| Journal of Food Bioactives, ISSN 2637-8752 print, 2637-8779 online |
| Journal website www.isnff-jfb.com |
Original Research
Volume 8, December 2019, pages 84-91
Comprehensive study of the lipid from whelk (Chlorostoma rusticum and Neverita didyma) with emphasis on characterization of phospholipid molecular species by shot-gun strategy
Zhong-Yuan Liua, #, Kai-Qi Ganga, #, Fa-Wen Yina, b, Hong-Kai Xiec, Liang Songa, b, Da-Yong Zhoua, b, *
aSchool of Food Science and Technology, Dalian Polytechnic University, Dalian, PR China, 116034
bNational Engineering Research Center of Seafood, Dalian, PR China, 116034
cBeijing Advanced Innovation Centre of Food Nutrition and Human Health, China Agricultural University, Beijing, China, 100083
#The authors contributed equally to this work.
*Corresponding author: Da-Yong Zhou, School of Food Science and Technology, Dalian Polytechnic University, Dalian, PR China, 116034. E-mail: zdyzf1@163.com
DOI: 10.31665/JFB.2019.8210
Received: December 20, 2019
Revised received & accepted: December 30, 2019
| Abstract | ▴Top |
In the present study, an effective shot-gun lipidomic methodology was established to determine the glycerophospholipid (GP) molecular species of two species of edible marine whelks (Chlorostoma rusticum and Neverita didyma). Simultaneously, the lipid content, lipid classes, phospholipid (PL) subclasses and fatty acid compositions were also investigated. Over 210 molecular species of GP including glycerophosphocholine, lysoglycerophosphocholine, glycerophosphoethanolamine, lysoglycerophosphoethanolamine, glycerophosphoserine, lysoglycerophosphoserine, glycerophosphoinositol and lysoglycerophosphoinositol were characterized in the two abovementioned whelk species. The predominant GP molecular species contained n-3 long chain polyunsaturated fatty acid (n-3 LC-PUFA), especially docosahexaenoic acid and eicosapentaenoic acid. Meanwhile, PL (57.70–58.86% of total lipids) and PUFA (21.69–37.68% of total FA) take large proportions in whelk lipids. Among PL, phosphatidylcholine (50.58–52.41 mol%) and phosphatidylethanolamine (27.67–32.73 mol%) were dominant. Therefore, marine whelks turn out to be promising source of n-3 LC-PUFA existed in PL form and thus directly contribute to the health benefits of consumer.
Keywords: Marine whelk; Lipid class composition; Phospholipid class composition; Phospholipid molecular species; n-3 LC-PUFA
| 1. Introduction | ▴Top |
Lipid is the main energy supply material for human body, which can provide more than twice the energy of sugar and protein in the metabolism (Barden and Decker, 2016). In addition, some fatty acids (FAs) are crucial for human body to remain healthy, especially n-3 long chain polyunsaturated fatty acids (n-3 LC-PUFAs) represented by docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), which play significant roles in various aspects toward human health such as cardiovascular protection, anti-inflammation, anti-hypertension, anti-allergies, brain development as well as renal function (Riediger et al., 2009). Therefore, it was recommended by many countries that a daily consumption of 250–1,000 mg of EPA/DHA provides health benefits (Kuratko and Salem, 2013).
Phospholipids (PLs), the major part of polar lipids in seafoods, have been reported to possess versatile biological function such as reducing cholesterol absorption, decreasing the risk of cardiovascular diseases, enhancing liver functions as well as brain function development (Ali et al., 2019; Arranz and Corredig, 2017). The current interest of PLs mainly comes from their effectiveness to incorporate n-3 LC-PUFAs to the cell membrane, as they exhibit better absorption and utilization than triglycerides (TAGs) (Cilla et al., 2016). Eggs, soybeans and fishes are main source of dietary PLs now, among which fishes are attracting more and more attention due to the abundant n-3 LC-PUFAs in PLs. In recent years, incremental studies have reported that marine shellfishes also are a rich source of PLs that contain n-3 LC-PUFAs. However, most of the published studies on characterization of PL molecular species of marine shellfish mainly concentrate on bivalve shellfish including oysters, clams, scallops, etc. (Boselli et al., 2012; Chen et al, 2012; Liu et al. 2017), but only few on univalve shellfish. To provide reliable nutritional and functional information and support for the better utilization of univalve shellfish, comprehensive characterization of lipid profiles in univalve shellfish is required.
Whelks are excellent candidate univalve shellfish for aquaculture and functional foods (Tang et al., 2016), among which Chlorostoma rusticum and Neverita didyma are the main edible species. An efficient shotgun lipidomic strategy was established in the present work to characterize the molecular species of glycerophospholipid (GP) including glycerophosphocholine (GPCho), lysoglycerophosphocholine (LGPCho), glycerophosphoethanolamine (GPEtn), lysoglycerophosphoethanolamine (LGPEtn), lycerophosphoserine (GPSer), lysoglycerophosphoserine (LGPSer), glycerophosphoinositol (GPIns), and lysoglycerophosphoinositol (LGPIns) in the two abovementioned species of whelks. Meanwhile, the lipid content and quality of lipid classes, PL subclasses and FA compositions were also investigated. This work will reveal the specific health benefits of lipids in whelks in detail as well as provide theoretical basis for utilization of whelk as a novel source of functional food.
| 2. Materials and methods | ▴Top |
2.1. Materials
Two species of marine edible whelks (Chlorostoma rusticum and Neverita didyma) that harvested in July were selected and purchased from a local market in Dalian, Liaoning, China. After husking by hand, about 800 g of meat were lyophilized with a 2KBTES-55 freeze-dryer (VirTis Co., Gardiner, NY, USA) for 70 h, crushed into a fine powder and stored at –30 °C for further use. GP standards of GPCho 12:0/12:0, GPEtn 12:0/12:0, GPSer 12:0/12:0 and GPIns 8:0/8:0 were purchased from Avanti Polar Lipids, INC (Alabaster, AL, USA). Deuterated chloroform (CDCl3), methanol (MeOD), triethyl phosphate (TEP), cesium carbonate (CsCO3) and deuterated water (D2O) were purchased from Aladdin Reagent Co., Ltd. (Shanghai, China). Other reagents were of analytical grade from Damao Chemical Reagent Co., Ltd. (Tianjin, China).
2.2. Lipid extraction
Total lipid from the two species of whelks Chlorostoma rusticum and Neverita didyma was extracted according to the widely adopted methyl tert-butyl ether (MTBE) method (Matyash et al., 2008), with little modification. Briefly, 10 g whelk powder sample was put in a 50 mL polytetrafluoroethylene tubes, and then an organic reagent mixture of methanol (15 mL) and MTBE (50 mL) was added to each sample. After stirring the mixture at 30 °C for 1 h, 12.5 mL of deionized water was added to the polytetrafluoroethylene tubes. Shaking the tubes for complete mixing, then the system was centrifuged at 7,800 g for 10 min. After collecting and transferring all the organic phase to a new glass, and then the rest was re-extracted following the same procedure. The organic phase was collected and evaporated under a tender nitrogen flow at 35 °C. The lipids from the two whelk materials were stored at –80 °C for less than 2 weeks for further analysis.
2.3. Determination of lipid class composition
An Iatroscan MK-6S thin layer chromatography-flame ionization detection (TLC-FID) Analyzer (Iatron Inc., Tokyo, Japan) was used to determine the lipid class composition according to Yin et al. (2015).
2.4. Determination of fatty acid composition
FA compositions were first derived with methanol to form FA methyl ester then determined with an Agilent 7890B GC-5977C MSD (Agilent, Palo Alto, CA, USA) equipped with HP-5-MS capillary column (30 m × 0.25 mm, 0.25 μm) (Agilent) according to the procedure previously described (Yin et al., 2015).
2.5. PL classes quantification by 31P NMR
The determination of PL classes was conducted with an Avance III 400 MHz NMR spectrometer (Bruker, Karlsruhe, Germany) according to one previous study (Gang et al., 2018).
2.6. Mass spectrometric analysis of whelk lipids
In this study, qualitative and quantitative of PL molecular species were achieved by using a hybrid API 4000 Qtrap (AB Sciex, Foster City, CA, USA) quadrupole-linear ion trap (QqLIT) mass spectrometer (MS) with a Turbo V electrospray ionization (ESI) source interface, and a computer platform equipped with HPLC-MS/MS Solution Analyst software 1.6.1 (AB Sciex, Foster City, CA, USA). Whelk lipids were dissolved in a mixture of chloroform and methanol (2:1, v/v), which contained 0.1% of formic acid to improve the ionization efficiency, and directly introduced into the ESI source though a syringe pump after filtering by a 0.22 µm organic membrane. The whelk lipid concentration was 50 μg/mL, and the flow rate for analysis of GPCho/LGPCho, GPEtn/LGPEtn, GPSer/LGPSer and GPIns/LGPIns was 2, 10, 10 and 20 µL/min, respectively. In characteristic scan mode, precursor-ion scanning (PIS) and neutral loss scanning (NLS) was operated in the positive ion mode for PL identification, while the enhanced product ion (EPI) scanning that conducted in the negative ion mode for FA explication. The instrument parameters were identical with our previous study (Liu et al., 2017).
The semi-quantitation of the same GP molecular species between lipids recovered from different whelk species was achieved by using an internal standard as previously described (Liu et al., 2017; Yin et al., 2016). In this study, four GP standards including GPCho 12:0/12:0 (inner standard for GPCho/LGPCho), GPEtn 12:0/12:0 (inner standard for GPEtn/LGPEtn), GPSer 12:0/12:0 (inner standard for GPSer/LGPSer) and GPIns 8:0/8:0 (inner standard for GPIns/LGPIns) were added to the samples with concentration of 1.00, 1.50, 0.15, and 0.15 µg/mL, respectively, before injection for MS analysis.
2.7. Statistical analysis
The experiments were conducted in triplicate. Data were presented as mean ± standard deviation (SD). The experimental data for statistical analysis was achieved by using SPSS 16.0 software (SPSS Inc., Chicago, IL, USA). Independent-samples T text was adopted to compare the differences between means. P values < 0.05 were considered significant.
| 3. Results and discussion | ▴Top |
3.1. Lipid content
The dried meat of Chlorostoma rusticum and Neverita didyma contained 13.67 and 7.12% lipids, respectively. Previous studies showed that the lipid contents of whelks Mytilus galloprovincial, Neptunea lyrata and Bulimulus dealbatus were 9.6, 10.8 and 7.3% (dry basis) (Miletic et al., 1991; Beach et al., 2009; Zarai et al., 2011), respectively, which indicate that the lipid contents varied in different species of whelks.
3.2. Lipid class compositions
As shown in Table 1, total lipids that extracted from the two species of whelks had similar lipid profiles. They were both consisted of PL, TAG, diacylglycerols (DAG), monoacylglycerols (MAG), free fatty acids (FFA) and cholesterols (CHO), among which PL (52.70–58.86% of total lipids) and TAG (31.91–32.01% of total lipids) constituted the major part of the lipids. Similarly, previous studies have also reported that PL was dominant in whelk lipids. For example, similar observations indicated PL accounted for 56.3 and 59.8% of total lipids, respectively, in whelks Turbo cornutus (Saito and Aono, 2014) and Haustrum scobina (Carrasco et al., 2016).
 Click to view | Table 1. Class compositions (%) of the lipids recovered from two species of whelks |
PLs, especially GPs, act as the principal structural constituents of cell or biological membranes. In the perception of lipid nutrition, it has been commonly accepted that PL are highly effective in delivering their FA residues for incorporation into the membranes and altered the FA composition of membrane PLs within specific cell type. Consequently, cellular functions including signaling and transport, as well as the membrane bound enzymes activity could be modulated or greatly affected by dietary PLs (Küllenberg et al., 2012). Therefore, dietary consumption of PLs contributes to a series of health benefits such as reducing the risk of cardiovascular disease, reducing blood cholesterol levels, reducing inflammatory reactions, enhancing the body’s immunity, preventing hepatic disorders, and improving brain function (Küllenberg et al., 2012; Restuccia et al., 2012). The current study implied that Chlorostoma rusticum and Neverita didyma contained a large amount of PL, which indicate their potential health benefits.
3.3. Phospholipid class composition
As demonstrated in Table 2, phosphocholine (PC), phosphoethanolamine (PE), phosphatidylinositol (PI), phosphatidylserine (PS), phosphatidic acid (PA), phosphatidylglycerol (PG) and lysophosphatidylcholine (LPC) were observed in the whelk lipids. Obviously, PC (50.58–52.41 mol%) and PE (27.67–32.73 mol%) were dominant. In contrast to PC and PE, LPC (3.20–8.86 mol%) and PG (3.76–11.05 mol%) just take a small proportion. However, PI (2.10 mol%), PS (4.81 mol%) and PA (2.81 mol%) were only detected in the lipid of Neverita didyma. Consist with this work, previous similar study also indicated that PC (62.81%) and PE (11.70%) were dominant PL classes of lipid in sea snail Chromodoris tinctoria (Zhukova, 2014).
 Click to view | Table 2. Phospholipid class compositions (mol%) of the lipids recovered from two species of whelks |
Different types of PLs vary in their headgroup, and therefore have different health effects. For example, PC, also known as lecithin, is the most abundant PL of all mammalian cell types and subcellular organelles, and is essential for brain land liver function, lipid metabolism and transport, cell membranes signaling, cell composition and repair (Asomaning et al., 2017); PE, which is the second most abundant PL in mammalian membranes, is also essential for membrane integrity and cell division (Calzada et al, 2016); PS, though a quantitatively minor membrane PL part, it is widely distributed throughout organelle membranes except endoplasmic reticulum, and it is required for human brain biochemistry, physiology, and function (Glade and Smith, 2015); PA, the simplest GP present in cells, is now a widely accepted second messenger for its important role in cellular signaling and membrane dynamics in all eukaryotes (Kooijman and Burger, 2009). With the diverse PL classes exist in the lipids in of Chlorostoma rusticum and Neverita didyma, they turn out to be potential diet for the diverse nutritional and healthy functions.
3.4. Fatty acid composition
The FA composition showed significantly difference between the two species. As shown in Table 3, the lipid from Chlorostoma rusticum included large level of monounsaturated fatty acids (MUFA) (43.10% of total FA), medium level of saturated fatty acids (SFA) (35.21% of total FA) and low level of PUFA (21.69% of total FA). Among PUFA, DHA and EPA only accounted for 0.40 and 3.17% of total FA, respectively. However, Neverita didyma had a proportion of PUFA (37.68% of total FA), MUFA (32.06% of total FA) and SFA (30.48% of total FA), which was close to the ratio of 1:1:1 that recommend by WHO. In addition, n-3 PUFA like DHA and EPA both accounted for over 11% of total FA.
 Click to view | Table 3. Fatty acid compositions (%) of the lipids recovered from two species of whelks |
Previous studies also have shown that FA profiles varied significantly among different species of whelks. For example, PUFA accounted for 44.8% of total FA in whelk Rapana Thomasiana, furthermore, DHA and EPA accounted for 5.68 and 7.77% of total FA, respectively (Christie et al., 1988). However, according to Saito and Aono (2014), lipid from sea snail Turbo cornutus contained a relative low percentage of PUFA (28.60%). Moreover, there was no DHA but low amount of EPA (3.20% of total FA) in Turbo cornutus lipid.
3.5. Characterization of glycerophospholipid molecular species
In the present study, GP molecular species were characterized with the shotgun lipidomic approach through character scan mode. After PIS scan with m/z 184 in positive ion mode for GPCho/LGPCho, NLS scan with m/z 141, 185 and 260 in positive ion mode for GPEtn/LGPEtn, GPSer/LGPSer and GPIns/LGPIns, respectively, the candidate compounds of each class of the afore-mentioned GP showed visible first-stage MS signals in the PIS or NLS MS spectra (Figure 1). The measured m/z value represent the molecular ion ([M]+) for GPCho/LGPCho, and the quasi-molecular ion ([M+H]+) of GPEtn/LGPEtn, GPSer/LGPSer and GPIns/LGPIns, respectively. Thus, the molecular mass of the unknown GP could be determined.
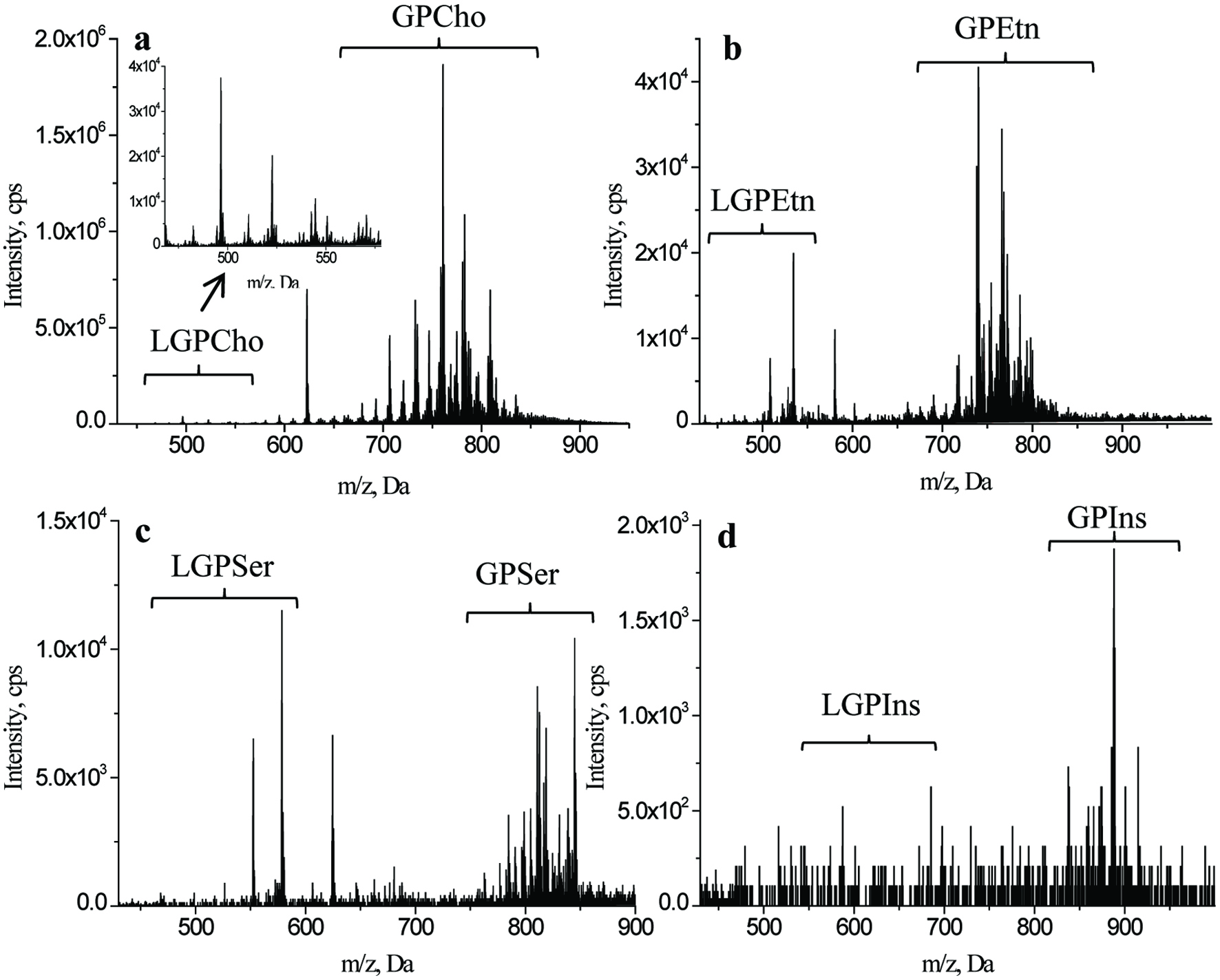 Click for large image | Figure 1. Specific detection of glycerophospholipids in lipids recovered from two species of whelks by using direct infusion mass spectrometric approaches. a-d, first-stage MS spectra of glycerophosphocholines (GPCho)/lysoglycerophosphocholines (LGPCho), glycerophosphoethanolamines (GPEtn)/lysoglycerophosphoethanolamines (LGPEtn), glycerophosphoserines (GPSer)/lysoglycerophosphoserines (LGPSer), and glycerophosphatidylinositol (GPIns)/lysoglycerophosphatidylinositol (LGPIns), respectively. |
Common structural for the GP molecular species follows the format x:y, in which x and y representing the number of carbons and double bonds of FA esterified in GP, respectively (Peterson and Cummings, 2006). For the lysoglycerophospholipid (LGP) molecular species, the structure could be distinguished directly according to the measured m/z by the formula established in our previous (Liu et al., 2017; Yin et al., 2016). Follow this procedure, the molecular species of all detected LGP in whelk lipids were characterized (Table s1).
For an unknown GP, which contain two FAs esterified to the sn-1 and sn-2 positions of the glycerol backbone, MS and MS/MS data are both essential for molecular structure elucidation. The total number of carbons and double bonds of the two FAs can be tentatively deduced according to the formulas we previously developed based on their measured molecular mass (Liu et al., 2017; Yin et al., 2016). After EPI scan in the negative ion mode, at least one FAs of the unknown GP can be ascertained by the FA anions ([RCOO]–) in MS/MS data acquired (Table s2). With the known information of the total number of carbons and double bonds of the two FAs, and one of the FA, the other FA of GP can be easily deduced. Moreover, other fragments such as lyso-GP anions due to the loss of FA were also observed in MS/MS data (Table s2), which further confirmed the reliability of characterization of molecular species. Furthermore, an empirical rule that the sn-2 of GP is the preferred position for the more unsaturated fatty acids was used to determine the distribution of the two FAs in the glycerol backbone (Napolitano et al., 1992). Through the above strategy, all detected phosphatidyl GPCho, GPEtn, GPSer and GPIns were characterized with unsaturated FA at sn-2 position. (Table s2).
The strategy for structural characterization of plasmalogen (plasmenyl and plasmanyl) GP was similar with that of phosphatidyl GP. However, it was hard to distinguish a plasmanyl GP x:y from a plasmenyl GP x:(y−1) for their same first-stage MS and MS/MS data due to the specific fatty acid fragment (ether-like anions, [RCO]–) of alkyl ether x:y and vinyl ether x:(y−1) that released from the sn-1 position of the plasmanyl and plasmenyl GP. Therefore, in the present work, a pair of possible structures including a plasmanyl GP x:y and a plasmenyl GP x:(y−1) corresponding to the same first-stage MS and MS/MS data were counted as one GP because they could not be differentiated (Table s2).
3.6. Glycerophospholipid molecular species in different whelk species
As shown in Tables 4, s3 and s4, more than 210 species of GP including GPCho, LGPCho, GPEtn, LGPEtn, GPSer, LGPSer, GPIns and LGPIns were characterized in lipids extracted from the two species of whelks. Or rather, at least 43 and 54 species of GPCho, 41 and 46 species of GPEtn, 27 and 36 species of GPSer, as well as 13 and 12 species of GPIns were characterized. Among them, 18:0/22:6, 16:0/18:1, 16:0/20:5 and 18:1/20:4 were the predominant species of the phosphatidyl subclass, 16:0/20:4 and 20:0/20:1 might be the predominant species of the plasmenyl subclass, 18:0/20:5, 16:0/22:6 and 20:0/20:1 might be the predominant species of the plasmanyl subclass (Table s4). In addition, for the lyso-GP, at least 14 and 21 species of LGPCho, 27 and 24 species of LGPEtn, 25 and 25 species of LGPSer, and 22 and 18 species of LGPIns were characterized from Chlorostoma rusticum and Neverita didyma, respectively (Tables 4, s1–s4). Among them, 18:1, 20:1, 20:5, 22:5 and 22:6 were the predominant species of the lyso-GP (Table 4).
 Click to view | Table 4. Major molecular species of glycerophosphocholines, glycerophosphoethanolamines, glycerophosphatidylserines, glycerophosphatidylinositols and their lyso- counterparts in lipids recovered from two species of whelks. The ratio of the intensity of the first-stage MS signal of a glycerophospholipid in the positive ion mode to that of the corresponding internal standard was used to represent its relative amount for comparison |
The analysis of FA compositions in PL has also proved that some classes of PL from whelk are abundant in PUFA. For example, Saito and Hashimoto (2010) reported that PC from black snail (Ifremeria nautilei) contained a large proportion of PUFA, especially EPA (8.70%), while Stuart et al (1998) reported that PE in snail Cepaea nemoralis was also rich in n-3 PUFA, particularly EPA (6.34%) and docosapentenoic acid (DPA) (8.24%). Meanwhile, some scholars devoted on PL molecular species in lipids from marine bivalves have also observed that the predominant species of GP contained PUFA. For example, the predominant GP species in lipids from mussel, clams and oysters normally contained 20:5 and 22:6 (Boselli et al., 2012; Chen et al., 2012; Liu et al., 2017; Yin et al., 2016). In this study, Chlorostoma rusticum and Neverita didymas had a large amount of PL molecular species that contained 20:5 and 22:6 in the structure. As described above, FA composition analysis indicated that there were no other 20:5 and 22:6 than EPA and DHA present in lipids form the whelk lipids. Therefore, the two whelks serve as potential source for PL enriched EPA and DHA.
It is well accepted that n-3 LC-PUFA, especially EPA, DPA and DHA, play significant roles in various aspects of human health, especially in reducing the risk of cardiovascular disease, hypertension, inflammation, allergies, immune, renal disorders and enhancing brain function (Riediger et al., 2009). Therefore, daily consumption of 250–1,000 mg of EPA/DHA for health benefit is widely recommended (Kuratko et al., 2013). The dietary sources of n-3 LC-PUFA are mainly existed in TAG and PL form, however, n-3 LC-PUFA in the PL form has captured extensive attention by consumers and scholars due to their higher bioavailability (Cook et al., 2016; Nicolson, 2014; Yurko-Mauro et al., 2015), tissue-delivery capacity (Antebi et al., 2004; Cansell, 2010; Liu et al., 2014) as well as health promoting effects (Ramprasath et al., 2013; Sampalis et al., 2003; Ulven et al., 2011) compared with n-3 LC-PUFA in TAG form. Therefore, the nutritional and healthy functions of whelk may partly be ascribed to the n-3 LC-PUFA enriched in PL.
| 4. Conclusion | ▴Top |
Dried whelks (Chlorostoma rusticum and Neverita didyma) tissues contained 13.67 and 7.12% of lipids, respectively. The lipids mainly consist of PL, TAG, DAG, MAG, FFA and CHO, among which PL (52.70–58.86% of total lipids) and TAG (31.91–32.01% of total lipids) were dominant. For PL, PC (50.58–52.41 mol%) was the major components. Compared with Chlorostoma rusticum, Neverita didyma had higher level of PUFA (37.68% of total FA), especially EPA (11.00% of total FA) and DHA (11.10% of total FA). At least 212 and 236 GP molecular species were characterized, respectively, in whelks Chlorostoma rusticum and Neverita didyma. Based on the amount of the molecular species containing EPA and DHA, Neverita didyma was the better fit species for GP. Obviously, whelks Chlorostoma rusticum and Neverita didyma are rich source of n-3 LC-PUFA existed in PL form. Therefore, the lipid quality of marine whelks may account for much of their nutritional and bioactive functions.
| Supporting information | ▴Top |
Table s1. Lysoglycerophosphocholines in lipids recovered from two species of whelks. The ratio of the intensity of the first-stage MS signal of a lysoglycerophosphocholine in the positive ion mode to that of the corresponding internal standard was used to represent its relative amount for comparison.
Table s2. Structural identification of glycerophosphocholines in whelk lipids according to first-stage MS and MS/MS data.
Table s3. Glycerophosphocholines in lipids recovered from two species of whelks. The ratio of the intensity of the first-stage MS signal of a glycerophosphocholine in the positive ion mode to that of the corresponding internal standard was used to represent its relative amount for comparison.
Table s4. The number of molecular species of glycerophosphocholines, glycerophosphoethanolamines, glycerophosphatidylserines, glycerophosphatidylinositols, lysoglycerophosphocholines, lysoglycerophosphoethanolamines, lysoglycerophosphatidylserines and lysoinglycerophosphatidylinositols in lipids recovered from two species of whelks.
Acknowledgments
This work was financially supported by “The National Natural Science Foundation of China (31871759; U1808203)”, “National Key R&D Program of China (2018YFD0901002)”, and “Project of Distinguished Professor of Liaoning Province (2015-153)”.
| References | ▴Top |