation as well as its ability to suppress platelet growth. A further class of bioactive derivative is 10-gingerol, which decreases cell proliferation and inhibits cellular expression (Kubra and Rao, 2012; Zaid et al., 2022). In a pilot study that investigated ginger’s effects on proliferation, apoptosis, and differentiation in normal-appearing colonic mucosa; it was found that 2g of ginger reduced proliferation and increased apoptosis and differentiation relative to proliferation in the crypt differentiation zone (Citronberg, et al., 2013). Ginger aqueous extract contains polyphenols and flavonoids-flavonols that have anticancer properties (Choudhury, et al., 2010). In colorectal cancer (CRC), cyclooxygenase (COX)-produced prostaglandin E2 (PGE2) tissues increase, which is an early sign of the disease. The ginger root significantly reduced the expression of COX-1 protein, which is responsible for an increased risk of CRC. It shows that ginger has the potential to reduce eicosanoid levels and to reduce inflammation in the colon (Jiang, et al., 2013; Zick, et al., 2011). Ginger is a chemopreventive (Zick et al., 2015). Ginger has been found to have anti-skin-cancer chemopreventive effects. Ginger ethanol extract pre-applied to the skin of Sensitivity to carcinogenesis mice model significantly inhibited tissue plasminogen activator (TPA)-induced cyclooxygenase and lipoxygenase activities, ODC mRNA expression, and epidermal edema and hyperplasia (Katiyar, et al., 1996). Research has established that gingerol is able to scavenge the peroxyl radicals produced during pulse radiolysis (Park et al., 1998; Ahmad et al., 2001; Chauhan et al., 2021). 6-gingerol inhibits tumor growth and metastasis through the inhibition of cyclooxygenase-2 (COX-2) and the p38 MAPK-NF-κB pathway (mitogen-activated protein kinase-necrosis factor-kappa B) (Kim et al., 2005a; Kim et al., 2005b). The possible mechanisms of gingerol’s effects on treating liver cancer were studied by network pharmacology, in-silico experiments, as well as in-vitro experiments on human liver cancer HepG2 cells. Molecular docking revealed that gingerol derivatives had good PI3K and Akt binding activity. Researchers report that gingerol derivatives and compound gingerol (compound gingerol is composed of 6gingerol, 8gingerol, and 10gingerol in a ratio of 7:1.5:1.5) inhibit HepG2 cell proliferation, and each administration group can significantly increase the apoptosis rate of HepG2 cells and the fluorescence intensity of the nucleus, thereby blocking the cell cycle. In Western Blot and real-time quantitative PCR experiments, gingerol derivatives and compound gingerol had a significant effect on decreasing the expression of Akt and p-Akt and increasing the expression of Bax/Bcl-2 (Su, et al., 2022). Cancer can be prevented and treated with ginger. Clinical trials are needed to support the efficacy of the drug. In colon cancer, the signaling pathway PI3K-Akt involves the serine/threonine kinase Akt, as well as the genes AKT1, PIK3CA, MAPK3, and TP53. Human colon cancer is associated with a misregulation of the PI3K-Akt pathway, the most commonly activated signaling pathway in cancer (Wang et al., 2020b; Li et al., 2020). In the study, phosphoinositol 3-kinase signaling was found to be an active biological process in ginger in controlling cancer-related pathways through interactions with PI3K/Akt (Kiyama, 2020). Ginger’s anticancer mechanism is directly influenced by its PI3K-Akt signaling pathways and EGFR kinase inhibitor resistance (Zhang et al., 2021b).
3.3. Neurodegenerative agent
Several studies have shown ginger to be analgesic (Phillips et al., 1993; Black, et al., 2010; Rondanelli, et al., 2020). In Alzheimer’s disease (AD), abnormal aggregation of neurons and synaptic damage is caused by abnormal accumulation of amyloid beta (Aβ) and tau proteins (Busche and Hyman, 2020). Aβ is considered by many to be a key pathogen that contributes to AD pathogenesis, but the exact mechanisms are still unclear. Several studies have found protective effects of ginger on amyloid beta (Aβ) neurotoxicity. The ginger treatment inhibited the inflammatory markers NF-κB and interleukin (IL)-1β resulting in behavioral dysfunction and neuronal death caused by Aβ1–40 plaques as well as increasing antioxidant enzymes superoxide dismutase and catalase (SOD and CAT) (Choi, et al., 2018). There is evidence that 6-gingerone, 6-shogaol, 6-paradol, and dehydrozingerone may provide benefits in diseases related to aging and neurological disorders (Ma et al., 2021; Srinivasan et al., 2019). Researchers assessed acetylcholinesterase (AChE) as the most promising molecular target for ginger compounds with multiple molecular targets in Alzheimer’s disease (Azam, et al., 2014; Wheeler, 2003). Additionally, ginger compounds inhibit the activity of butyrylcholinesterase (BChE) by binding to Trp82 and Tyr332 residues (Cuya and França, 2020; Cuya et al., 2018; Schepici, et al., 2021). A reduction in clinical symptoms of mice with Experimental autoimmune encephalomyelitis (EAE) as well as a reduction of expression of IL-27 and IL-33 in their spinal cords is also observed (Jafarzadeh et al, 2014). In studies on migraine headaches, ginger was observed to have both abortive and prophylactic effects without causing any side effects (Mustafa and Srivastava, 1990). In a meta-analysis of placebo-controlled randomized controlled trials (RCTs), ginger was shown to be very effective and safe in treating migraine patients with pain outcomes as measured after two hours (Chen and Cai, 2021).
3.4. Antiviral
The antiviral and immune-modulating properties of herbal extracts have been extensively studied (Das, et al., 2021; Yasmin et al., 2020; Mukhtar, et al., 2008). Studies have reported that allicin from ginger has anti-influenza properties (Hornung, et al., 1994; Sahoo et al., 2016). The Dengue virus infection significantly enhanced matrix metalloproteinase (MMP)-2 proteolytic activity, but not matrix metalloproteinase-9. Ethylenediaminetetraacetic acid (EDTA-MMP inhibitor) reduced this enhancement. An aqueous extract from Zingiber officinale Roscoe was confirmed to modulate expression levels of MMP-2, MMP-9, TIMP-1, and TIMP-2 in a concentration-dependent manner. Gingerols and shogaols, which are naturally occurring compounds in Zingiber officinale, have been associated with its anti-MMP activity (Sharma, et al., 2015). In vitro, the anti-chikungunya effect of Zingiber officinale (62.50 μg/mL) aquatic plant extract was tested using the Vero cell line. Additionally, Zingiber officinale is used as an antiviral treatment against Chikungunya virus that prevents drug resistance (Kaushik, et al., 2020). Fresh ginger blocks the human respiratory syncytial virus (HRSV) (San Chang et al., 2013). Ginger chemicals as antiviral agents against a variety of viruses, as well as their mechanisms of action, Table 2.
 Click to view | Table 2. Ginger extract as an antiviral agent |
Recently, several studies have focused their attention on the interaction between ginger compounds and the spike protein of Coronaviruses. PLPro (papain-like protease) and 3CLPro (chymotrypsin-like protease) are essential for the survival of SARS-CoV-2 in humans (Li, et al., 2020). Natural products are being investigated as a potential targeted therapy to treat SARS-CoV-2. A structure-based molecular docking study found that ginger phytocompounds like 8-Gingerol, 10-Gingerol, and 6-Gingerol are potent inhibitors of PLpro, with high affinity for binding (Goswami, et al., 2020). Ginger components were shown to have high binding affinity to the protease (Mpro) of CoV-2, the virus that causes SARS (Jahan et al., 2021).
| 4. Conclusion | ▴Top |
The promising target profiles of natural compounds enable them to be considered as ideal drug development candidates since they contain signature events that are dispersed throughout. There are several compounds in ginger extract that have antiviral and antibiotic properties. Ginger extract can be used as a treatment for various diseases. Ginger is considered a natural alternative to treat cancer and other diseases due to its phytochemical properties and pharmacological effects. It is a popular healthy food because of its phytochemical properties and pharmacological effects. Ginger compounds can relieve fevers, reduce pain, and suppress coughs, making them helpful to minimize the effects of a winter cold. It has been shown to be effective in treating several ailments such as high blood pressure, cough, cold, swelling, nausea, rheumatoid disorders, vomiting, bronchitis, indigestion, gastric ulcers, infectious diseases, and poisonings. There is evidence that ginger is anti-inflammatory, anti-cancer, anti-tumor, anti-microbial, antiemetic, hepatoprotective, and neuroprotective. Clinical trials show that ginger can reduce nausea and vomiting in women during early pregnancy. Ginger inhibits proinflammatory cytokines, including nuclear factor kappa B (NF-κB) and immune system activation, as well as many other cellular processes. Ginger inhibits reactive oxygen species, inducible nitric oxide synthase, superoxide dismutase, glutathione, heme oxygenase, and inducible nitric oxide synthase. Research on ginger and its bioactive components, including both preclinical and clinical studies, is shown to have positive effects on cardiovascular disease, neurology, and cancer. These results reinforce the need for further In vivo and clinical studies.
Acknowledgments
None.
None.
Conflict of interest
The authors declare no conflict of interest.
| References | ▴Top |